ACIDBASE DISORDERS dr Husnil Kadri M Kes Biochemistry

ACID-BASE DISORDERS dr. Husnil Kadri, M. Kes Biochemistry Departement Medical Faculty Of Andalas University Padang
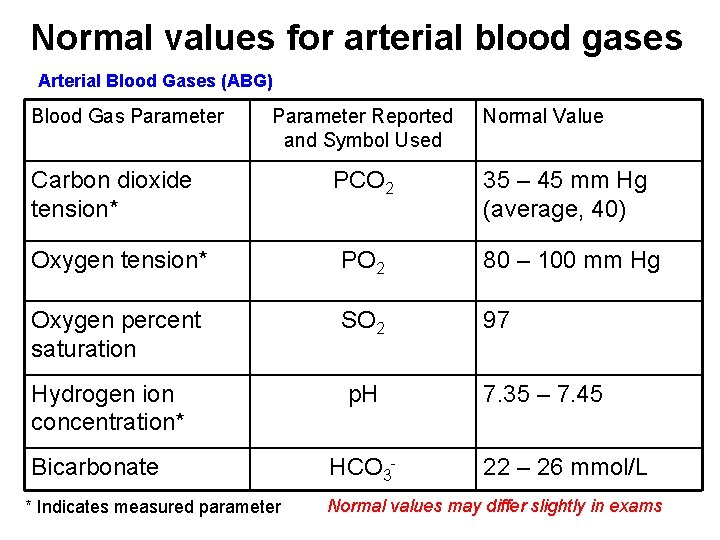
Normal values for arterial blood gases Arterial Blood Gases (ABG) Blood Gas Parameter Reported and Symbol Used Normal Value Carbon dioxide tension* PCO 2 35 – 45 mm Hg (average, 40) Oxygen tension* PO 2 80 – 100 mm Hg Oxygen percent saturation SO 2 97 Hydrogen ion concentration* p. H 7. 35 – 7. 45 Bicarbonate * Indicates measured parameter HCO 3 - 22 – 26 mmol/L Normal values may differ slightly in exams

GANGGUAN KESEIMBANGAN ASAM-BASA TRADISIONAL DISORDER p. H PRIMER RESPON KOMPENSASI ASIDOSIS METABOLIK HCO 3 - p. CO 2 ALKALOSIS METABOLIK HCO 3 - p. CO 2 ASIDOSIS RESPIRATORI p. CO 2 HCO 3 - ALKALOSIS RESPIRATORI p. CO 2 HCO 3 -

Normal Compensatory Response • Any primary disturbance in acid-base homeostasis invokes a normal compensatory response. • A primary metabolic disorder leads to respiratory compensation, and a primary respiratory disorder leads to an acute metabolic response due to the buffering capacity of body fluids. • A more chronic compensation (1 -2 days) due to alterations in renal function.

Mixed Acid - Base Disorder • Most acid-base disorders result from a single primary disturbance with the normal physiologic compensatory response and are called simple acid-base disorders. • In certain cases, however, particularly in seriously ill patients, two or more different primary disorders may occur simultaneously, resulting in a mixed acid-base disorder. • The net effect of mixed disorders may be additive (eg, metabolic acidosis and respiratory acidosis) and result in extreme alteration of p. H; • or they may be opposite (eg, metabolic acidosis and respiratory alkalosis) and nullify each other’s effects on the p. H.

