AcidBase Balance and Arterial Blood Gases Chapter 17

Acid-Base Balance and Arterial Blood Gases Chapter 17 Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Introduction • Maintain a steady balance between acids and bases to achieve homeostasis • Health problems lead to imbalance • Diabetes mellitus • Chronic obstructive pulmonary disease (COPD) • Kidney disease Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

p. H • Measure of H+ ion concentration • Blood is slightly alkaline at p. H 7. 35 to 7. 45 • <7. 35 is acidosis • >7. 45 is alkalosis Copyright © 2014 by Mosby, an imprint of Elsevier Inc.
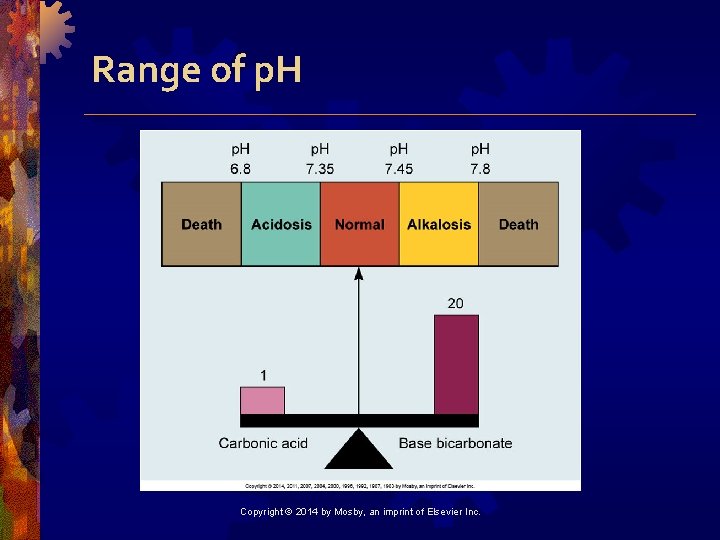
Range of p. H Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Acid-Base Regulation • Metabolic processes produce acids that must be neutralized and excreted • Regulatory mechanisms • Buffers : react immediately • Respiratory system : responds within minutes and reaches maximum effectiveness in hours • Renal system : response takes 2 -3 days maximum response, but kidneys can maintain balance indefinitely in chronic imbalances Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Buffer System • Act chemically to change strong acids to weak acids or bind acids • Carbonic acid–bicarbonate, phosphate, protein, and hemoglobin buffers • Also shifting of hydrogen in and out of cell Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Carbonic acid (H 2 CO 3)/Bicarbonate (HCO 3–) Buffer • HCl + Na. H 2 CO 3 Na. Cl + H 2 CO 3 • Strong acid + strong base is buffered into salt and weak acid Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Respiratory System Regulation • CO 2 + H 2 O H 2 CO 3 H++ HCO 3– • Respiratory center in medulla controls breathing • Increased respirations lead to increased CO 2 elimination and decreased CO 2 in blood • Decreased respirations lead to CO 2 retention Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Renal System Regulation • Conserves bicarbonate and excretes acid • Three mechanisms for acid elimination • Secrete free hydrogen • Combine H+ with ammonia (NH 3) • Excrete weak acids Copyright © 2014 by Mosby, an imprint of Elsevier Inc.
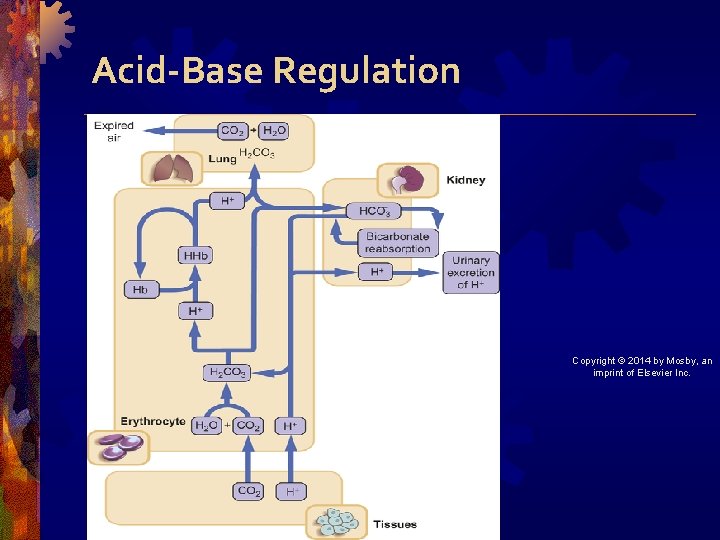
Acid-Base Regulation Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Alterations in Acid-Base Balance • Imbalances occur when compensatory mechanisms fail • Classification of imbalances : acid-base imbalance seen when ratio is 1: 20 between acid-base content is altered • Compensatory process may be inadequate because either the pathophysiologic process is overwhelming or there is insufficient time for compensation process to function • Imbalances include: • Respiratory (CO 2) or metabolic (HCO 3) • Acidosis or alkalosis Copyright © 2014 by Mosby, an imprint of Elsevier Inc. • Acute or chronic

