AcidBase Definitions Acid substance that can donate hydrogen

Acid-Base

Definitions • Acid: substance that can donate hydrogen ions • Base: substance that can accept hydrogen ions

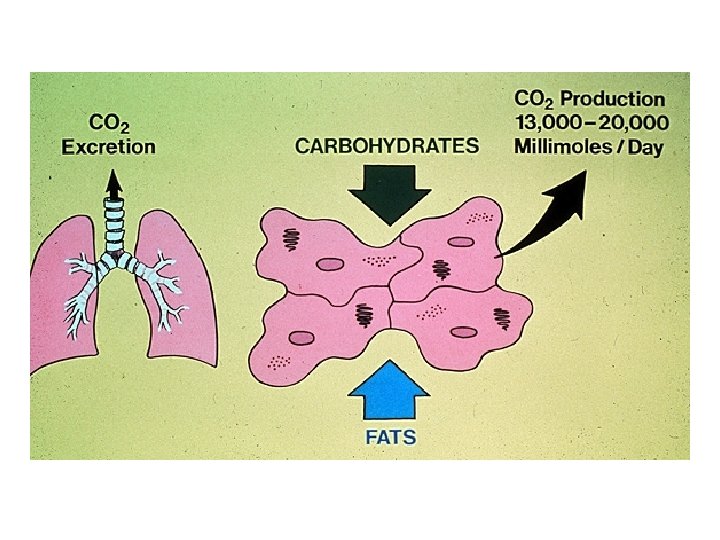
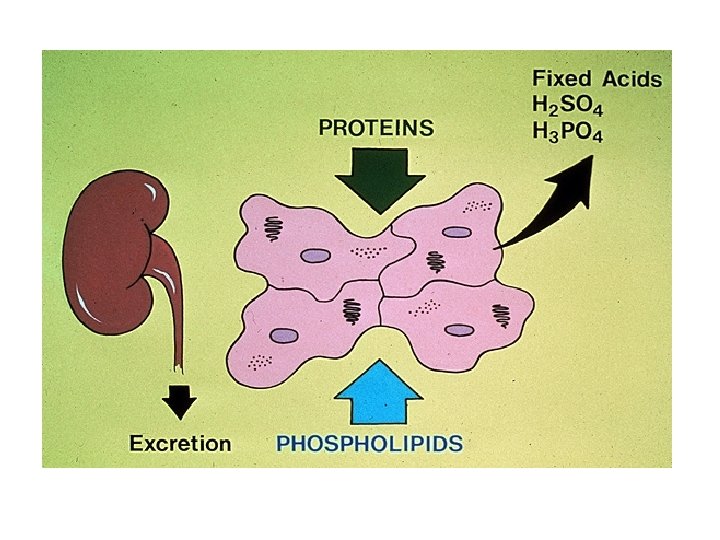
Types of acids • Carbonic acid (volatile acid, carbon dioxide, ~ 15000 mmol/d). Eliminated by the lungs • Non-carbonic acids (nonvolatile acids such as phosphoric and sulfuric acids, 50 -100 meq/d). Combine with buffers and subsequently excreted by the kidney

Clinical p. H range • p. H between 7. 80 and 6. 80 (H+concentrations between 16 -160 neq/l) are the extremes of p. H compatible with life • Clinical laboratories measured p. H, carbon dioxide, and oxygen in arterial samples • Bicarbonate concentration can be calculated from the Henderson equation • Laboratories measure total CO 2 concentration (dissolved CO 2 plus bicarbonate concentration, ~25 -26 meq/l) in venous samples

Plasma bicarbonate concentration • Laboratories measure total CO 2 concentration (dissolved carbon dioxide plus bicarbonate concentration, ~25 -26 meq/l) • As a result, total CO 2 concentration exceeds plasma bicarbonate concentration by 1. 0 to 1. 5 meq/l • Normal plasma bicarbonate concentration is approximately 24 m. Eq/l

Definitions • Reduced p. H (elevated hydrogen ion concentration) equals acidemia • Increased p. H (reduced hydrogen ion concentration equals alkalemia) • Process that lowers p. H = acidosis • Process that increases p. H = alkalosis