KLASIFIKASI GANGGUAN KESEIMBANGAN ASAM BASA BERDASARKAN PRINSIP STEWART Fencl V, Jabor A, Kazda A, Figge J. Diagnosis of metabolic acid-base disturbances in critically ill patients. Am J Respir Crit Care Med 2000 Dec; 162(6): 2246 -51
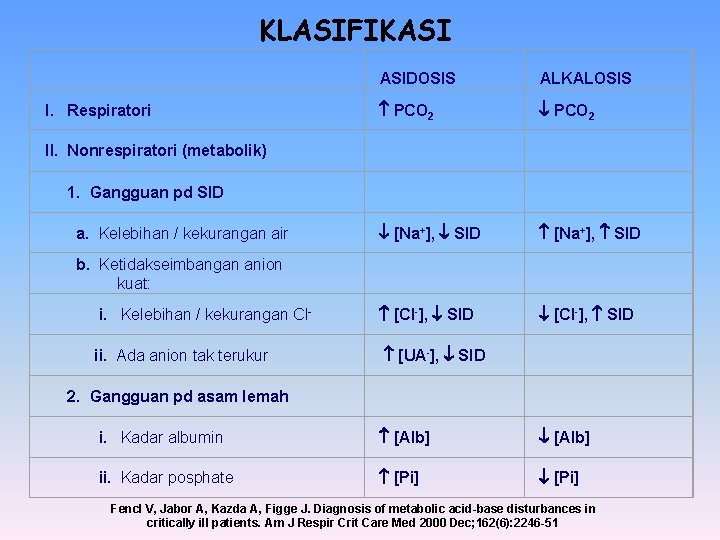
KLASIFIKASI I. Respiratori II. Nonrespiratori (metabolik) 1. Gangguan pd SID a. Kelebihan / kekurangan air b. Ketidakseimbangan anion kuat: i. Kelebihan / kekurangan Cl ii. Ada anion tak terukur 2. Gangguan pd asam lemah i. Kadar albumin ii. Kadar posphate ASIDOSIS ALKALOSIS PCO 2 [Na+], SID [Cl-], SID [UA-], SID [Alb] [Pi] Fencl V, Jabor A, Kazda A, Figge J. Diagnosis of metabolic acid-base disturbances in critically ill patients. Am J Respir Crit Care Med 2000 Dec; 162(6): 2246 -51
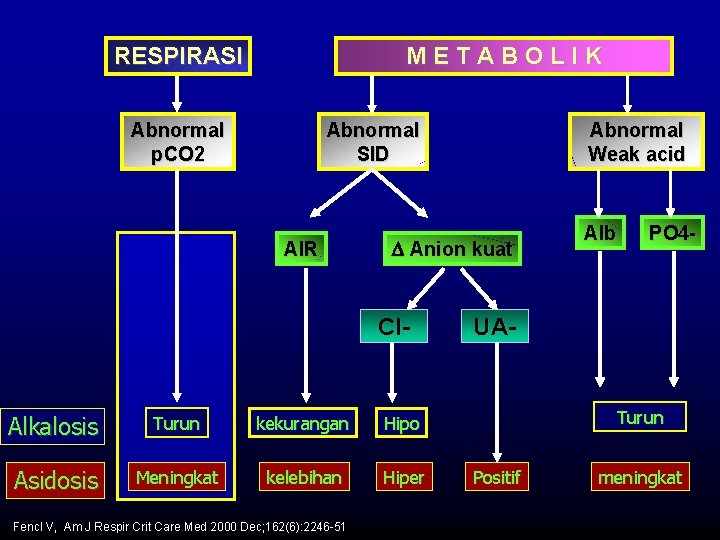
