AcidBase Equilibria Chapter 16 Models of AcidBase Behavior

Acid-Base Equilibria Chapter 16

Models of Acid-Base Behavior

1. Arrhenius Acids and Bases n Arrhenius Acid –produce H+ n HCl(aq) H+ + Cln Arrhenius Base- produce OH+ n Na. OH(aq) Na + OH

2. Bronsted-Lowry Acids and Bases n B-L acid-base rxns involve a + transfer of a proton (H ) n B-L Acid = donates H+ n B-L Base = accepts H+
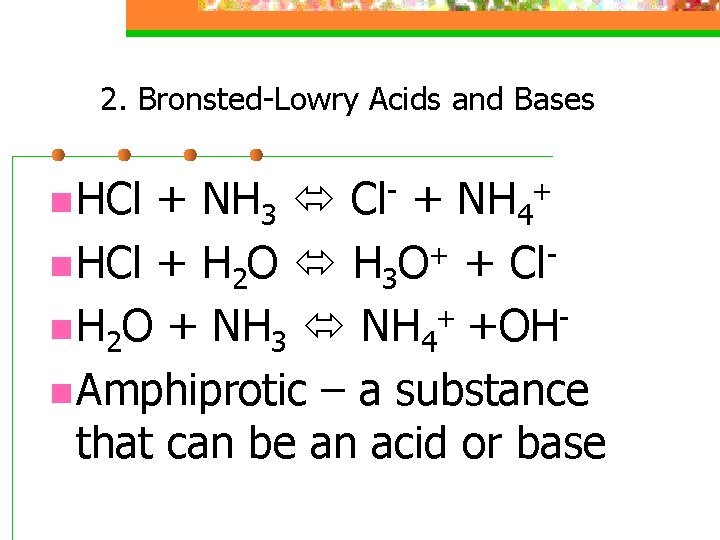
2. Bronsted-Lowry Acids and Bases n HCl + NH 3 Cl- + NH 4+ n HCl + H 2 O H 3 O+ + Cl+ n H 2 O + NH 3 NH 4 +OH n Amphiprotic – a substance that can be an acid or base

2. Bronsted-Lowry Acids and Bases conjugate acid-base pairs differ only by a proton

Sample Exercise 16. 1 Identifying Conjugate Acids and Bases (a) What is the conjugate base of each of the following acids: HCl. O 4, H 2 S, PH 4+, HCO 3–? (b) What is the conjugate acid of each of the following bases: CN-, SO 42–, H 2 O, HCO 3– ?

Sample Exercise 16. 2 Writing Equations for Proton-Transfer Reactions The hydrogen sulfite ion (HSO 3–) is amphiprotic. (a) Write an equation for the reaction of HSO 3– with water, in which the ion acts as an acid (b) Write an equation for the reaction of HSO 3– with water, in which the ion acts as a base.

The Autoionization of Water n acid n The base ion product of water, Kw=[H 3 O+][OH-] = 1 x 10 -14
![The Autoionization of Water n n n [H+] =[OH-] is neutral [H+] > [OH-] The Autoionization of Water n n n [H+] =[OH-] is neutral [H+] > [OH-]](http://slidetodoc.com/presentation_image/2e806d8ccf59afd7b31c3abc927b9d7a/image-10.jpg)
The Autoionization of Water n n n [H+] =[OH-] is neutral [H+] > [OH-] is acidic [H+] < [OH-] is basic
![Sample Exercise 16. 5 Calculating [H+] from [OH-] n Calculate the concentration of H+(aq) Sample Exercise 16. 5 Calculating [H+] from [OH-] n Calculate the concentration of H+(aq)](http://slidetodoc.com/presentation_image/2e806d8ccf59afd7b31c3abc927b9d7a/image-11.jpg)
Sample Exercise 16. 5 Calculating [H+] from [OH-] n Calculate the concentration of H+(aq) in (a) a solution in which [OH–] is 0. 010 M, (b) a solution in which [OH–] is 1. 8 × 10– 9 M.

