AcidBase Concepts Chapter 15 1 Arrhenius AcidBase Concept

Acid-Base Concepts -- Chapter 15 1. Arrhenius Acid-Base Concept (last semester) Acid: H+ supplier Base: OH– supplier 2. Brønsted-Lowry Acid-Base Concept (more general) (a) Definition (H+ transfer) Acid: H+ donor Base: H+ Acceptor + H+ Conjugate Acid-Base Pairs: Base – H+ Acid
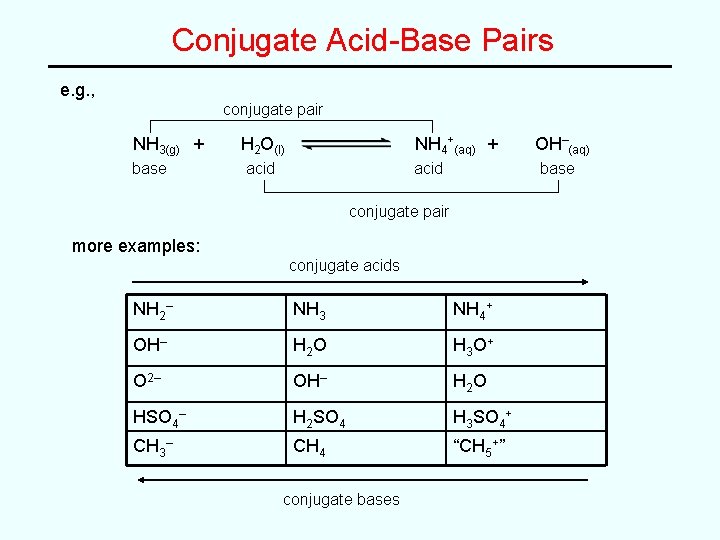
Conjugate Acid-Base Pairs e. g. , conjugate pair NH 3(g) + base H 2 O(l) NH 4+(aq) + acid base conjugate pair more examples: conjugate acids NH 2– NH 3 NH 4+ OH– H 2 O H 3 O + O 2– OH– H 2 O HSO 4– H 2 SO 4 H 3 SO 4+ CH 3– CH 4 “CH 5+” conjugate bases OH–(aq)