Bicarbonate buffer system • CO 2 + H 20 H 2 CO 3 H+ + HCO 3 • If a closed system, p. Ka = 6. 1 (normal p. H= 7. 40) • We are an open system via the lungs excreting CO 2 making this system a highly efficient buffer
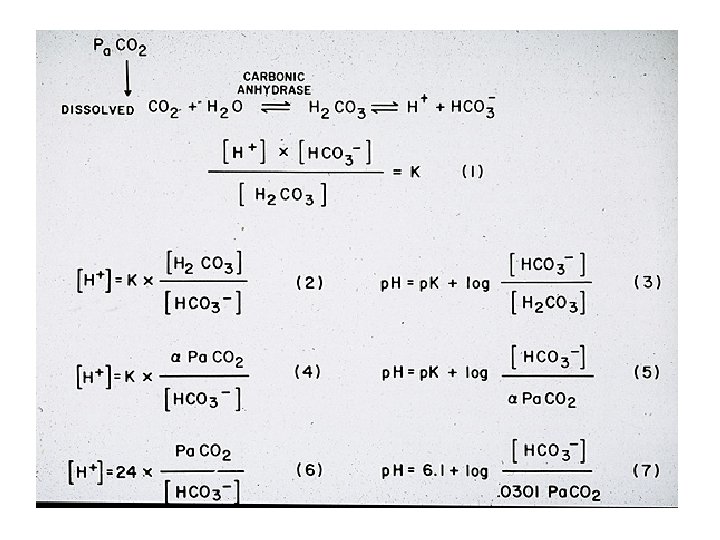

Determinants of p. H • (p. H) H+ = 24 ( CO 2) (HCO 3) • p. H must be converted to H+ (n. Eq/L). p. H = 7. 40 = 40 n. Eq/L • 7. 40 = 24 (40/24)
![p. H vs. [H+] • p. H 7. 0 7. 1 7. 2 7. p. H vs. [H+] • p. H 7. 0 7. 1 7. 2 7.](http://slidetodoc.com/presentation_image/3164604785e08068740b84de9b501695/image-12.jpg)
p. H vs. [H+] • p. H 7. 0 7. 1 7. 2 7. 3 7. 4 7. 5 7. 6 7. 7 7. 8 • [H+] 100 80 64 50 40 32 25 20 16 [H+] = 80 – decimal digits of p. H

Normals • Normal p. H – 7. 35 -7. 45 (7. 40) – p. H = -log[H+] – [H+] = 24 x p. CO 2/[HCO 3 -] • Normal p. CO 2 – 36 -44 mm Hg (40 mm. Hg) • Normal HCO 3– 22 -26 meq/L (24 meq/l)

Metabolic Disorders • Processes that directly alter bicarbonate concentration Metabolic acidosis: decreased bicarbonate • Metabolic alkalosis: increased bicarbonate

Respiratory Disorders • • Processes that directly alter CO 2 Respiratory acidosis: increased CO 2 Respiratory alkalosis: decreased CO 2 Buffer effect: slightly increased HCO 3 with respiratory acidosis. Slightly decreased HCO 3 with respiratory alkalosis

Buffering • Prevent wide changes in p. H in response to the addition of base or acid • Bicarbonate is the major extracellular buffer (can be easily measured) • There also intracellular buffers

Effects of Buffers on p. H • Bicarbonate is the major extracellular buffer. There also intracellular buffers. • The presence of buffers attenuates changes in p. H in response to acid-base disorders. • Immediate onset • Isohydric principle (all buffers change in the same direction)
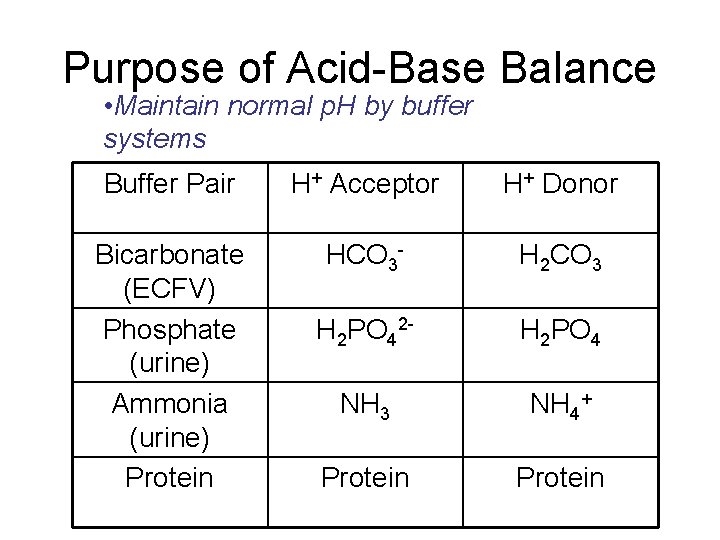
Purpose of Acid-Base Balance • Maintain normal p. H by buffer systems Buffer Pair H+ Acceptor H+ Donor Bicarbonate (ECFV) Phosphate (urine) Ammonia (urine) Protein HCO 3 - H 2 CO 3 H 2 PO 42 - H 2 PO 4 NH 3 NH 4+ Protein