RESPIRASI METABOLIK Abnormal p. CO 2 Abnormal Weak acid Abnormal SID AIR Anion kuat Cl- Alkalosis Turun kekurangan Hipo Asidosis Meningkat kelebihan Hiper Fencl V, Am J Respir Crit Care Med 2000 Dec; 162(6): 2246 -51 Alb PO 4 - UA- Turun Positif meningkat
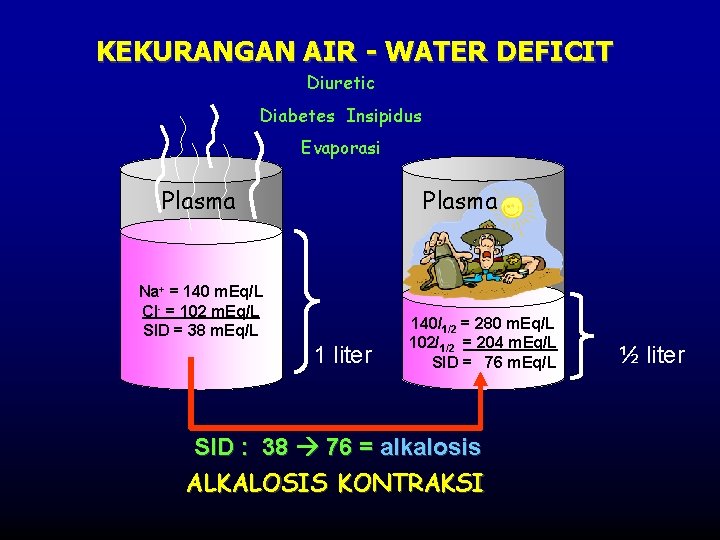
KEKURANGAN AIR - WATER DEFICIT Diuretic Diabetes Insipidus Evaporasi Plasma Na+ = 140 m. Eq/L Cl- = 102 m. Eq/L SID = 38 m. Eq/L 1 liter 140/1/2 = 280 m. Eq/L 102/1/2 = 204 m. Eq/L SID = 76 m. Eq/L SID : 38 76 = alkalosis ALKALOSIS KONTRAKSI ½ liter
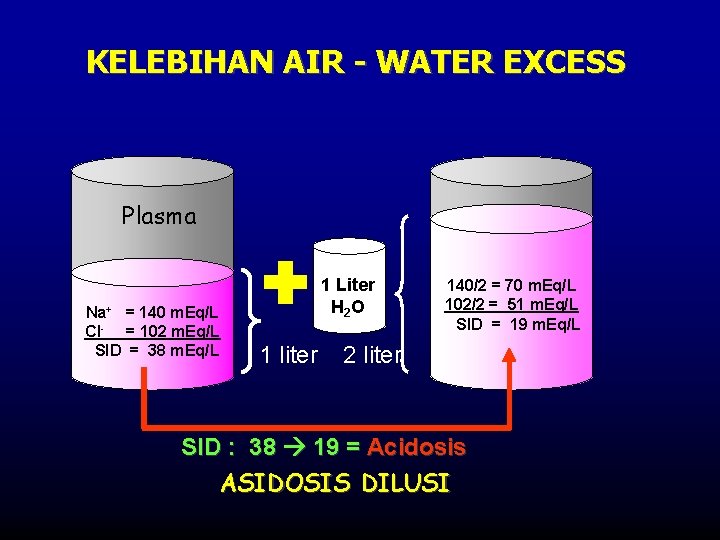
KELEBIHAN AIR - WATER EXCESS Plasma Na+ = 140 m. Eq/L Cl- = 102 m. Eq/L SID = 38 m. Eq/L 1 Liter H 2 O 1 liter 140/2 = 70 m. Eq/L 102/2 = 51 m. Eq/L SID = 19 m. Eq/L 2 liter SID : 38 19 = Acidosis ASIDOSIS DILUSI
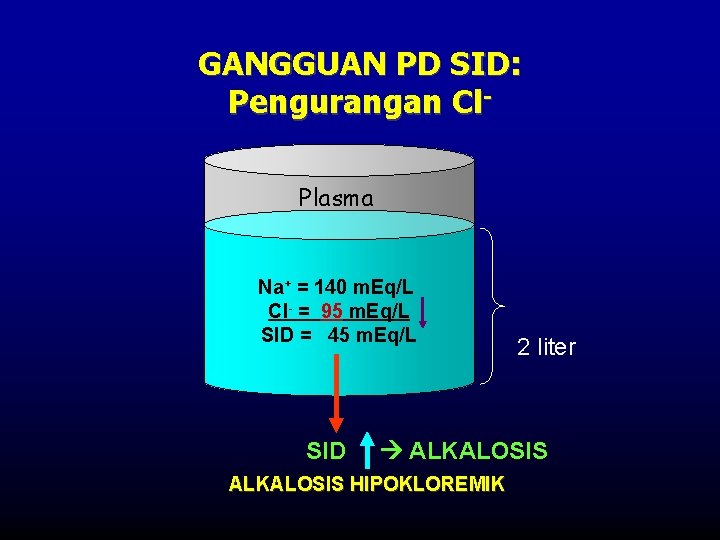
GANGGUAN PD SID: Pengurangan Cl. Plasma Na+ = 140 m. Eq/L Cl- = 95 m. Eq/L SID = 45 m. Eq/L SID 2 liter ALKALOSIS HIPOKLOREMIK