Amphoteric Substances Molecules or ions that can function as both acids and bases (e. g. H 2 O itself!) e. g. the bicarbonate ion, HCO 3– + OH– --> H 2 O + CO 32– acid base HCO 3– + HCl --> H 2 CO 3 + Cl– base acid
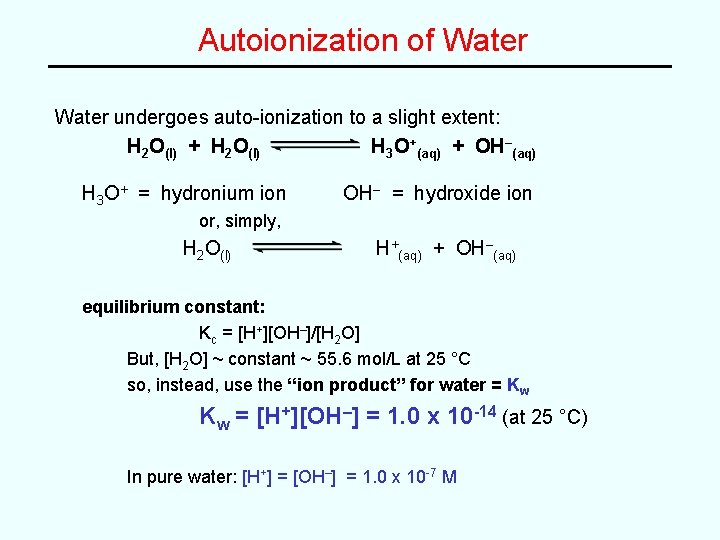
Autoionization of Water undergoes auto-ionization to a slight extent: H 2 O(l) + H 2 O(l) H 3 O+(aq) + OH–(aq) H 3 O+ = hydronium ion OH– = hydroxide ion or, simply, H 2 O(l) H+(aq) + OH–(aq) equilibrium constant: Kc = [H+][OH–]/[H 2 O] But, [H 2 O] ~ constant ~ 55. 6 mol/L at 25 °C so, instead, use the “ion product” for water = Kw Kw = [H+][OH–] = 1. 0 x 10 -14 (at 25 °C) In pure water: [H+] = [OH–] = 1. 0 x 10 -7 M
![The p. H Scale Kw = [H+][OH–] = 1. 0 x 10 -14 The The p. H Scale Kw = [H+][OH–] = 1. 0 x 10 -14 The](http://slidetodoc.com/presentation_image/10a5eed8edd1adf44cc5b2ff9fe24aa9/image-5.jpg)
The p. H Scale Kw = [H+][OH–] = 1. 0 x 10 -14 The p. H scale: p. H = – log [H+] In general: p. X = – log X e. g. p. OH = – log [OH–] and, in reverse: [H+] = 10–p. H mole/L [OH–] = 10–p. OH mole/L two sig figs Since Kw = [H+][OH–] = 1. 0 x 10– 14 p. Kw = p. H + p. OH = 14. 00 two sig figs! Notice the sig figs!
![Relative Acidity of Solutions neutral solution [H+] = [OH–] = 1. 0 x 10– Relative Acidity of Solutions neutral solution [H+] = [OH–] = 1. 0 x 10–](http://slidetodoc.com/presentation_image/10a5eed8edd1adf44cc5b2ff9fe24aa9/image-6.jpg)
Relative Acidity of Solutions neutral solution [H+] = [OH–] = 1. 0 x 10– 7 M p. H = p. OH = 7. 00 acidic solution [H+] > 10– 7 (i. e. more H+ than in pure water) p. H < 7. 00 [OH–] < 10– 7 and p. OH > 7. 00 e. g. if [H+] = 1. 00 x 10– 3 M then p. H = 3. 000 and p. OH = 11. 000 Basic solution [H+] < 10– 7 (i. e. less H+ than in pure water) p. H > 7. 00 [OH–] > 10– 7 and p. OH < 7. 00 e. g. if [OH–] = 1. 00 x 10– 3 M then p. OH = 3. 000 and p. H = 11. 000
![Example Problem The water in a soil sample was found to have [OH–] equal Example Problem The water in a soil sample was found to have [OH–] equal](http://slidetodoc.com/presentation_image/10a5eed8edd1adf44cc5b2ff9fe24aa9/image-7.jpg)
Example Problem The water in a soil sample was found to have [OH–] equal to 1. 47 x 10– 9 mole/L. Determine [H+], p. H, and p. OH. Answer: [H+] = Kw/[OH–] = (1. 00 x 10– 14)/(1. 47 x 10– 9) = 6. 80 x 10– 6 p. H = – log [H+] = – log (6. 80 x 10– 6) = 5. 167 (acidic!) p. OH = 14. 00 – p. H = 14. 00 – 5. 167 = 8. 833 {or, p. OH = – log [OH–] = – log (1. 47 x 10– 9) = 8. 833}
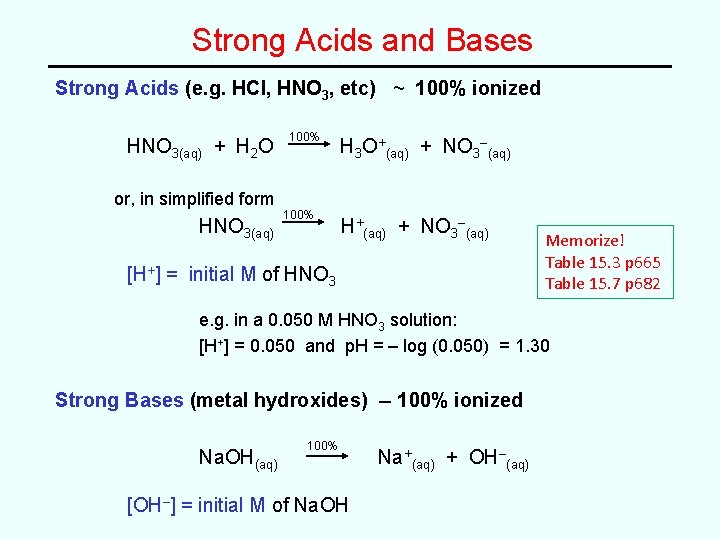
Strong Acids and Bases Strong Acids (e. g. HCl, HNO 3, etc) ~ 100% ionized HNO 3(aq) + H 2 O or, in simplified form HNO 3(aq) 100% H 3 O+(aq) + NO 3–(aq) H+(aq) + NO 3–(aq) [H+] = initial M of HNO 3 Memorize! Table 15. 3 p 665 Table 15. 7 p 682 e. g. in a 0. 050 M HNO 3 solution: [H+] = 0. 050 and p. H = – log (0. 050) = 1. 30 Strong Bases (metal hydroxides) -- 100% ionized Na. OH(aq) 100% [OH–] = initial M of Na. OH Na+(aq) + OH–(aq)

