ACIDBASE BALANCE PYRAMID POINTS Description of each acidbase

ACID-BASE BALANCE

PYRAMID POINTS • Description of each acid-base disturbance • Causes of each acid-base disturbance • Assessment findings of each acid-base disturbance • Treatment and evaluation of response to each acid-base disturbance • Allen’s test • Reading arterial blood gas results

RESPIRATORY ACIDOSIS • DESCRIPTION – The total concentration of buffer base is lower than normal, with a relative increasing hydrogen ions (H+) concentration; thus, a greater number of H+ are circulating in the blood than can be absorbed by the buffer system

RESPIRATORY ACIDOSIS • CAUSES – Due to primary defects in the function of the lungs or changes in normal respiratory patterns – Any condition that causes an obstruction of the airway or depresses respiratory status can cause respiratory acidosis – Hypoventilation: Carbon dioxide (CO 2) is retained and the H+ increase, leading to the acid state; carbonic acid is retained and the p. H goes down

RESPIRATORY ACIDOSIS • CAUSES – Medications: Sedatives, narcotics, and anesthetics depress the respiratory center, leading to hypoventilation; carbon dioxide is retained and the H+ increases – Bronchitis: Inflammation causes airway obstruction resulting in inadequate oxygenation – Atelectasis: Excessive mucus collection, with the collapse of alveolar sacs caused by mucus plugs, infectious drainage, or anesthetic medications, results in decreased respirations

RESPIRATORY ACIDOSIS • CAUSES – Brain trauma: Excessive pressure on the respiratory center or medulla oblongata depresses respirations – Emphysema: Loss of elasticity of alveolar sacs restricts air flow in and out (primarily out), leading to an increased CO 2 level – Asthma: Spasms due to allergens, irritants, or emotions cause the smooth muscles of the bronchioles to constrict

RESPIRATORY ACIDOSIS • CAUSES – Pulmonary edema: Extracellular accumulation of fluid in acute congestive heart failure (CHF) causes disturbances in alveolar diffusion and perfusion – Bronchiectasis: Bronchi become dilated due to inflammation and destructive changes and weakness in the walls of the bronchi occur

RESPIRATORY ACIDOSIS • ASSESSMENT – In an attempt to compensate, the respiratory rate and depth increase – Headache – Restlessness – Mental status changes, such as drowsiness and confusion – Visual disturbances

RESPIRATORY ACIDOSIS • ASSESSMENT – Diaphoresis – Cyanosis as the hypoxia becomes more acute – Hyperkalemia – Rapid, irregular pulse – Dysrhythmias leading to ventricular fibrillation

RESPIRATORY ACIDOSIS • IMPLEMENTATION – Monitor for signs of respiratory distress – Administer oxygen as prescribed – Place client in semi-Fowler position unless contraindicated – Encourage and assist the client to turn, cough, and deep breathe – Prepare to administer respiratory treatments as prescribed – Encourage hydration to thin secretions unless excess fluid intake is contraindicated

RESPIRATORY ACIDOSIS • IMPLEMENTATION – Suction client if necessary – Reduce restlessness by improving ventilation rather than by the administration of tranquilizers, sedatives, or narcotics, because they further depress respirations – Monitor electrolyte values, particularly the potassium level – Administer antibiotics for infection or other medications as prescribed

RESPIRATORY ALKALOSIS • DESCRIPTION – A deficit of carbonic acid (H 2 CO 3) and a decrease in H+ concentration; results from the accumulation of base or from a loss of acid without a comparable loss of base in the body fluids

RESPIRATORY ALKALOSIS • CAUSES – Due to conditions that cause overstimulation of the respiratory status – Hyperventilation: Rapid respirations cause the blowing off of CO 2 and this leads to a decrease in H 2 CO 3 – Hysteria: Often neurogenic in nature and related to a psychoneurosis; however, this condition leads to vigorous breathing and excessive exhaling of CO 2 – Hypoxia: Causes respiratory stimulation with resultant carbonic acid deficit

RESPIRATORY ALKALOSIS • CAUSES – Overventilation by mechanical ventilators: The administration of oxygen and the depletion of CO 2 can occur from mechanical ventilation; the client may be hyperventilated – Conditions that increase metabolism, such as fever – Pain or brain trauma: Causes overstimulation of the respiratory center in the brain stem with resultant carbonic acid deficit – Salicylates: Stimulate the respiratory center causing hyperventilation