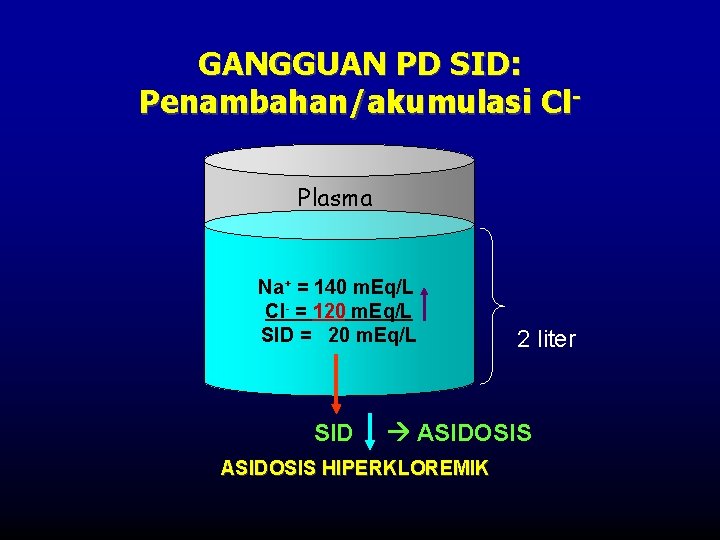
GANGGUAN PD SID: Penambahan/akumulasi Cl. Plasma Na+ = 140 m. Eq/L Cl- = 120 m. Eq/L SID = 20 m. Eq/L SID 2 liter ASIDOSIS HIPERKLOREMIK
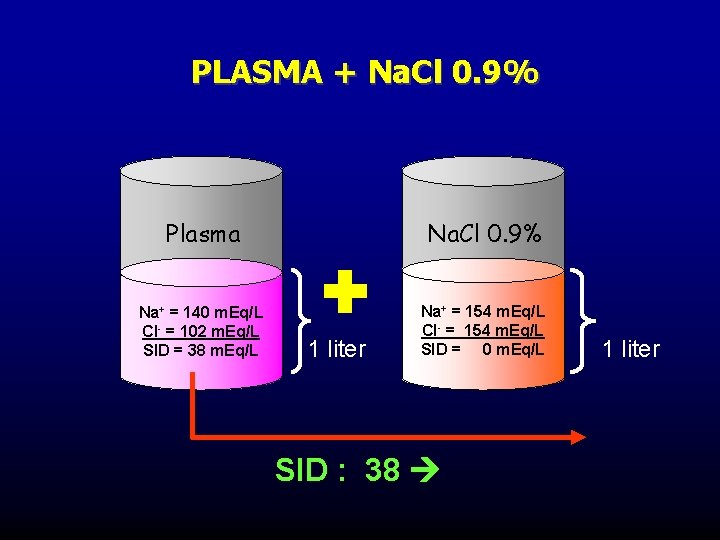
PLASMA + Na. Cl 0. 9% Plasma Na. Cl 0. 9% Na+ = 140 m. Eq/L Cl- = 102 m. Eq/L SID = 38 m. Eq/L Na+ = 154 m. Eq/L Cl- = 154 m. Eq/L SID = 0 m. Eq/L 1 liter SID : 38 1 liter
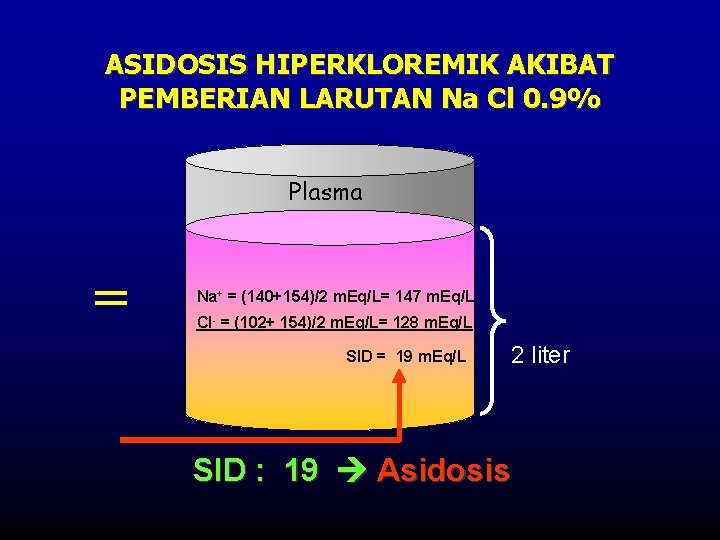
ASIDOSIS HIPERKLOREMIK AKIBAT PEMBERIAN LARUTAN Na Cl 0. 9% Plasma = Na+ = (140+154)/2 m. Eq/L= 147 m. Eq/L Cl- = (102+ 154)/2 m. Eq/L= 128 m. Eq/L SID = 19 m. Eq/L 2 liter SID : 19 Asidosis
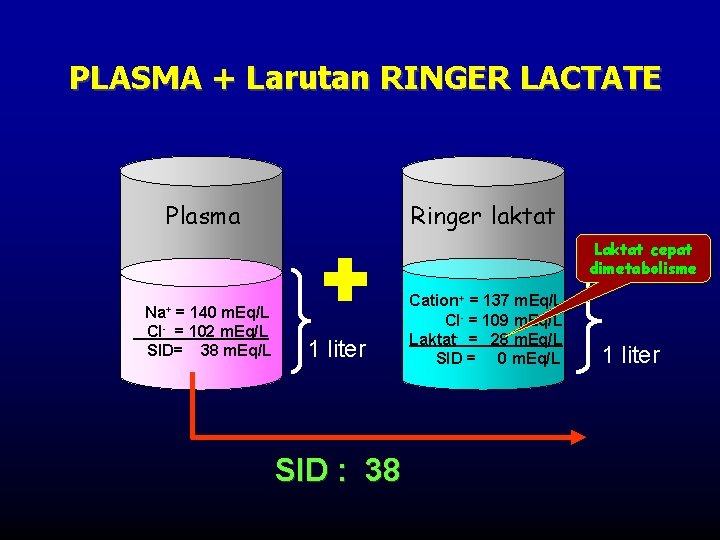
PLASMA + Larutan RINGER LACTATE Plasma Ringer laktat Laktat cepat dimetabolisme Na+ = 140 m. Eq/L Cl- = 102 m. Eq/L SID= 38 m. Eq/L 1 liter SID : 38 Cation+ = 137 m. Eq/L Cl- = 109 m. Eq/L Laktat- = 28 m. Eq/L SID = 0 m. Eq/L 1 liter