Example Problem What mass of Ba(OH)2 (171. 34 g/mole) is required to prepare 250 m. L of a solution with a p. H of 12. 50? First: Write the reaction. Ba(OH)2(aq) 100% Ba 2+(aq) + 2 OH–(aq) so, [OH–] = 2 x M of Ba(OH)2 solution (2: 1 ratio) Second: Determine [OH–]. p. OH = 14. 00 – p. H = 14. 00 – 12. 50 = 1. 50 [OH–] = 10– 1. 50 = 0. 032 M Third: How much Ba(OH)2 is needed for that much OH–? 250 m. L soln x 0. 0079 mol OH– x 0. 032 mol OH– 1000 m. L soln = 0. 0079 mol OH– 1 mole Ba(OH)2 171. 34 g Ba(OH)2 x 1 mol Ba(OH)2 2 mol OH– = 0. 68 g Ba(OH)2

Sample Problem (a) Determine the p. H of a 1. 75 M solution of HNO 3. (b) Now suppose that 500. 0 m. L of a 1. 50 M Ba(OH)2 solution is added to 1. 00 L of the above HNO 3 solution. Determine the p. H of the resulting solution. (c) What additional volume (in m. L) of 1. 50 M Ba(OH)2 must be added to the mixture in part (b) to bring the p. H to 7. 00?

Sample Problem (a) Determine the p. H of a 1. 75 M solution of HNO 3. a) p. H = – 0. 243 (b) Now suppose that 500. 0 m. L of a 1. 50 M Ba(OH)2 solution is added to 1. 00 L of the above HNO 3 solution. Determine the p. H of the resulting solution. b) p. H = 0. 78 (c) What additional volume (in m. L) of 1. 50 M Ba(OH)2 must be added to the mixture in part (b) to bring the p. H to 7. 00? c) 83 m. L Ba(OH)2 soln

Weak Acids and Bases (a) Weak Acids -- Less than 100% ionized (equilibrium !) In general: HA is a weak acid, A– is its conjugate base HA(aq) + H 2 O H 3 O+(aq) + A–(aq) or, simply, HA(aq) H+(aq) + A–(aq) Acid Dissociation Constant: Ka [H+][A–] Ka = [HA] Relative acid strength: Weak acid: Ka < ~10– 3 Moderate acid: Ka ~ 1 to 10– 3 Strong acid: Ka > 1

Example Problem Hypochlorous acid, HOCl, has a p. Ka of 7. 52. What is the p. H of an 0. 25 M solution of HOCl? What is the percent ionization? First: Write equation and ICE table. ⇌ HOCl(aq) H+(aq) + OCl–(aq) I 0. 25 M 0 0 C –x +x +x E 0. 25 – x x x Second: Write equilibrium expression. p. Ka = – log Ka, so 3. 0 x 10– 8 = Ka = 10–p. Ka = 10– 7. 52 = 3. 0 x 10– 8 [H+][Cl–] [HOCl] = (x)(x) (0. 25 –x) = x 2 (0. 25 –x) Third: Solve for [H+] and p. H. Since Ka is very small, assume x << 0. 25 x 2/(0. 25) ≈ 3. 0 x 10– 8 x ≈ 8. 7 x 10– 5 (OK!) p. H = –log (8. 7 x 10– 5) = 4. 06 (solution is acidic!)

Example Problem, cont. Fourth: Determine percent ionization. • % ionization = (amount HA ionized)/(initial) x 100% = (8. 7 x 10– 5)/(0. 25) x 100% = 0. 035%

Weak Bases (b) Weak Bases • In general: B is a weak base, HB+ is its conjugate acid B(aq) + H 2 O(l) HB+(aq) + OH–(aq) Base Dissociation Constant: e. g. NH 3(aq) + H 2 O(l) Kb = [NH 4+][OH–] [NH 3] Kb = [HB+][OH–] [B] NH 4+(aq) + OH–(aq) = 1. 8 x 10– 5 p. Kb = -log Kb = 4. 74 Note: Since OH– rather than H+ appears here, first find [OH–] or p. OH, and then convert to p. H Example problem: p. H of 0. 25 M solution of NH 3? Set up conc. table as usual, solve for x = [OH–] = 2. 1 x 10– 3 p. OH = 2. 68 p. H = 11. 32 (basic!)

