AcidBase Equilibria and Solubility Equilibria Chapter 16 Copyright

Acid-Base Equilibria and Solubility Equilibria Chapter 16 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

The common ion effect is the shift in equilibrium caused by the addition of a compound having an ion in common with the dissolved substance. The presence of a common ion suppresses the ionization of a weak acid or a weak base. Consider mixture of CH 3 COONa (strong electrolyte) and CH 3 COOH (weak acid). CH 3 COONa (s) Na+ (aq) + CH 3 COO- (aq) CH 3 COOH (aq) H+ (aq) + CH 3 COO- (aq) common ion 16. 2
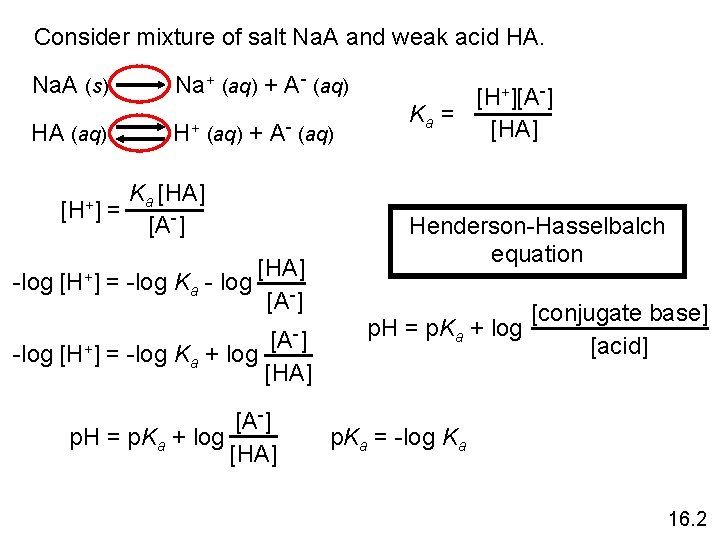
Consider mixture of salt Na. A and weak acid HA. Na. A (s) Na+ (aq) + A- (aq) HA (aq) H+ (aq) + A- (aq) [H+] Ka [HA] = [A-] -log [H+] = -log Ka - log [HA] [A-] -] [A -log [H+] = -log Ka + log [HA] [A-] p. H = p. Ka + log [HA] [H+][A-] Ka = [HA] Henderson-Hasselbalch equation p. H = p. Ka + log [conjugate base] [acid] p. Ka = -log Ka 16. 2

What is the p. H of a solution containing 0. 30 M HCOOH and 0. 52 M HCOOK? Mixture of weak acid and conjugate base! HCOOH (aq) Initial (M) Change (M) Equilibrium (M) Common ion effect 0. 30 – x 0. 30 0. 52 + x 0. 52 H+ (aq) + HCOO- (aq) 0. 30 0. 00 0. 52 -x +x +x 0. 30 - x x 0. 52 + x [HCOO-] p. H = p. Ka + log [HCOOH] [0. 52] = 4. 01 p. H = 3. 77 + log [0. 30] HCOOH p. Ka = 3. 77 16. 2

A buffer solution is a solution of: 1. A weak acid or a weak base and 2. The salt of the weak acid or weak base Both must be present! A buffer solution has the ability to resist changes in p. H upon the addition of small amounts of either acid or base. Consider an equal molar mixture of CH 3 COOH and CH 3 COONa Add strong acid H+ (aq) + CH 3 COO- (aq) Add strong base OH- (aq) + CH 3 COOH (aq) CH 3 COO- (aq) + H 2 O (l) 16. 3
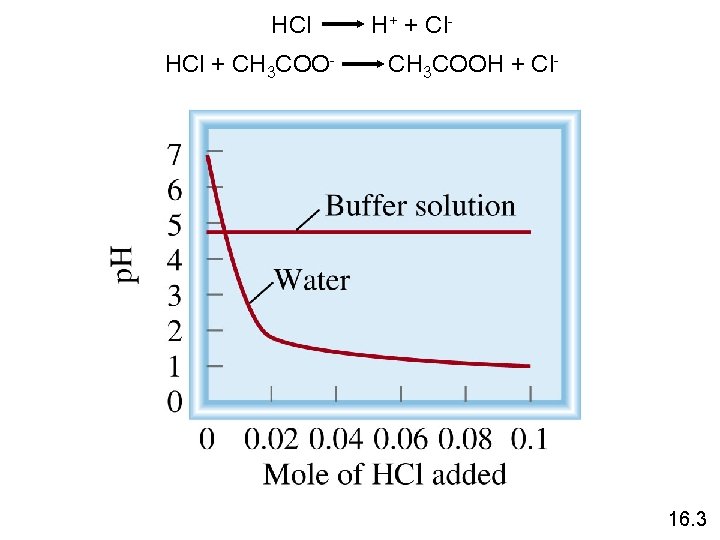
HCl + CH 3 COO- H+ + Cl. CH 3 COOH + Cl- 16. 3