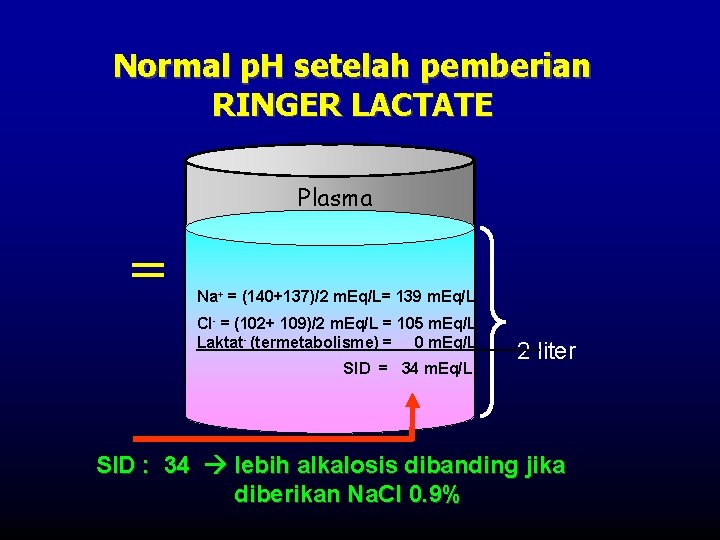
Normal p. H setelah pemberian RINGER LACTATE Plasma = Na+ = (140+137)/2 m. Eq/L= 139 m. Eq/L Cl- = (102+ 109)/2 m. Eq/L = 105 m. Eq/L Laktat- (termetabolisme) = 0 m. Eq/L SID = 34 m. Eq/L 2 liter SID : 34 lebih alkalosis dibanding jika diberikan Na. Cl 0. 9%
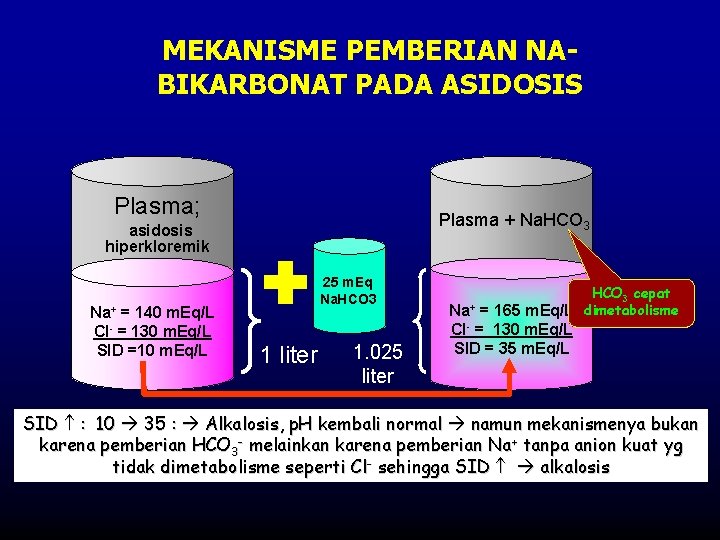
MEKANISME PEMBERIAN NABIKARBONAT PADA ASIDOSIS Plasma; asidosis Plasma + Na. HCO 3 hiperkloremik Na+ = 140 m. Eq/L Cl- = 130 m. Eq/L SID =10 m. Eq/L 25 m. Eq Na. HCO 3 1 liter 1. 025 liter Na+ HCO 3 cepat = 165 m. Eq/L dimetabolisme Cl- = 130 m. Eq/L SID = 35 m. Eq/L SID : 10 35 : Alkalosis, p. H kembali normal namun mekanismenya bukan karena pemberian HCO 3 - melainkan karena pemberian Na+ tanpa anion kuat yg tidak dimetabolisme seperti Cl- sehingga SID alkalosis