Sample Problem The nitrite ion (NO 2–) is a weak base with a p. Kb value of 10. 85. (a) Write a balanced net ionic equation for the major equilibrium reaction that is occurring in an aqueous solution of sodium nitrite (Na. NO 2). (b) Calculate the p. H of a 0. 25 M solution of sodium nitrite (Na. NO 2). Clearly state and justify any assumptions that you make.
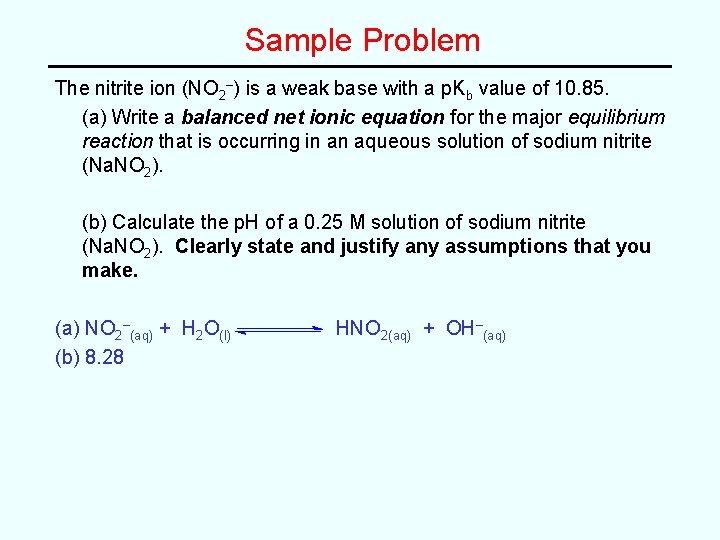
Sample Problem The nitrite ion (NO 2–) is a weak base with a p. Kb value of 10. 85. (a) Write a balanced net ionic equation for the major equilibrium reaction that is occurring in an aqueous solution of sodium nitrite (Na. NO 2). (b) Calculate the p. H of a 0. 25 M solution of sodium nitrite (Na. NO 2). Clearly state and justify any assumptions that you make. (a) NO 2–(aq) + H 2 O(l) (b) 8. 28 HNO 2(aq) + OH–(aq)

Salts of Weak Acids and Bases • A) conjugate acid-base pairs (HA and A–) – Ka: HA – Kb: A- + H 2 O For any conjugate acid-base pair: Ka Kb = K w p. Ka + p. Kb = 14. 00 H + + A– HA + OH–
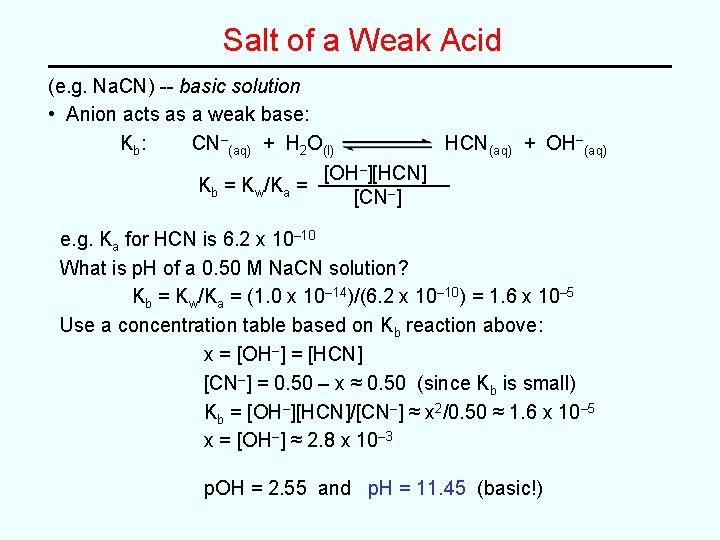
Salt of a Weak Acid (e. g. Na. CN) -- basic solution • Anion acts as a weak base: Kb : CN–(aq) + H 2 O(l) HCN(aq) + OH–(aq) [OH–][HCN] Kb = Kw/Ka = [CN–] e. g. Ka for HCN is 6. 2 x 10– 10 What is p. H of a 0. 50 M Na. CN solution? Kb = Kw/Ka = (1. 0 x 10– 14)/(6. 2 x 10– 10) = 1. 6 x 10– 5 Use a concentration table based on Kb reaction above: x = [OH–] = [HCN] [CN–] = 0. 50 – x ≈ 0. 50 (since Kb is small) Kb = [OH–][HCN]/[CN–] ≈ x 2/0. 50 ≈ 1. 6 x 10– 5 x = [OH–] ≈ 2. 8 x 10– 3 p. OH = 2. 55 and p. H = 11. 45 (basic!)