3. Lewis Acids and Bases n. A lewis acid is an electron pair acceptor n A lewis base is an electron pair donor n Doesn’t pertain to just H+
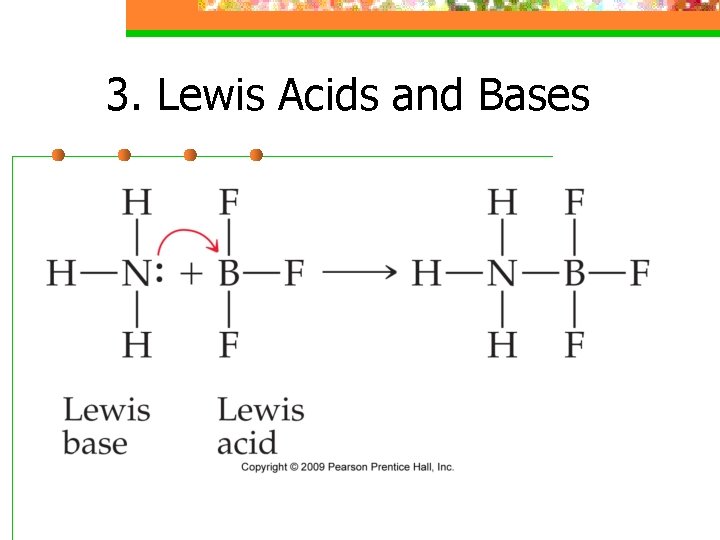
3. Lewis Acids and Bases

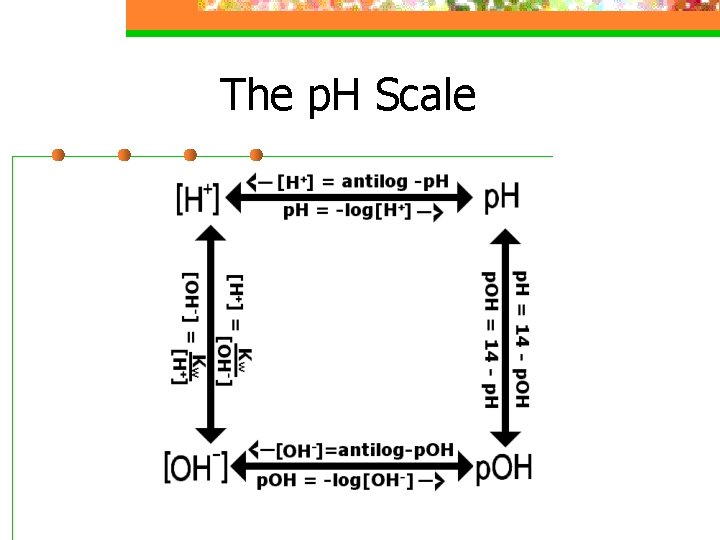
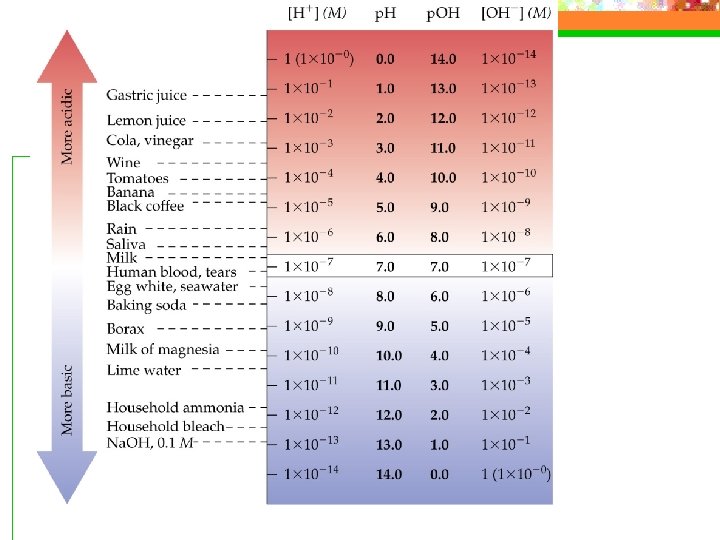
The p. H Scale n 0 ---- 7 ---- 14 Acid Neutral Basic
![The p. H Scale --- key equations n p. H = -log[H+] n [H+]=10 The p. H Scale --- key equations n p. H = -log[H+] n [H+]=10](http://slidetodoc.com/presentation_image/2e806d8ccf59afd7b31c3abc927b9d7a/image-15.jpg)
The p. H Scale --- key equations n p. H = -log[H+] n [H+]=10 -p. H n The p. H decreases as [H+] increases n p. OH = -log[OH-] n [OH-]=10 -p. OH n p. H + p. OH = 14
The p. H Scale