Secondary(Compensatory) Mechanisms • In addition to buffering mechanisms, additional secondary (compensatory) physiologic responses occur in response to changes in p. H. • Invariably present in simple acid-base disorders (if not present, it is a mixed disorder)

Compensatory mechanisms • The respiratory system compensates for metabolic disorders by altering CO 2 (via the lungs, rapid onset, minutes) • Compensation for respiratory disorders occurs by alterations in bicarbonate concentration (via the kidney, slower onset 1 -2 days)

Mechanisms that Buffer an Acid Load Buffer systems Extracellula r fluid (primarily bicarbonate) Increased rate and Lungs depth of breathing to decrease CO 2 Immediate (HCO 3 - + H+ ↔ H 2 CO 3 ↔ CO 2 + H 20) Minutes to hours Buffer systems (phosphate, bicarbonate, protein) Intracellular fluid 2 -4 hours Hydrogen ion excretion, bicarb reabsorption, & Kidneys Hours to days
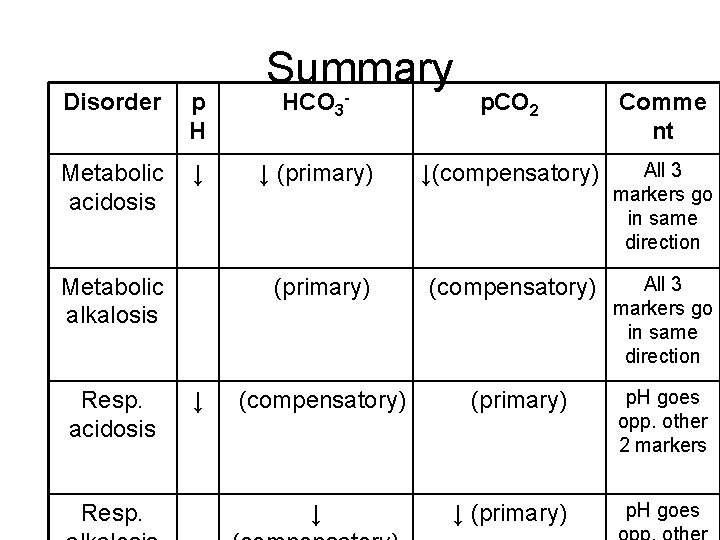
Disorder p H Metabolic acidosis ↓ Metabolic alkalosis Resp. acidosis Resp. ↓ Summary HCO 3 - p. CO 2 Comme nt ↓ (primary) ↓(compensatory) All 3 markers go in same direction (primary) (compensatory) All 3 markers go in same direction (compensatory) (primary) ↓ ↓ (primary) p. H goes opp. other 2 markers p. H goes

Golden rules: Simple acid-base disorders • 1) PCO 2 and HCO 3 always change in the same direction. • 2) The secondary physiologic compensatory mechanisms must be present. • 3) The compensatory mechanisms never fully correct p. H.

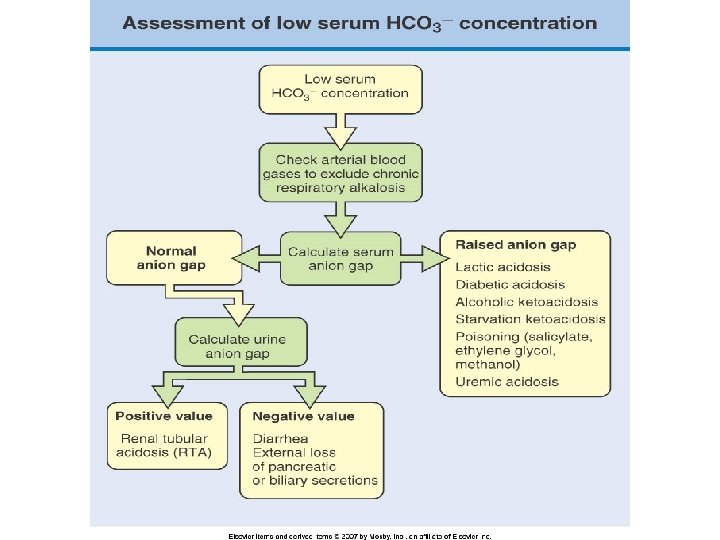
Metabolic acidosis • Process that reduces plasma bicarbonate concentration • Etiology: • Decreased renal acid excretion • Direct bicarbonate losses (GI tract or urine) • Increased acid generation (exogenous or endogenous)