Salt of a Weak Base (e. g. NH 4 Cl) -- Acidic Solution • Cation acts as a weak acid: Ka : NH 4+ H+ + NH 3 [H+][NH 3] Ka = Kw/Kb = [NH 4+] e. g. Kb for NH 3 is 1. 8 x 10– 5 What is p. H of a 0. 50 M NH 4 Cl solution? Ka = Kw/Kb = (1. 0 x 10– 14)/(1. 8 x 10– 5) = 5. 6 x 10– 10 Use a concentration table based on Ka reaction above: x = [H+] = [NH 3] [NH 4+] = 0. 50 – x ≈ 0. 50 (since Ka is small) Ka = [H+][NH 3]/[NH 4+] ≈ x 2/0. 50 ≈ 5. 6 x 10– 10 x = [H+] ≈ 1. 7 x 10– 5 p. H = 4. 77 (acidic!)

Sample Problem The p. Ka value for HCN is 9. 21. What molar concentration of Na. CN is required to make a solution with a p. H of 11. 75?

Sample Problem The p. Ka value for HCN is 9. 21. What molar concentration of Na. CN is required to make a solution with a p. H of 11. 75? Answer: 2. 0 M Na. CN
![Polyprotic Acids e. g. diprotic acids, H 2 A, undergo stepwise dissociation: [HA–][H+] Ka Polyprotic Acids e. g. diprotic acids, H 2 A, undergo stepwise dissociation: [HA–][H+] Ka](http://slidetodoc.com/presentation_image/10a5eed8edd1adf44cc5b2ff9fe24aa9/image-23.jpg)
Polyprotic Acids e. g. diprotic acids, H 2 A, undergo stepwise dissociation: [HA–][H+] Ka 1 = H 2 A HA– + H+ [H 2 A] HA– A 2– + H+ Ka 2 = [A 2–][H+] [HA–] Usually, Ka 1 >> Ka 2 so that: The 1 st equilibrium produces most of the H+ and [HA–] but the 2 nd equilibrium determines [A 2–]

Example Problem Ascorbic acid (vitamin C), H 2 C 6 H 2 O 6, is an example of a diprotic acid with Ka 1 = 7. 9 x 10– 5 and Ka 2 = 1. 6 x 10– 12. For a 0. 10 M solution of ascorbic acid, determine the p. H and the concentrations of the mono anion, HC 6 H 2 O 6–, and the dianion, C 6 H 2 O 62–. Based on the first equilibrium: x = [H+] ≅ [HA–] and [H 2 A] = 0. 10 – x ≅ 0. 10 Ka 1 = 7. 9 x 10– 5 ≅ x 2 / (0. 10) ∴ x ≅ 2. 8 x 10– 3 so p. H = 2. 55 Must use the 2 nd equilibrium to find [A 2–]: Ka 2 = [A 2–][H+] / [HA–] but, from above ∴ Ka 2 ≅ [A 2–] (a general result for H 2 A!) [A 2–] ≅ 1. 6 x 10– 12 [H+] ≅ [HA–]

How to tell if it’s acidic or basic? Anions Anion that is conjugate base of a weak acid is itself a weak base. Exception: H 2 O!! An anion that is a conjugate base of a strong acid is p. H-neutral. Anion of a polyprotic acid is amphoteric, e. g. H 2 PO 4–. Cations Cation that is conjugate acid of a weak base is itself a weak acid. A cation that is a conjugate acid of a strong base is p. H-neutral. Small, highly charged metal cations form weakly acidic solutions (not Group I or II), e. g. Al 3+(aq) + 6 H 2 O(l) Al(H 2 O)63+(aq) Neutrals weak acid Weak bases: Table 15. 8, p 683 and Fig. 15. 12, p 687 Weak acids: Tables 15. 4, p 666, and 15. 12, p 687, and Fig. 15. 12. Mixed Salts where the cation is acidic and the anion basic form solutions where the p. H depends on the relative strengths of the acid and base.