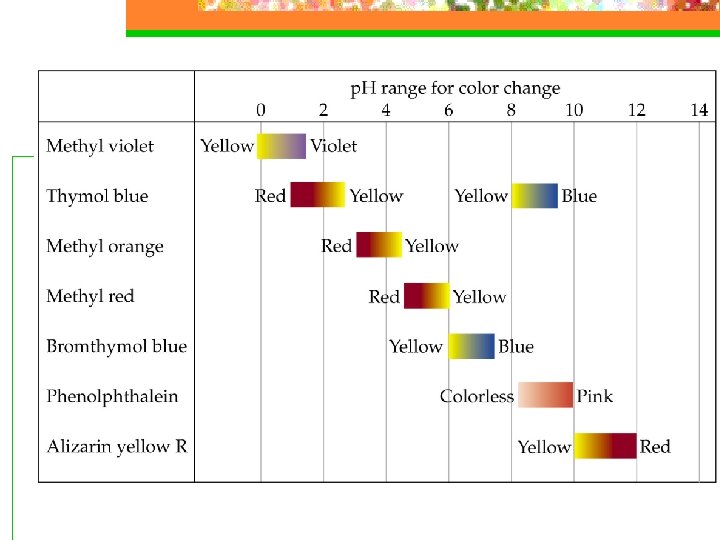
The p. H Scale n Measuring p. H n 1. p. H meters n 2. Titration and Acid-Base Indicators-colored substance that can exist as an acid or base, each having its own color


p. H meter simulation http: //group. chem. iastate. edu/Greenbo we/sections/projectfolder/flashfiles/acid basep. H/ph_meter. html n Acid-Base Solutions http: //phet. colorado. edu/en/simulation/ac id-base-solutions p. H Scale http: //phet. colorado. edu/en/simulation/ph n
![Sample Exercise 16. 7 Calculating [H+] from p. H A sample of freshly pressed Sample Exercise 16. 7 Calculating [H+] from p. H A sample of freshly pressed](http://slidetodoc.com/presentation_image/2e806d8ccf59afd7b31c3abc927b9d7a/image-22.jpg)
Sample Exercise 16. 7 Calculating [H+] from p. H A sample of freshly pressed apple juice has a p. H of 3. 76. Calculate [H+], [OH-], and p. OH.

Strong Acids and Bases n 7 strong acids ? ? ? n Strong acids are strong electrolytes and completely ionize in aqueous soln

Strong Acids and Bases n In aqueous solutions of + strong acids, the [H ] equals the original concentration of acid, calculate the p. H from [H+]

Sample Exercise 16. 8 Calculating the p. H of a Strong Acid What is the p. H of a 0. 040 M solution of HCl. O 4?

Strong Acids and Bases n Strong bases ? ? ? n Strong bases are strong electrolytes and completely ionize in aqueous soln
![Strong Acids and Bases n In aqueous solns of strong bases, the [OH-] depends Strong Acids and Bases n In aqueous solns of strong bases, the [OH-] depends](http://slidetodoc.com/presentation_image/2e806d8ccf59afd7b31c3abc927b9d7a/image-27.jpg)
Strong Acids and Bases n In aqueous solns of strong bases, the [OH-] depends on the coefficient of the equation n Calculate p. H from p. OH

Sample Exercise 16. 9 Calculating the p. H of a Strong Base What is the p. H of (a) a 0. 028 M solution of Na. OH (b) a 0. 0011 M solution of Ca(OH)2?

Weak Acids n weak acids are partially ionized which is represented as an equilibrium reaction n HA + H 2 O H 3 O+ + An or HA H+ + A-

Weak Acids n Ka is the acid-dissociation constant and the magnitude of it indicates the tendency of the acid to ionize. n The larger the Ka, the stronger the acid.