Causes of metabolic acidosis • 1) increased acid generation • Lactic acidosis, Ketoacidosis, ingestion of acids (aspirin, ethylene glycol, methanol), dietary protein intake (animal source) • 2) loss of bicarbonate • Gastrointestinal (diarrhea, intestinal fistulas) • Renal: type 2 proximal renal tubular acidosis

Causes of metabolic acidosis • 1) decreased acid excretion (impaired NH 4+ excretion) • Renal failure (reduced GFR) decreased ammonium excretion • Type I (distal) renal tubular acidosis • Type 4 renal tubular acidosis (hypoaldosteronism)

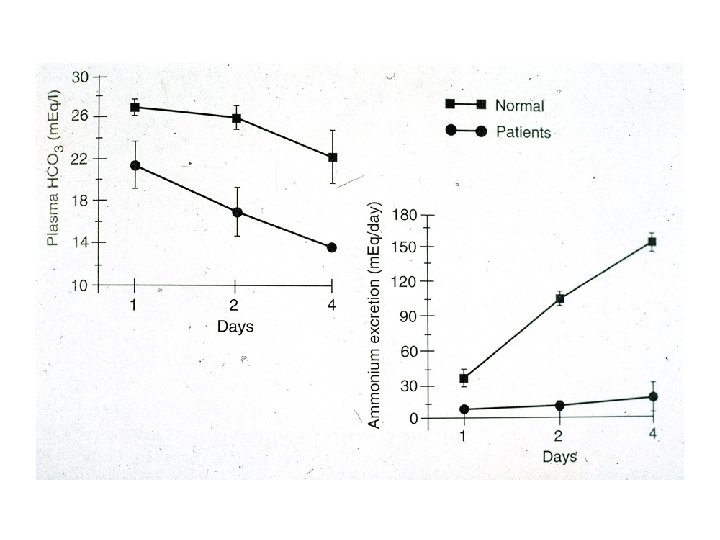
Respiratory acidosis • Induced by hypercapnia (decreased alveolar ventilation) • Buffering mechanisms raise plasma bicarbonate concentration (rapid but limited response, ~1 -2 meq/l) • Kidney minimizes the change in extracellular p. H by increasing acid excretion (NH 4+) generating new bicarbonate ions (delayed response, 2 -3 days).


Respiratory alkalosis • Reduced carbon dioxide due to increased alveolar ventilation • Buffering processes lower plasma bicarbonate concentration (rapid but limited response, ~1 -2 meq/l) • Kidney response is to reduce net acid excretion (eliminate bicarbonate into the urine or decrease ammonium excretion). Delayed response, 1 -2 days)
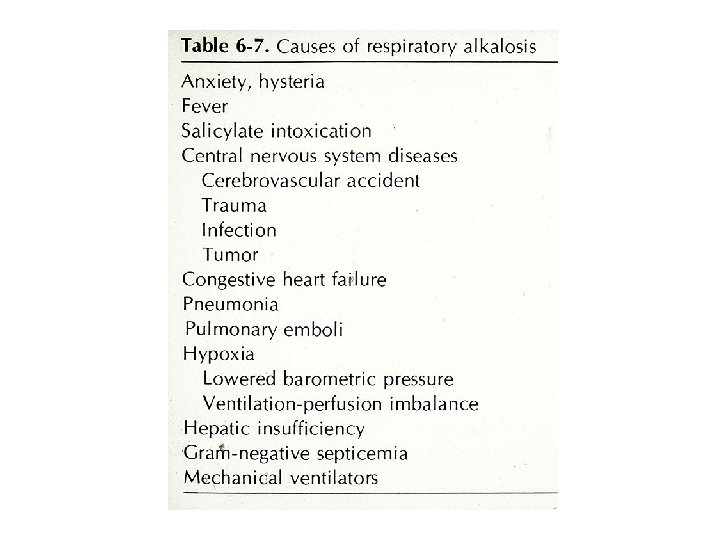