More Sample Problems 1. Write balanced chemical equations for the important equilibrium that is occurring in an aqueous solution of each of the following. (a) HCl. O (b) (NH 4)2 SO 4 (c) KCl (e) Na. CHO 2 2. Write the appropriate equilibrium constant expressions (Ka, etc) for the above reactions. 3. Determine the p. H of a 0. 100 M solution of each of the above compounds. (Use equilibrium constants from the textbook as needed).
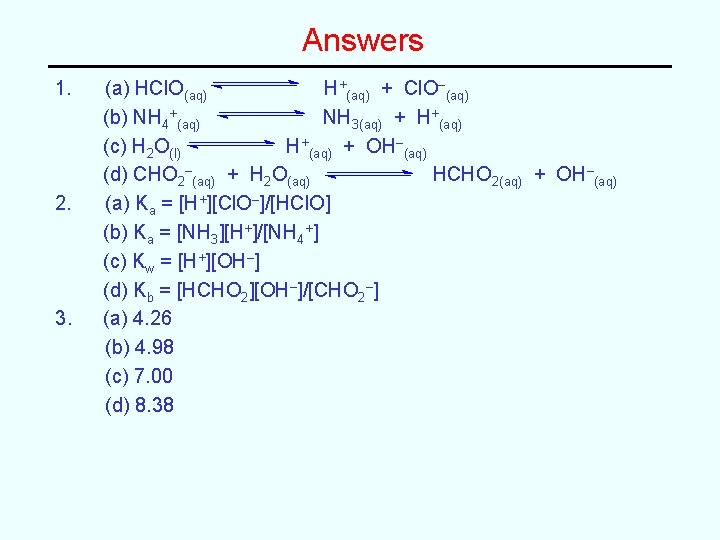
Answers 1. 2. 3. (a) HCl. O(aq) H+(aq) + Cl. O–(aq) (b) NH 4+(aq) NH 3(aq) + H+(aq) (c) H 2 O(l) H+(aq) + OH–(aq) (d) CHO 2–(aq) + H 2 O(aq) HCHO 2(aq) + OH–(aq) (a) Ka = [H+][Cl. O–]/[HCl. O] (b) Ka = [NH 3][H+]/[NH 4+] (c) Kw = [H+][OH–] (d) Kb = [HCHO 2][OH–]/[CHO 2–] (a) 4. 26 (b) 4. 98 (c) 7. 00 (d) 8. 38
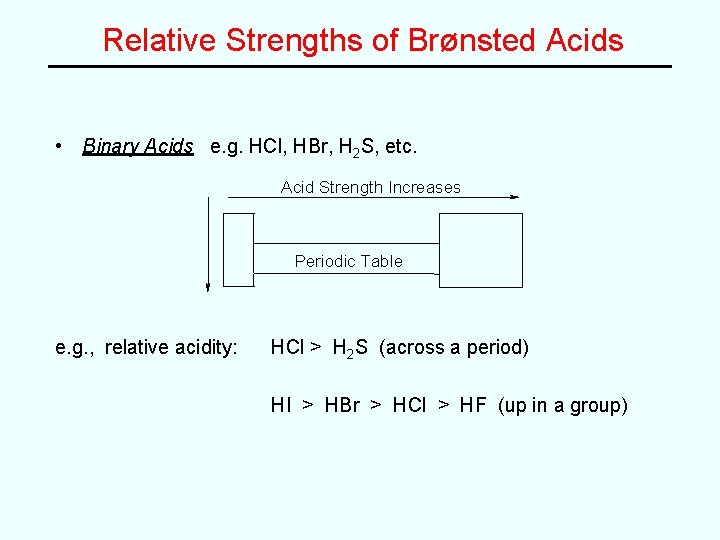
Relative Strengths of Brønsted Acids • Binary Acids e. g. HCl, HBr, H 2 S, etc. Acid Strength Increases Periodic Table e. g. , relative acidity: HCl > H 2 S (across a period) HI > HBr > HCl > HF (up in a group)
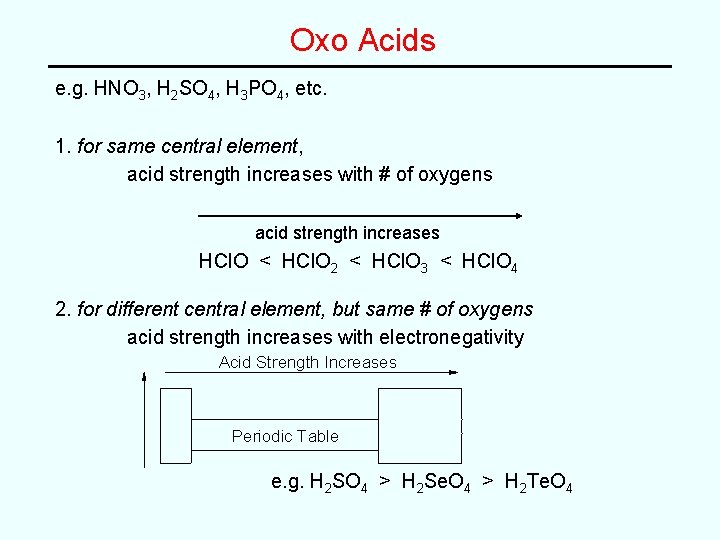
Oxo Acids e. g. HNO 3, H 2 SO 4, H 3 PO 4, etc. 1. for same central element, acid strength increases with # of oxygens acid strength increases HCl. O < HCl. O 2 < HCl. O 3 < HCl. O 4 2. for different central element, but same # of oxygens acid strength increases with electronegativity Acid Strength Increases Periodic Table e. g. H 2 SO 4 > H 2 Se. O 4 > H 2 Te. O 4
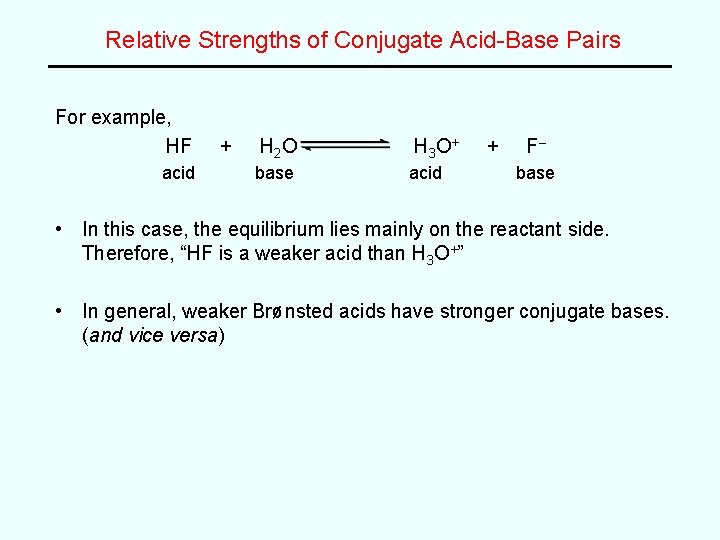
Relative Strengths of Conjugate Acid-Base Pairs For example, HF acid + H 2 O H 3 O+ base acid + F– base • In this case, the equilibrium lies mainly on the reactant side. Therefore, “HF is a weaker acid than H 3 O+” • In general, weaker Brønsted acids have stronger conjugate bases. (and vice versa)