Weak Acids n It is possible to calculate Ka from p. H

Sample Exercise 16. 10 Calculating Ka from Measured p. H A student prepared a 0. 10 M solution of formic acid (HCOOH) and measured its p. H. The p. H at 25 ºC was found to be 2. 38. Calculate Ka formic acid at this temperature.

Weak Acids n Or you can calculate p. H from Ka n What is the p. H of 0. 30 M -5 HC 2 H 3 O 2 ? (Ka = 1. 8 x 10 )

Sample Exercise 16. 11 Calculating Percent Ionization A 0. 10 M solution of formic acid (HCOOH) contains 4. 2 × 10– 3 M H+(aq). Calculate the percentage of the acid that is ionized.

Warm up: Sample Exercise 16. 12 Using Ka to Calculate p. H (1) Calculate the p. H of a 0. 20 M solution of HCN. (Ka = 4. 9 x 10 -10. ) (2) Calculate the p. H of 0. 20 M HNO 3.

Sample Exercise 16. 13 Using Ka to Calculate Percent Ionization n Calculate the percentage of HF molecules ionized in (a) a 0. 10 M HF solution, (b) a 0. 010 M HF solution

Weak Acids n Polyprotic Acids have more than one ionizable H atom n It is always easier to remove the first proton from a polyprotic acid than the second n Ex. H 2 CO 3

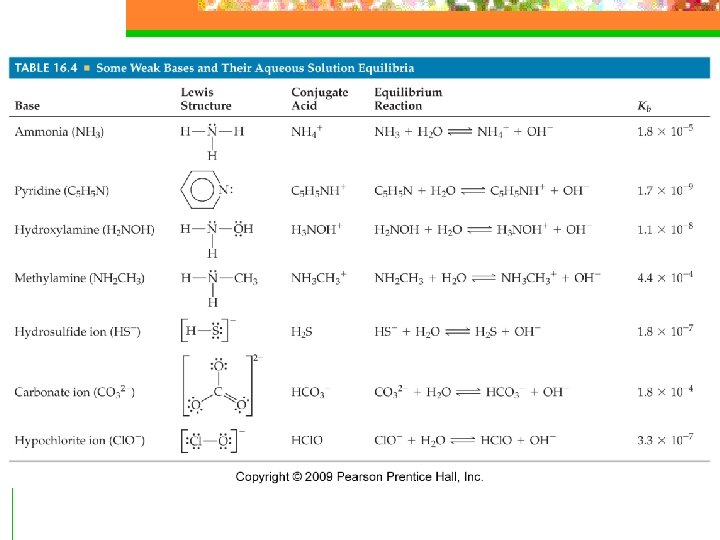
Weak Bases n Weak bases react with water by taking away a proton forming the conjugate acid of the base and OH- ions + n B + H 2 O BH + OH

Weak Bases n Types of Weak Bases n 1. Neutral substances that have an atom with a nonbonding pair of electrons that can serve as a proton acceptor (substances w/N) 2. Anions of weak acids ; conjugate base of weak acids
Weak Bases
Weak Bases

Sample Exercise 16. 15 Using Kb to Calculate OH¯ Calculate the p. H of a 0. 15 M solution of NH 3. Kb = 1. 8 x 10 -5