RESPIRATORY ALKALOSIS • ASSESSMENT – Initially the hyperventilation and respiratory stimulation will cause abnormal rapid respirations (tachypnea); in an attempt to compensate, respiratory rate and depth go down

RESPIRATORY ALKALOSIS • ASSESSMENT – Headache – Lightheadedness, vertigo – Mental status changes – Paresthesias, such as tingling of the fingers and toes – Hypokalemia, hypocalcemia – Tetany, convulsions

RESPIRATORY ALKALOSIS • IMPLEMENTATION – Provide emotional support and reassurance to the client – Encourage appropriate breathing patterns – Assist with breathing techniques and breathing aids as prescribed – Voluntary holding of breath – Rebreathe exhaled CO 2 as prescribed – Rebreathing mask as prescribed – Carbon dioxide breaths as prescribed

RESPIRATORY ALKALOSIS • IMPLEMENTATION – Provide cautious care with ventilator clients so that the client is not forced to take breaths too deeply or rapidly – Monitor electrolyte values, particularly potassium and calcium levels – Administer medications as prescribed – Prepare to administer calcium gluconate for tetany as prescribed

METABOLIC ACIDOSIS • DESCRIPTION – The total concentration of buffer base is lower than normal, with a relative increase in the H+ concentration; occurs as a result of losing too many bases and holding too many acids without sufficient bases

METABOLIC ACIDOSIS • CAUSES – Diabetes mellitus/diabetic ketoacidosis: An insufficient supply of insulin causes increased fat metabolism, leading to an excess accumulation of ketones or other acids; the bicarbonate then ends up being exhausted – Renal insufficiency/failure: Increased waste products of protein metabolism are retained, excessive acids build up, and bicarbonate is unable to maintain acid-base balance

METABOLIC ACIDOSIS • CAUSES – Insufficient metabolism of carbohydrates: When an insufficient supply of oxygen is available for the proper burning of carbohydrates, glucose, and water, lactic acid increases and lactic acidosis results – Excessive ingestion of acetylsalicylic acid (aspirin): Causes an increase in the H+ concentration – Severe diarrhea: Intestinal and pancreatic secretions are normally alkaline; therefore, excessive loss of base leads to acidosis

METABOLIC ACIDOSIS • CAUSES – Malnutrition: Improper metabolism of nutrients causes fat catabolism, leading to an excess buildup of ketones and acids – High-fat diet: A high intake of fat causes a much too rapid accumulation of the waste products of fat metabolism, leading to a buildup of ketones and acids

METABOLIC ACIDOSIS • ASSESSMENT – In an attempt to blow off the extra CO 2 and compensate for the acidosis, hyperpnea with Kussmaul's respirations occurs – Headache – Nausea, vomiting, diarrhea – Fruity smelling breath due to improper fat metabolism – Central nervous system (CNS) depression: Mental dullness, drowsiness, stupor, coma – Twitching, convulsions – Hyperkalemia

METABOLIC ACIDOSIS • IMPLEMENTATION – Assess level of consciousness (LOC) for CNS depression – Monitor I&O and assist with fluid and electrolyte replacement as prescribed – Prepare to administer IV solutions such as normal saline, 5% dextrose and 1/2 normal saline, sodium lactate, or bicarbonate to increase the buffer base – Initiate safety and seizure precautions – Monitor the serum potassium level closely; when acidosis is being treated, potassium will move back into the cell and the serum potassium level will drop

METABOLIC ACIDOSIS • IMPLEMENTATION IN DIABETES MELLITUS/DIABETIC KETOACIDOSIS – Insulin is given to hasten the movement of serum glucose into the cell, thereby decreasing the concurrent ketosis – When glucose is being properly metabolized, the body will stop converting fats to glucose – Monitor for circulatory collapse due to polyuria which may result from the hyperglycemic state, because polyuria or diuresis may lead to extracellular volume deficit

METABOLIC ACIDOSIS • IMPLEMENTATION IN RENAL FAILURE – In renal failure, dialysis may be used to remove protein and waste products, thereby lessening the acidosis state – A diet low in protein and high in calories will lessen the amount of protein waste products due to protein catabolism; this in turn, will lessen the acidosis