Lewis Acid-Base Concept (most general) Definition (electron pair transfer) Acid: e– pair acceptor Base: e– pair donor Lewis acids -- electron deficient molecules or cations Lewis bases -- electron rich molecules or anions. (have one or more unshared e– pairs)
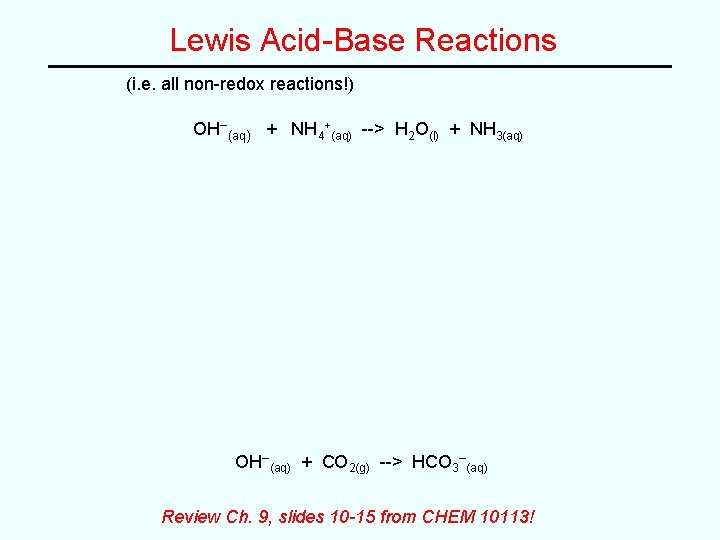
Lewis Acid-Base Reactions (i. e. all non-redox reactions!) OH–(aq) + NH 4+(aq) --> H 2 O(l) + NH 3(aq) OH–(aq) + CO 2(g) --> HCO 3–(aq) Review Ch. 9, slides 10 -15 from CHEM 10113!