Respiratory disorders • Acute respiratory acid base disorders always have a greater change in p. H than chronic disorders • Plasma Cl changes equally and inversely with plasma HCO 3. • Plasma anion gap does not change with respiratory disorders • Plasma sodium is not directly altered by acid base disorders

Metabolic alkalosis • Processes that raise plasma bicarbonate concentration • Etiology: Loss of hydrogen ion from the GI tract (vomiting) or into the urine (diuretic therapy) • Excessive urinary net acid excretion (primary hyperaldosteronism)

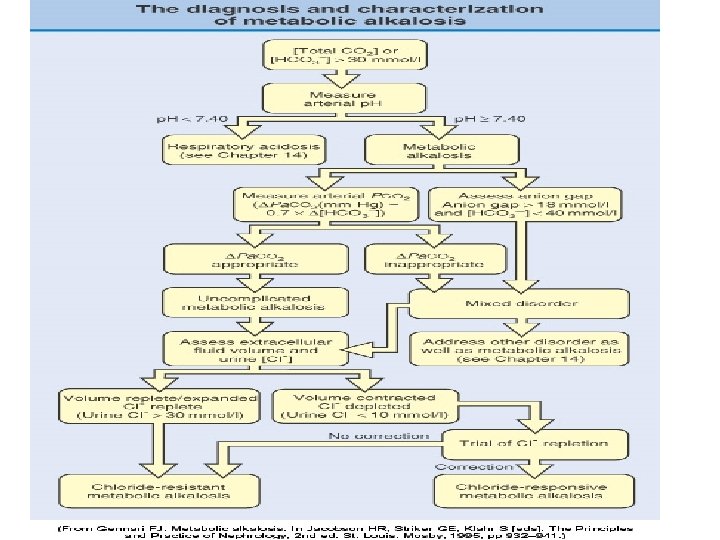

Metabolic alkalosis • Urine chloride concentration: • Cl responsive: urine Cl <20 meq/l (usually <10 meq/l • Cl resistant: urine Cl > 20 meq/l (usuallly >50 meq/l)

Expected p. H Changes for Respiratory Disorders • Acute Respiratory Acidosis: HCO 3 - increases 1 m. Eq for each 10 mm increase in PCO 2 • Chronic Respiratory Acidosis: HCO 3 - increases 4 m. Eq for each 10 mm increase in PCO 2 • Acute Respiratory Alkalosis: HCO 3 - decreases 2 m. Eq for each 10 mm decrease in PCO 2 • Chronic Respiratory Alkalosis: HCO 3 - decreases 5 m. Eq for each 10 mm decrease in PCO