Types of Acid-Base Imbalances • Resp imbalances affect carbonic acid concentration leading to respiratory acidosis (or increased carbonic acid) and decreased carbonic acid leading to respiratory alkalosis • Metabolic imbalances affect base bicarbonate , so decreased bicarb leads to metabolic acidosis and increased bicarb leads to metabolic alkalosis Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Clinical Manifestations of Acid-Base Imbalances • Based on p. H, not source of imbalance • Acidosis • Central nervous system (CNS) depression • Kussmaul respirations • Alkalosis • CNS irritability • Hypocalcemia. Copyright © 2014 by Mosby, an imprint of Elsevier Inc

Blood Gas Values • Arterial blood gas (ABG) values provide information about • Acid-base status • Underlying cause of imbalance • Body's ability to regulate p. H • Overall oxygen status Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Interpretation of ABGs • Diagnosis in six steps • Evaluate p. H : 7. 35 -7. 45 ; p. H is a measure of H+ ion concentration. p. H less than 6. 8 or greater than 7. 8 equals death • Analyze Pa. CO 2 35 -45 • Analyze HCO 3– 22 -26 • Determine if CO 2 or HCO 3– matches the alteration • Decide if the body is attempting to compensate : when compensatory mechanism fail we see imbalances which include resp-carbonic acid concentration and metabolic which affect bicarb Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Normal Blood Gas Values Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Acid-Base Mnemonic—ROME • Respiratory • Opposite • Alkalosis • Acidosis ↑ p. H ↓ Pa. CO 2 ↓ p. H ↑ Pa. CO 2 • Metabolic • Equal • Acidosis • Alkalosis ↓ p. H ↓ HCO 3 ↑ p. H ↑ HCO 3 Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Sample ABG Interpretation Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 1: Jeri i. Stockphoto/Thinkstockk • Jeri is a 22 -year-old female who has been on a 3 -day party binge. • Her friends bring her to the ED after being unable to awaken her. • Assessment reveals shallow respirations with a rate of 8/min, diminished breath sounds, and decreased level of consciousness. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 1: Jeri 1. What type of acid-base imbalance would you expect Jeri to have? 2. What is causing it? 3. What type of compensation would you expect or not expect? Explain. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 1: Jeri i. Stockphoto/Thinkstock 1. What would Jeri's ABGs look like? 2. What is the treatment? 3. See next slide Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Respiratory Acidosis • Carbonic acid excess caused by • Hypoventilation : increased CO 2 • Respiratory failure • Compensation • Kidneys conserve HCO 3– and secrete H+ into urine Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 2: Mayna Brand X Pictures/Thinkstock • Mayna is an 18 -year-old female who presents to the ED after a sexual assault. She is hysterical and in severe emotional distress. • Her blood pressure is 140/96, heart rate 104, respiratory rate 38, and oxygen saturation 96%. Lung sounds are clear. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 2: Mayna Brand X Pictures/Thinkstock 1. What type of acid-base imbalance would you expect Mayna to have? 2. What is causing it? 3. What type of compensation would you expect or not expect? Explain. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 2: Mayna Brand X Pictures/Thinkstock 1. What do Mayna's ABGs look like? 2. What is the treatment? 3. See next slide Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Respiratory Alkalosis • Carbonic acid deficit caused by • Hypoxemia from acute pulmonary disorders • Hyperventilation • Primary cause is hypoxemia from acute pulmonary disorders; anxiety, CNS disorders and mechanical overventilation; also increase vent rate and decreased Pa. CO 2 • Compensation • Rarely occurs when acute : renal excretion of HCO 3 but not enough time to compensate • Can buffer with bicarbonate shift • Renal compensation if chronic Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 3: Alan • Alan is a 17 -year-old male who comes to the clinic with c/o feeling “bad, ” fatigue, constant thirst, and frequent urination. • Focused assessment reveals rapid deep respirations (rate 28) with a fruity breath odor. • A capillary blood glucose is 484 mg/d. L. i. Stockphoto/Thinkstock