Which of the following are buffer systems? (a) KF/HF (b) KBr/HBr, (c) Na 2 CO 3/Na. HCO 3 (a) KF is a weak acid and F- is its conjugate base buffer solution (b) HBr is a strong acid not a buffer solution (c) CO 32 - is a weak base and HCO 3 - is its conjugate acid buffer solution 16. 3

Calculate the p. H of the 0. 30 M NH 3/0. 36 M NH 4 Cl buffer system. What is the p. H after the addition of 20. 0 m. L of 0. 050 M Na. OH to 80. 0 m. L of the buffer solution? NH 4+ (aq) [NH 3] p. H = p. Ka + log [NH 4+] start (moles) end (moles) H+ (aq) + NH 3 (aq) p. Ka = 9. 25 0. 029 0. 001 NH 4+ (aq) + OH- (aq) 0. 028 0. 0 [0. 30] p. H = 9. 25 + log = 9. 17 [0. 36] 0. 024 H 2 O (l) + NH 3 (aq) 0. 025 final volume = 80. 0 m. L + 20. 0 m. L = 100 m. L [NH 4 +] 0. 028 0. 025 = [NH 3] = 0. 10 [0. 25] p. H = 9. 25 + log = 9. 20 [0. 28] 16. 3
![Solubility Equilibria Ag. Cl (s) Ksp = [Ag+][Cl-] Mg. F 2 (s) Ag 2 Solubility Equilibria Ag. Cl (s) Ksp = [Ag+][Cl-] Mg. F 2 (s) Ag 2](http://slidetodoc.com/presentation_image_h2/6ee8bbfaf0e6805d9d0438389858cf85/image-9.jpg)
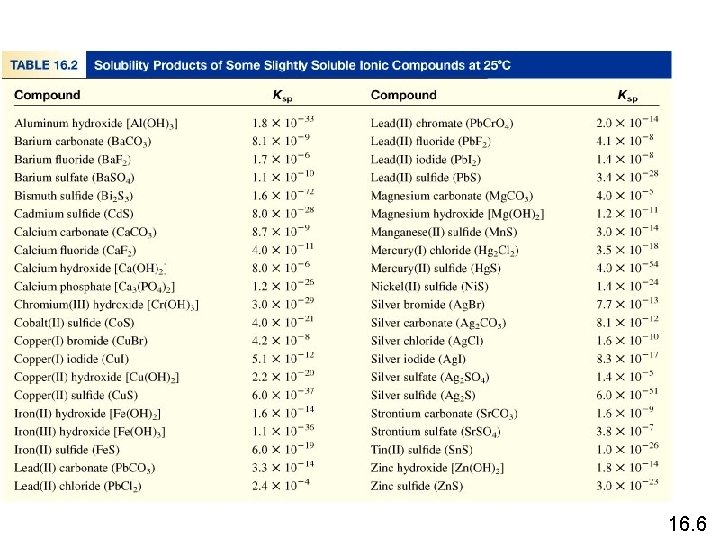