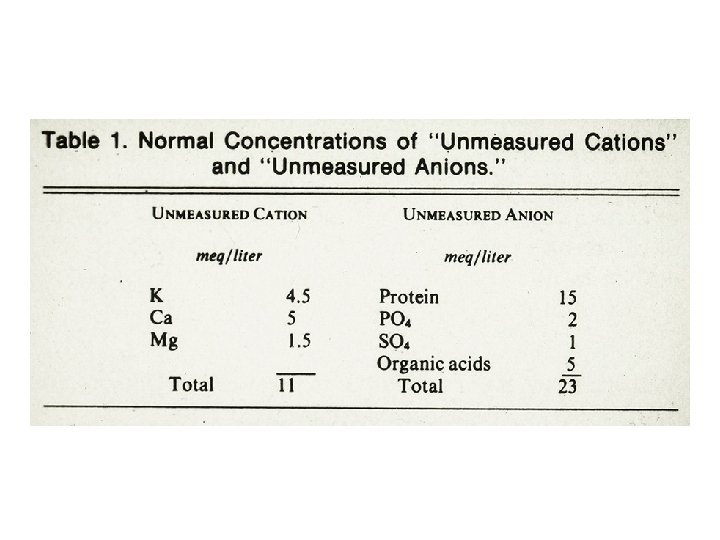
Plasma anion gap Strong acids (HA) fully dissociate at physiologic p. H (7. 40) into H+ and AH+ is buffered by HC 03 A- is either excreted into the urine (normal plasma anion gap, increased plasma chloride concentration) Or, A- is reabsorbed by the kidney and retained in plasma, as an unmeasured anion (increased plasma anion gap, minimal change in plasma chloride concentration)
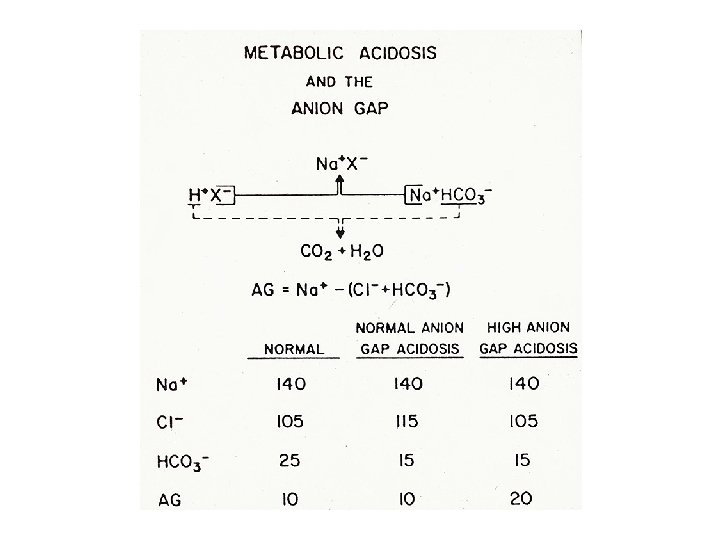


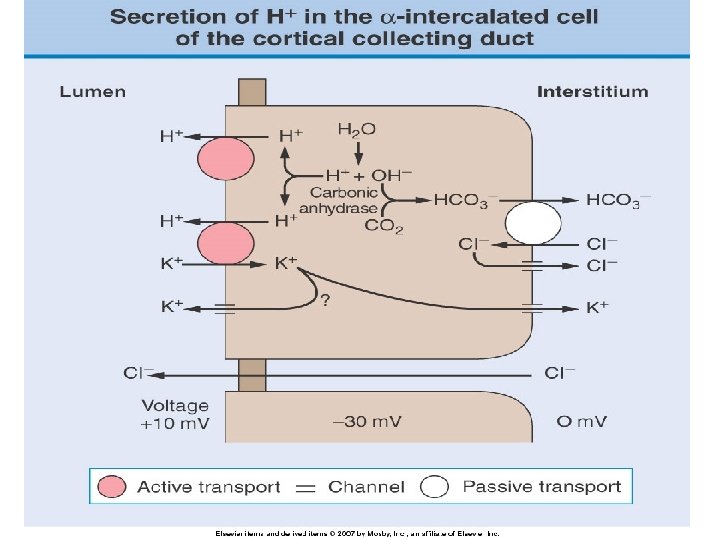
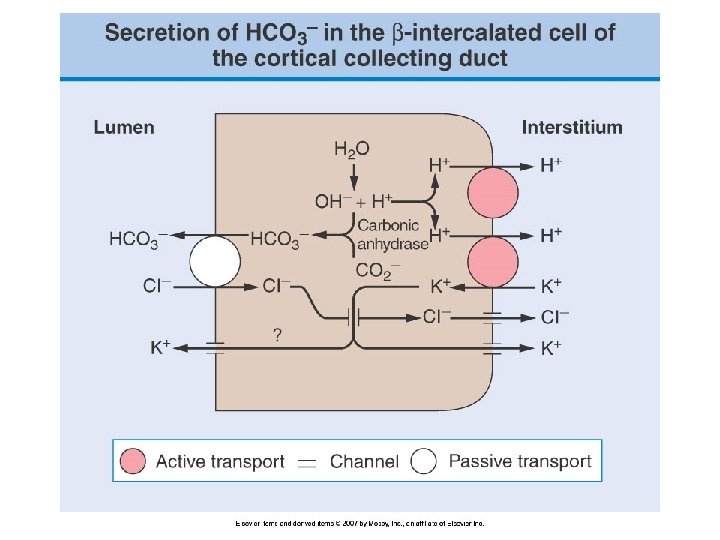
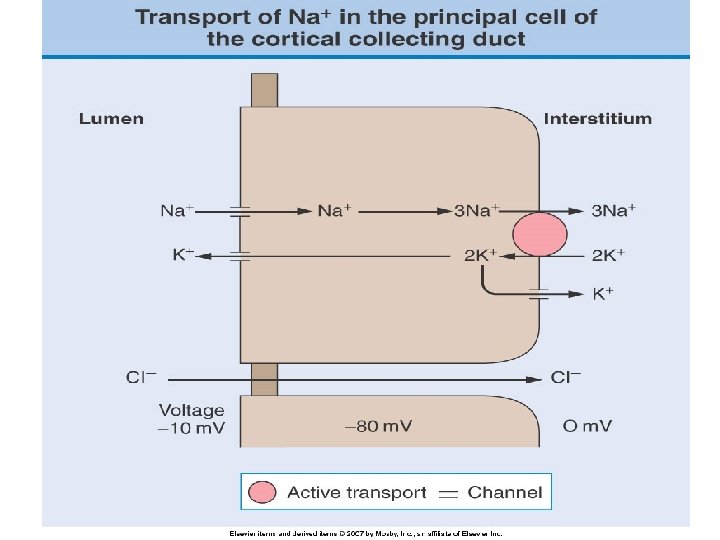
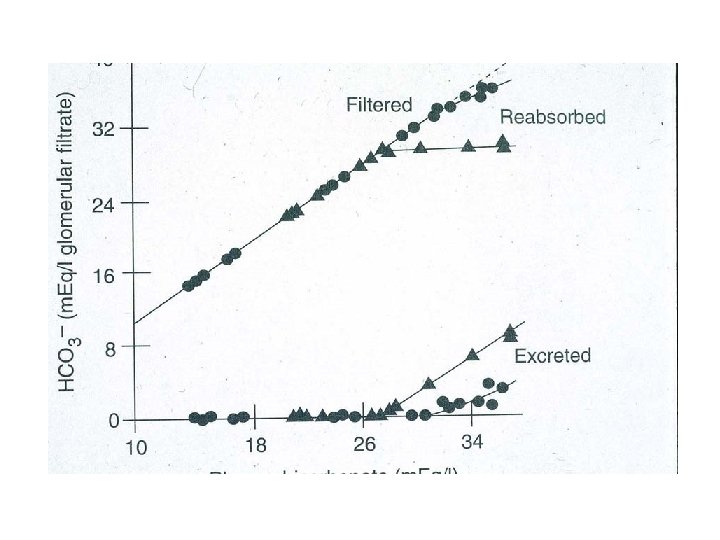
Renal acid excretion • All of the filter of bicarbonate must be reabsorbed (primarily in the proximal tubule and loop of Henle) • Final excretion of the daily acid load occurs primarily in the collecting duct (approximately 50 -100 meq/d)