UA = Unmeasured Anion: Laktat, acetoacetate, salisilat, metanol dll. K K HCO 3 - SID HCO 3 - Keto. A- A- Na+ SID Na+ Cl- Normal Cl. Lactic/Keto asidosis Ketosis
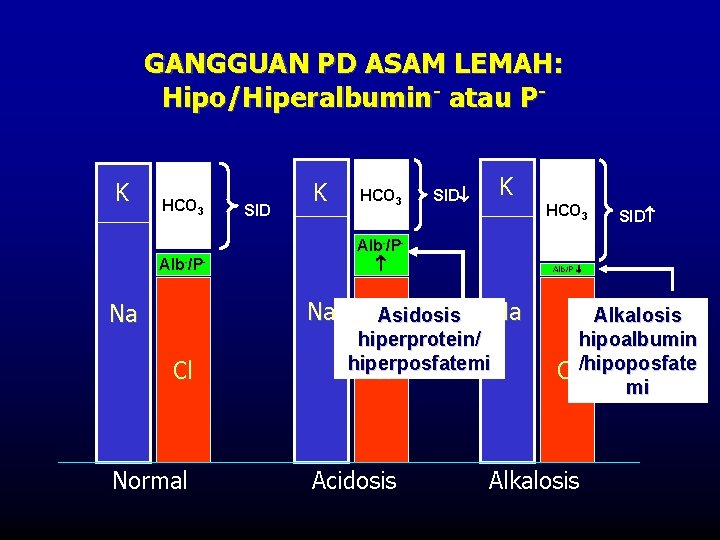
GANGGUAN PD ASAM LEMAH: Hipo/Hiperalbumin- atau PK HCO 3 SID K Na Cl Normal SID K HCO 3 Alb-/P- Na HCO 3 Alb/P Na Asidosis hiperprotein/ hiperposfatemi Cl Acidosis SID Alkalosis hipoalbumin Cl /hipoposfate mi Alkalosis