Solubility Equilibria Ag. Cl (s) Ksp = [Ag+][Cl-] Mg. F 2 (s) Ag 2 CO 3 (s) Ca 3(PO 4)2 (s) Ag+ (aq) + Cl- (aq) Ksp is the solubility product constant Mg 2+ (aq) + 2 F- (aq) Ksp = [Mg 2+][F-]2 2 Ag+ (aq) + CO 32 - (aq) Ksp = [Ag+]2[CO 32 -] 3 Ca 2+ (aq) + 2 PO 43 - (aq) Ksp = [Ca 2+]3[PO 43 -]2 Dissolution of an ionic solid in aqueous solution: Q < Ksp Unsaturated solution Q = Ksp Saturated solution Q > Ksp Supersaturated solution No precipitate Precipitate will form 16. 6
16. 6
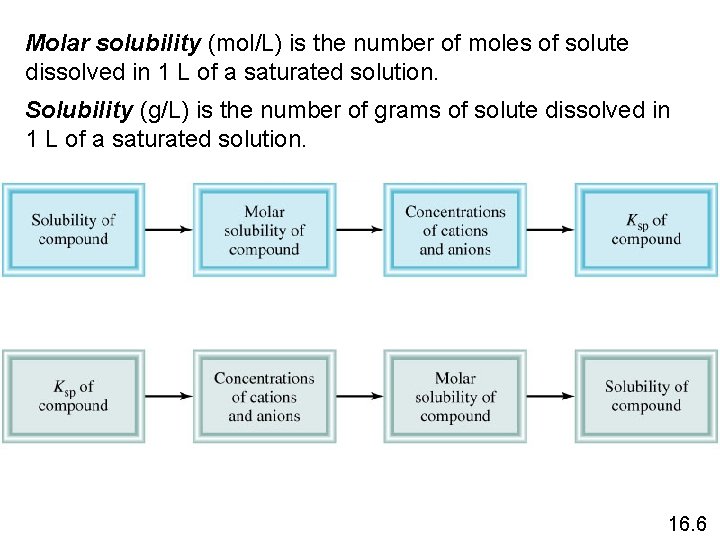
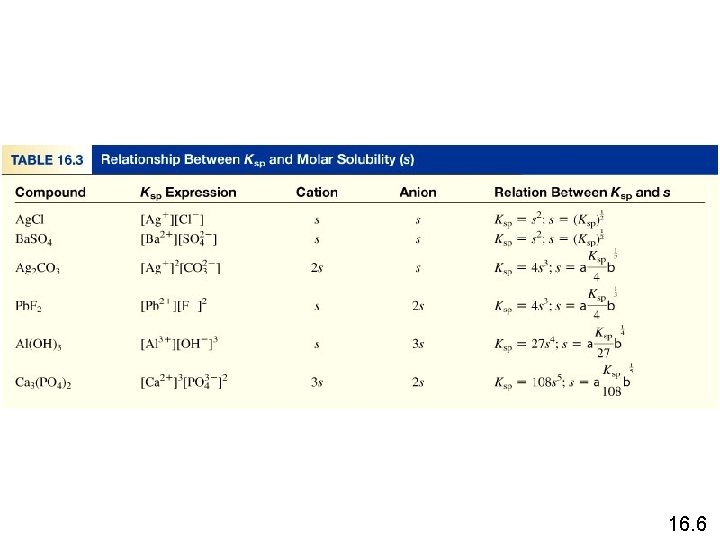
Molar solubility (mol/L) is the number of moles of solute dissolved in 1 L of a saturated solution. Solubility (g/L) is the number of grams of solute dissolved in 1 L of a saturated solution. 16. 6

What is the solubility of silver chloride in g/L ? Ag. Cl (s) Initial (M) Change (M) Equilibrium (M) [Ag+] = 1. 3 x 10 -5 M Ag+ (aq) + Cl- (aq) 0. 00 +s +s s s [Cl-] = 1. 3 x 10 -5 M Ksp = 1. 6 x 10 -10 Ksp = [Ag+][Cl-] Ksp = s 2 s = Ksp s = 1. 3 x 10 -5 mol Ag. Cl 143. 35 g Ag. Cl Solubility of Ag. Cl = x = 1. 9 x 10 -3 g/L 1 L soln 1 mol Ag. Cl 16. 6
16. 6