METABOLIC ALKALOSIS • DESCRIPTION – A deficit of H 2 CO 3 and a decease in H+ concentration; results from the accumulation of base or from a loss of acid without a comparable loss of base in the body fluids

METABOLIC ALKALOSIS • CAUSES – Results from a malfunction of metabolism leading to an increased amount of available basic solution in the blood and a decrease in available acids in the blood – Ingestion of excess sodium bicarbonate causes an increase in the amount of base in the blood – Excessive vomiting or gastrointestinal suctioning leads to an excessive loss of acids

METABOLIC ALKALOSIS • CAUSES – Diuretics: The loss of H+ and chloride causes a compensatory increase in the bicarbonate in the blood – Hyperaldosteronism: Increased renal tubular reabsorption of sodium occurs with the resultant loss of hydrogen ions – Massive transfusion of whole blood: The citrate anticoagulant used for the storage of blood is metabolized to bicarbonate

METABOLIC ALKALOSIS • ASSESSMENT – In an attempt to compensate, respiratory rate and depth go down to conserve CO 2 – Nausea, vomiting, diarrhea – Restlessness – Numbness and tingling in the extremities – Twitching in the extremities – Hypokalemia – Hypocalcemia – Dysrhythmias: tachycardia

METABOLIC ALKALOSIS • IMPLEMENTATION – Monitor potassium and calcium serum blood levels – Institute safety precautions – Prepare to administer medications as prescribed to promote the kidney excretion of bicarbonate – Prepare to replace potassium chloride as prescribed

OBTAINING AN ARTERIAL BLOOD GAS SPECIMEN • Obtain vital signs • Determine whether the client has an arterial line in place • Perform Allen’s test to determine the presence of collateral circulation • Assess factors that may affect the accuracy of the results such as changes in the oxygen settings, suctioning within the last 20 minutes, and client activities

OBTAINING AN ARTERIAL BLOOD GAS SPECIMEN • Provide emotional support to the client • Assist with the specimen draw by preparing a heparinized syringe • Apply pressure immediately to the puncture site following the blood draw for 5 minutes, and for 10 minutes if the client is taking anticoagulants • Appropriately label the specimen and transport on ice to the laboratory • Record the client's temperature and the type of supplemental oxygen that the client is receiving on the laboratory form

PERFORMING THE ALLEN’S TEST • Apply direct pressure over the client's ulnar and radial arteries simultaneously • While pressure is applied, ask the client to open and close the hand repeatedly; the hand should blanch • Release pressure from the ulnar artery while compressing the radial artery and assess the color of the extremity distal to the pressure point • If pinkness fails to return within 6 seconds, the ulnar artery is insufficient, indicating that the radial artery should not be used for obtaining a blood specimen
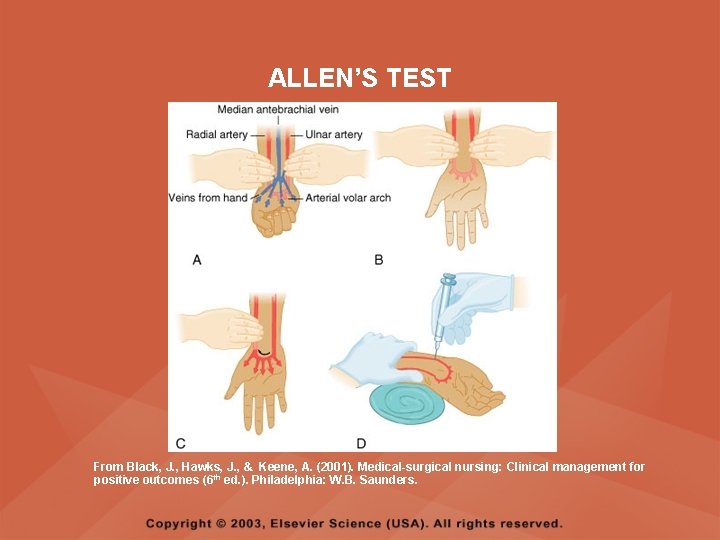
ALLEN’S TEST From Black, J. , Hawks, J. , & Keene, A. (2001). Medical-surgical nursing: Clinical management for positive outcomes (6 th ed. ). Philadelphia: W. B. Saunders.