• Calculate the anion gap. • Anion gap = Na+ - (Cl- + HCO 3 -). • Normal anion gap is 8 -15 m. Eq/L.

If the anion gap is elevated • Then compare the changes from normal between the anion gap and [HCO 3 -]. • If the change in the anion gap is greater than the change in the [HCO 3 -] from normal, then a metabolic alkalosis is present in addition to a gap metabolic acidosis. • If the change in the anion gap is less than the change in the [HCO 3 -] from normal, then a non gap metabolic acidosis is present in addition to a gap metabolic acidosis.

Anion Gap Acidosis: • Anion gap >12 m. Eq/L; caused by a decrease in [HCO 3 -] • balanced by an increase in an unmeasured acid ion from either endogenous production or exogenous ingestion (normochloremic acidosis).

Non anion Gap Acidosis: • Anion gap = 8 -12 m. Eq/L; caused by a decrease in [HCO 3 -] balanced by an increase in chloride (hyperchloremic acidosis). Renal tubular acidosis is a type of non gap acidosis • The anion gap is helpful in identifying metabolic gap acidosis, non gap acidosis, mixed metabolic gap and non gap acidosis. If an elevated anion gap is present, a closer look at the anion gap and the bicarbonate helps differentiate among (a) a pure metabolic gap acidosis (b) a metabolic non gap acidosis (c) mixed metabolic gap and non gap acidosis, and (d) a metabolic gap acidosis and metabolic alkalosis.
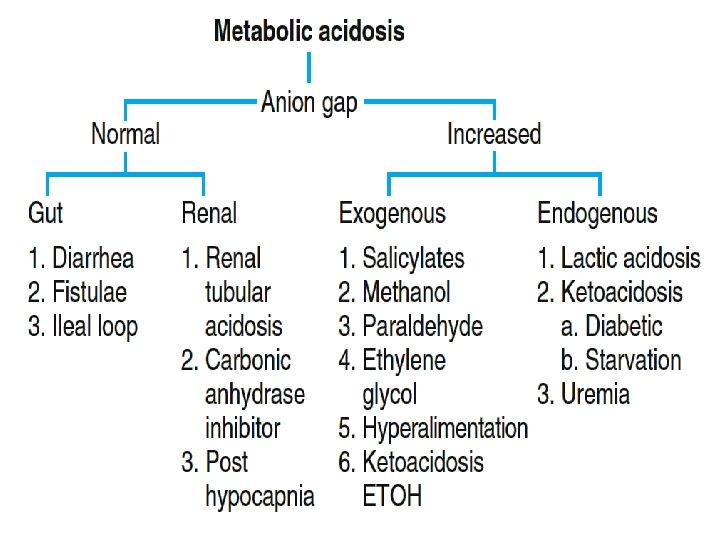

Increased Anion Gap Normal = 8 -15 May differ institutionally • Accumulation of organic acids (ketones, lactate) • Toxic Ingestions – methanol, ethylene glycol, salicylates • Reduced inorganic acid excretion – phosphates, sulfates • Decrease in unmeasured cations (unusual)

Increased AG Metabolic Acidosis: • Methanol • Uremia/Renal Failure • INH, Iron--lactate • Paraldehyde • Lactic Acidosis – Has many etiologies – Cyanide, CO, Toluene, HS – Poor perfusion • Ethylene glycol • Salicylates – Methyl salicylate • (Oil of wintergreen) – Mg salicylate Levraut J et al. Int Care Med 23: 417, 1997

Decreased or Negative Anion Gap Clin J Am Soc Nephrol 2: 162 -174, 2007 • Low protein most important • Albumin has many unmeasured negative charges • “Normal” anion gap (12) in cachectic person – Indicates anion gap metabolic acidosis • 2 -2. 5 m. Eq/liter drop in AG for every 1 g drop in albumin • Other etiologies of low AG: – Low K, Mg, Ca, increased globulins (Mult. Myeloma), Li, Br (bromism), I intoxication • Negative AG – more unmeasured cations than unmeasured anions – Bromide, Iodide, Multiple Myeloma

Sources 1. Achmadi, A. , George, YWH. , Mustafa, I. Pendekatan “Stewart” Dalam Fisiologi Keseimbangan Asam Basa. ppt. 2007 2. Magdy. A. Blood Gases and Acid-Base Disorders. ppt. 2011 3. Paphitou, N. Interpretation of Arterial Blood Gases and Acid-Base Disorders. PPT. 2011. 4. Rashid, FA. Respiratory mechanism in acidbase homeostasis. PPT. 2005. 5. Smith, SW. Acid-Base Disorders. www. acidbase. com 28
- Slides: 28