Yet More Sample Problems 1) Among the following, which is the strongest acid? HBr. O 3 HBr. O 4 H 2 Se. O 3 H 2 Se. O 4 H 2 Te. O 3 H 2 Te. O 4 2) Among the following, which is the weakest acid? H 2 Se NH 3 PH 3 H 2 S H 2 O As. H 3 3) The conjugate base of CH 3 OH is _______. 4) The conjugate acid of HF is _____. 5) Write the equation for the reaction of H 2 Se with As. H 3, then answer the questions below. a) b) Label the Bronsted acid, base, conjugate acid, and conjugate base. If Kc = 0. 078, label which is the strongest acid and strongest base in your equation.

Yet More Sample Problems 1) Among the following, which is the strongest acid? HBr. O 3 HBr. O 4 H 2 Se. O 3 H 2 Se. O 4 H 2 Te. O 3 H 2 Te. O 4 HBr. O 4 2) Among the following, which is the weakest acid? H 2 Se NH 3 PH 3 H 2 S H 2 O As. H 3 NH 3 3) The conjugate base of CH 3 OH is _______. CH 3 O– 4) The conjugate acid of HF is _____. H 2 F + 5) Write the equation for the reaction of H 2 Se with As. H 3, then answer the questions below. a) b) Label the Bronsted acid, base, conjugate acid, and conjugate base. If Kc = 0. 078, label which is the strongest acid and strongest base in your equation. H 2 Se(aq) + As. H 3(aq) 1) 2) As. H 4+(aq) + HSe–(aq) acid base conj. acid conj. base strongest acid is As. H 4+, strongest base is HSe–

And Finally… Consider the reaction of hydroxide ion with the nitrosyl cation, NO 2+, to form nitric acid. OH–(aq) + NO 2+(aq) --> HNO 3(aq) Write Lewis electron dot formulas (including formal charges and/or resonance forms if needed) for all three species in this reaction. Clearly indicate which reactant is the Lewis acid and which is the Lewis base. Use arrow(s) to illustrate the formation of any new chemical bond(s) during the reaction.
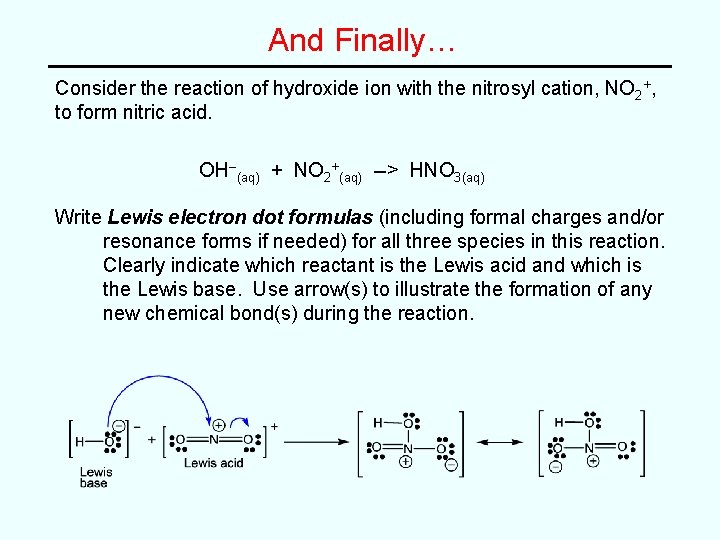
And Finally… Consider the reaction of hydroxide ion with the nitrosyl cation, NO 2+, to form nitric acid. OH–(aq) + NO 2+(aq) --> HNO 3(aq) Write Lewis electron dot formulas (including formal charges and/or resonance forms if needed) for all three species in this reaction. Clearly indicate which reactant is the Lewis acid and which is the Lewis base. Use arrow(s) to illustrate the formation of any new chemical bond(s) during the reaction.
- Slides: 36