ANALYZING ARTERIAL BLOOD GAS RESULTS • NORMAL ARTERIAL BLOOD GAS VALUES – p. H 7. 35– 7. 45 – PCO 2 35– 45 mm. Hg – HCO 3 22– 27 m. Eq/L – PO 2 80– 100 mm. Hg

ANALYZING ARTERIAL BLOOD GAS RESULTS • RESPIRATORY IMBALANCES – The respiratory function indicator is the PCO 2 – In a respiratory imbalance, you will find an opposite response between the p. H and the PCO 2; in other words, the p. H will be up with a PCO 2 down (alkalosis), or the p. H will be down with an elevated PCO 2 (acidosis) – Look at the p. H and the PCO 2 to determine if the condition is a respiratory problem

ANALYZING ARTERIAL BLOOD GAS RESULTS • RESPIRATORY IMBALANCES – Respiratory acidosis: The p. H is down; the PCO 2 is up – Respiratory alkalosis: The p. H is up; the PCO 2 is down

ANALYZING ARTERIAL BLOOD GAS RESULTS • METABOLIC IMBALANCES – The metabolic function indicator is the bicarbonate (HCO 3) – In a metabolic imbalance, you will find a corresponding response between the p. H and the HCO 3; in other words, the p. H will be up and the HCO 3 will be up (alkalosis), or the p. H will be down and the HCO 3 will be down (acidosis) – Look at the p. H and the HCO 3 to determine if the condition is a metabolic problem

ANALYZING ARTERIAL BLOOD GAS RESULTS • METABOLIC IMBALANCES – Metabolic acidosis: The p. H is down; the HCO 3 is down – Metabolic alkalosis: The p. H is up; the HCO 3 is up

ANALYZING ARTERIAL BLOOD GAS RESULTS • COMPENSATION – Respiratory acidosis and respiratory alkalosis – When compensation has occurred, the p. H will be within normal limits – The blood gas result reflects partial compensation if the HCO 3 is abnormal – The blood gas result reflects an uncompensated condition if the HCO 3 is normal

ANALYZING ARTERIAL BLOOD GAS RESULTS • COMPENSATION – Metabolic acidosis and metabolic alkalosis • When compensation has occurred, the p. H will be within normal limits • The blood gas result reflects partial compensation if the PCO 2 is abnormal • The blood gas result reflects an uncompensated condition if the PCO 2 is normal

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID POINTS • • In acidosis, the p. H is down In alkalosis, the p. H is up The respiratory function indicator is the PCO 2 The metabolic function indicator is the HCO 3

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID STEPS • PYRAMID STEP 1 – Look at the blood gas report. Look at the p. H. Is it up or down? If it is up, it reflects alkalosis. If it is down, it reflects acidosis.

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID STEPS • PYRAMID STEP 2 – Look at the PCO 2. Is it up or down? If it reflects an opposite response to the p. H, then you know that the condition is a respiratory imbalance. If it does not reflect an opposite response to the p. H, then move on to Pyramid Step 3.

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID STEPS • PYRAMID STEP 3 – Look at the HCO 3. Does the HCO 3 reflect a corresponding response with the p. H? If it does, then the condition is a metabolic imbalance.

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID STEPS • PYRAMID STEP 4 – Remember, compensation has occurred if the p. H is in a normal range of 7. 35– 7. 45 – If the p. H is not within normal range, look at the respiratory or metabolic function indicators

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID STEPS • STATE OF COMPENSATION AND RESPIRATORY IMBALANCES – If the condition is a respiratory imbalance, look at the HCO 3 to determine the state of compensation – If the HCO 3 is normal, then the condition is uncompensated. If the HCO 3 is abnormal, then the condition is partial compensation

ANALYZING ARTERIAL BLOOD GAS RESULTS PYRAMID STEPS • STATE OF COMPENSATION AND METABOLIC IMBALANCES – If the condition is a metabolic imbalance, look at the PCO 2 to determine the state of compensation – If the PCO 2 is normal, then the condition is uncompensated – If the PCO 2 is abnormal, then the condition is partial compensation
- Slides: 49