Case Study 3: Alan i. Stockphoto/Thinkstock 1. What type of acid-base imbalance would you expect Alan to have? 2. What is causing it? 3. What type of compensation would you expect or not expect? Explain. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 3: Alan i. Stockphoto/Thinkstock 1. What will Alan's ABGs look like? 2. What is the treatment? 3. See next slide Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Metabolic Acidosis • Excess carbonic acid or base bicarbonate deficit caused by : • Ketoacidosis • Lactic acid accumulation (shock) • Severe diarrhea : loss of bicarb • Kidney disease : inability to reabsorb bicarb and secrete H+ • Compensatory metabolic acidosis –body attempts to rid acid by Kussmauls breathing and blowing off CO 2 and kidneys excrete extra H+ Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Metabolic Acidosis • Compensatory mechanisms • Increased CO 2 excretion by lungs • Kussmaul respirations (deep and rapid) • Kidneys excrete acid • Anion gap • Na+ – (Cl – + HCO 3–) • Normal: 10– 14 mmol/L • Increased with acid gain Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 4: Anthony Banana. Stock/Thinkstock • Anthony is a 54 -year-old male with a history of nausea and vomiting for the past week. • He has been self-medicating himself with baking soda to control his abdominal discomfort. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 4: Anthony 1. What type of acid-base imbalance would you expect Anthony to have? 2. What is causing it? 3. What type of compensation would you expect or not expect? Explain. Banana. Stock/Thinkstock Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Case Study 4: Anthony Banana. Stock/Thinkstock 1. What will Anthony's ABGs look like? 2. What is the treatment? 3. See next slide Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Metabolic Alkalosis • Base bicarbonate excess caused by • Prolonged vomiting or gastric suction • Gain of HCO 3– : ingestion of baking soda • Compensatory mechanisms • Renal excretion of HCO 3– • Decreased respiratory rate to increase plasma CO 2 (limited) Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Practice ABG Interpretation Case 1 • What imbalance is this? • p. H 7. 33 • Pa. CO 2 67 mm Hg • Pa. O 2 47 mm Hg • HCO 3 37 m. Eq/L Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Practice ABG Interpretation Case 2 • What imbalance is this? • p. H 7. 18 • Pa. CO 2 38 mm Hg • Pa. O 2 70 mm Hg • HCO 3– 15 m. Eq/L Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Practice ABG Interpretation Case 3 • What imbalance is this? • p. H 7. 60 • Pa. CO 2 30 mm Hg • Pa. O 2 60 mm Hg • HCO 3– 22 m. Eq/L Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Practice ABG interpretation Case 4 • What imbalance is this? • p. H 7. 58 • Pa. CO 2 35 mm Hg • Pa. O 2 75 mm Hg • HCO 3– 50 m. Eq/L Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Practice ABG Interpretation Case 5 • What imbalance is this? • p. H 7. 28 • Pa. CO 2 28 mm Hg • Pa. O 2 70 mm Hg • HCO 3– 18 m. Eq/L Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

ABG Analysis Case 6 • ABG results are as follows: • p. H 7. 20 • Pa. CO 2 58 mm Hg • Pa. O 2 59 mm Hg • HCO 3– 24 m. Eq/L • Describe a patient who would have these ABGs, including history, assessment, and treatment. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

ABG Analysis Case 7 • ABG results are as follows: • p. H 7. 39 • Pa. CO 2 38 mm Hg • Pa. O 2 44 mm Hg • HCO 3– 24 m. Eq/L • Describe a patient who would have these ABGs, including history, assessment, and treatment. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

ABG Case Analysis Case 6 • ABG results are as follows: • p. H 7. 36 • Pa. CO 2 58 mm Hg • Pa. O 2 50 mm Hg • HCO 3 – 33 m. Eq/L • Describe a patient who would have these ABGs, including history, assessment, and treatment. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

ABG Analysis Case 7 • ABG results are as follows: • p. H 7. 50 • Pa. CO 2 28 mm Hg • Pa. O 2 85 mm Hg • HCO 3 – 24 m. Eq/L • Describe a patient who would have these ABGs, including history, assessment, and treatment. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

ABG Case Analysis Case 8 • ABG results are as follows: • p. H 7. 20 • Pa. CO 2 28 mm Hg • Pa. O 2 81 mm Hg • HCO 3– 18 m. Eq/L • Describe a patient who would have these ABGs, including history, assessment, and treatment. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

ABG Analysis Case 9 • ABG results are as follows: • p. H 7. 57 • Pa. CO 2 46 mm Hg • Pa. O 2 87 mm Hg • HCO 3– 38 m. Eq/L • Describe a patient who would have these ABGs, including history, assessment, and treatment. . Copyright © 2014 by Mosby, an imprint of Elsevier Inc

Audience Response Question A patient with an acid-base imbalance has an altered potassium level. The nurse recognizes that the potassium level is altered because a. Potassium is returned to extracellular fluid when metabolic acidosis is corrected. b. Hyperkalemia causes an alkalosis that results in potassium being shifted into the cells. c. Acidosis causes hydrogen ions in the blood to be exchanged for potassium from the cells. d. In alkalosis, potassium is shifted into extracellular fluid to bind excessive bicarbonate. Copyright © 2014 by Mosby, an imprint of Elsevier Inc.

Audience Response Question A patient has the following ABG results: p. H 7. 48, Pa. O 2 86 mm Hg, Pa. CO 2 44 mm Hg, HCO 3 29 m. Eq/L. When assessing the patient, the nurse would expect the patient to have a. Muscle cramping b. Warm, flushed skin c. Respiratory rate of 36 d. Blood pressure of 94/52 Copyright © 2014 by Mosby, an imprint of Elsevier Inc.
- Slides: 48