Titratable acidity • Phosphate homeostasis is maintained by urinary excretion of dietary phosphate • Monobasic phosphate is an effective urinary buffer, esp. at lower urinary p. H • Accounts for excretion of 10 to 40 m. Eq of hydrogen ion daily • Cannot be increased beyond this due to the fixed amount of phosphate in urine

Ammonium excretion • Contributes the major adaptive response to an acid load • Can be increased in response to physiologic needs • Normally 30 -40 m. Eq/d and maximal excretion is approximately 300 m. Eq/d • NH 4+ is lipid soluble and therefore trapped in the urinary lumen

Urine anion gap • An indirect estimate of urinary NH 4+ excretion • Urine Na + K minus urine Cl • Normally, ~ 10 meq/l • Becomes less positive and may even become neg with incr urinary NH 4 excretion (Cl- must accomany NH 4+)

Sodium and Chloride relationship • Law of electroneutrality: • Sodium concentration is not directly altered by acid base disorders • Plasma Cl is altered in all acid base disorders (except increased plasma anion gap metaboic acidosis) • Conclusion: If sodium concetration stays constant but chloride conc changes, an acid base disorder is present

Mixed Acid-base disorders • The presence of more than one simple acid-base disorder simultaneously: • Respiratory acidosis and metabolic acidosis (profound acidemia) • Respiratory alkalosis and metabolic alkalosis (profound alkalemia) • Metabolic alkalosis and respiratory acidosis • Metabolic acidosis and respiratory alkalosis

Examination/quiz • For this course, only simple acid-base disorders will be included in quizzes and examinations. • You have got to crawl before you walk!
- Slides: 55