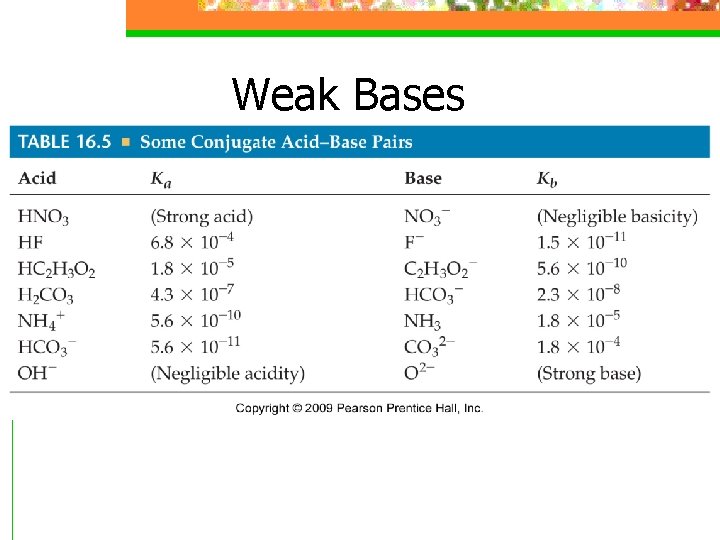
Relationship between Ka and Kb n Ka x Kb=Kw = 1. 0 x 10 -14 n p. Ka = -log. Ka n p. Kb=-log Kb n p. Ka + p. Kb = p. Kw = 14. 00 at 25 o. C

Relationship between Ka and Kb n As the strength of an acid increases (larger Ka), the strength of its conjugate base decreases (smaller Kb) n Or vice versa

Sample Exercise 16. 17 Calculating Ka or Kb for a Conjugate Acid -Base Pair Calculate (a) the base-dissociation constant, Kb, for the fluoride ion (F–); (b) the acid dissociation constant, Ka, for the ammonium ion (NH 4+).

Acid-Base Properties of Salt Solutions n salt solutions can be acidic, neutral, or basic n the p. H of a salt solution can be predicted by the ions that make up the salt

Acid-Base Properties of Salt Solutions n General Rules n 1. An anion that is the conjugate base of a SA will not affect the p. H of a solution (Cl )

Acid-Base Properties of Salt Solutions n 2. An anion that is the conjugate base of a WA will increase the p. H (CN )

Acid-Base Properties of Salt Solutions n 3. A cation of a SB will not + affect p. H (Na )

Acid-Base Properties of Salt Solutions n 4. A cation that is the conjugate acid of a WB will cause a decrease in p. H (NH 4+)

Acid-Base Properties of Salt Solutions n 5. With the exception of ions of group 1 A and heavier members of 2 A, metal ions will cause a decrease in p. H

Acid-Base Properties of Salt Solutions n 6. When a soln contains both the conj. base of a WA and the conj. acid of a WB, the ion with the largest Keq will have the greatest influence on p. H

Sample Exercise 16. 18 Determining Whether Salt Solutions Are Acidic, Basic, or Neutral Determine whether aqueous solutions of each of the following salts will be acidic, basic, or neutral: (a) Ba(CH 3 COO)2 (b) NH 4 Cl (c) CH 3 NH 3 Br (d) KNO 3 (e) Al(Cl. O 4)3.

Practice Exercise 16. 18 Determining Whether Salt Solutions Are Acidic, Basic, or Neutral In each of the following, indicate which salt in each of the following pairs will form the more acidic (or less basic) 0. 010 M solution: (a) Na. NO 3 or Fe(NO 3)3 (b) KBr or KBr. O; (c) CH 3 NH 3 Cl or Ba. Cl 2 (d) NH 4 NO 2 or NH 4 NO 3.

Acid-Base Behavior and Chemical Structure n Acidity increases with stronger attractions for central atoms electron pairs

Acid-Base Behavior and Chemical Structure n 1. Ionic charge n More positive ions are stronger acids Na+ < Ca 2+ < Al 3+ n Negative ions are weaker acids PO 43 - < HPO 42 - < H 2 PO 4 - < H 3 PO 4

Acid-Base Behavior and Chemical Structure n 2. Oxidation Number n For similar formulas, greater oxidation number, the stronger the acid HCl. O < HCl. O 2 < HCl. O 3 < HCl. O 4 H 2 SO 3 < H 2 SO 4

Acid-Base Behavior and Chemical Structure n 3. Electronegativity n For similar formulas with different central atoms, greater electronegativity, stronger acid H 2 CO 3 < H 2 SO 3 < HNO 3

Titration simulation n http: //group. chem. iastate. edu/Greenbo we/sections/projectfolder/flashfiles/stoic hiometry/acid_base. html n http: //chemilp. net/lab. Techniques/Acid. Base. Idicator. S imulation. htm n
- Slides: 59