If 2. 00 m. L of 0. 200 M Na. OH are added to 1. 00 L of 0. 100 M Ca. Cl 2, will a precipitate form? The ions present in solution are Na+, OH-, Ca 2+, Cl-. Only possible precipitate is Ca(OH)2 (solubility rules). Is Q > Ksp for Ca(OH)2? [Ca 2+]0 = 0. 100 M [OH-]0 = 4. 0 x 10 -4 M Q = [Ca 2+]0[OH-]02 = 0. 10 x (4. 0 x 10 -4)2 = 1. 6 x 10 -8 Ksp = [Ca 2+][OH-]2 = 8. 0 x 10 -6 Q < Ksp No precipitate will form 16. 6

What concentration of Ag is required to precipitate ONLY Ag. Br in a solution that contains both Br- and Cl- at a concentration of 0. 02 M? Ag. Br (s) Ag+ (aq) + Br- (aq) Ksp = 7. 7 x 10 -13 Ksp = [Ag+][Br-] -13 K 7. 7 x 10 sp -11 M = = 3. 9 x 10 [Ag+] = 0. 020 [Br-] Ag. Cl (s) [Ag+] Ag+ (aq) + Cl- (aq) Ksp = 1. 6 x 10 -10 Ksp = [Ag+][Cl-] Ksp 1. 6 x 10 -10 -9 M = = 8. 0 x 10 = 0. 020 [Cl-] 3. 9 x 10 -11 M < [Ag+] < 8. 0 x 10 -9 M 16. 7

The Common Ion Effect and Solubility The presence of a common ion decreases the solubility of the salt. What is the molar solubility of Ag. Br in (a) pure water and (b) 0. 0010 M Na. Br? Na. Br (s) Na+ (aq) + Br- (aq) Ag. Br (s) Ag+ (aq) + Br- (aq) [Br-] = 0. 0010 M Ksp = 7. 7 x 10 -13 Ag. Br (s) Ag+ (aq) + Br- (aq) s 2 = Ksp [Ag+] = s s = 8. 8 x 10 -7 [Br-] = 0. 0010 + s 0. 0010 Ksp = 0. 0010 x s s = 7. 7 x 10 -10 16. 8
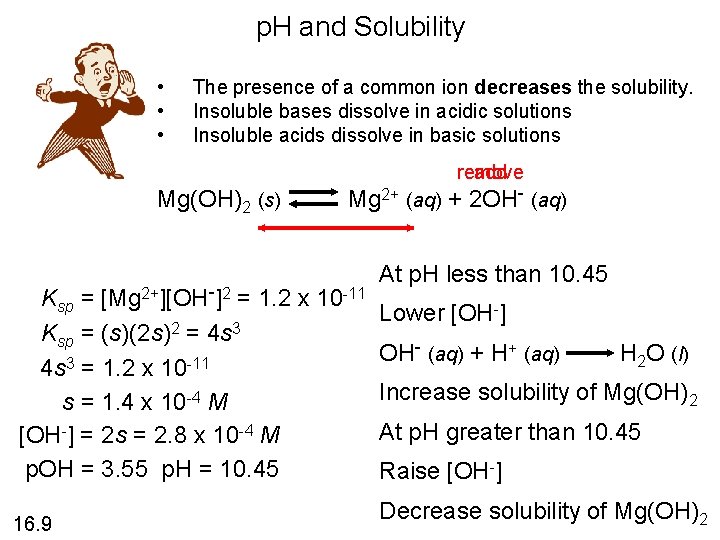
p. H and Solubility • • • The presence of a common ion decreases the solubility. Insoluble bases dissolve in acidic solutions Insoluble acids dissolve in basic solutions add remove Mg(OH)2 (s) Mg 2+ (aq) + 2 OH- (aq) Ksp = [Mg 2+][OH-]2 = 1. 2 x 10 -11 Ksp = (s)(2 s)2 = 4 s 3 = 1. 2 x 10 -11 s = 1. 4 x 10 -4 M [OH-] = 2 s = 2. 8 x 10 -4 M p. OH = 3. 55 p. H = 10. 45 16. 9 At p. H less than 10. 45 Lower [OH-] OH- (aq) + H+ (aq) H 2 O (l) Increase solubility of Mg(OH)2 At p. H greater than 10. 45 Raise [OH-] Decrease solubility of Mg(OH)2
- Slides: 17