AcidBase Balance AcidBase Disturbances ASSOC PROF DR SUAT

Acid-Base Balance &Acid-Base Disturbances ASSOC. PROF. DR. SUAT BIÇER YEDITEPE UNIVERSITY FACULTY OF MEDICINE DEPARTMENT OF CHILD HEALTH AND PEDIATRICS SUBDIVISION OF PEDIATRIC EMERGENCY

p. H of arterial blood is maintained between 7. 38 and 7. 42 Acid-base balance is maintained by interaction of the lungs, kidneys, and systemic buffering systems. Disturbances in acid-base balance are initially stabilized by chemical buffering, compensated for by pulmonary or renal regulation of CO 2 or , and ultimately corrected when the primary cause of the acid-base disturbance is eliminated.

Renal regulation of acid-base balance is accomplished by the reabsorption of filtered primarily in the proximal tubule, and the excretion of H+ or in the distal nephron to match the net input of acid or base. Alkalinization of the urine may occur when an absolute or relative excess of bicarbonate exists. However, urinary alkalinization will not occur if there is a deficiency of Na+ or K+, because must also be retained to maintain electroneutrality

In contrast, the urine may be acidified if an absolute or relative decrease occurs in systemic. In this setting, proximal tubular reabsorption and distal tubular H+ excretion are maximal. A “paradoxical aciduria” with low urinary p. H may be seen in the setting of hypokalemic metabolic alkalosis and systemic K+ depletion wherein H+ is exchanged and excreted in preference to K+ in response to mineralocorticoid.
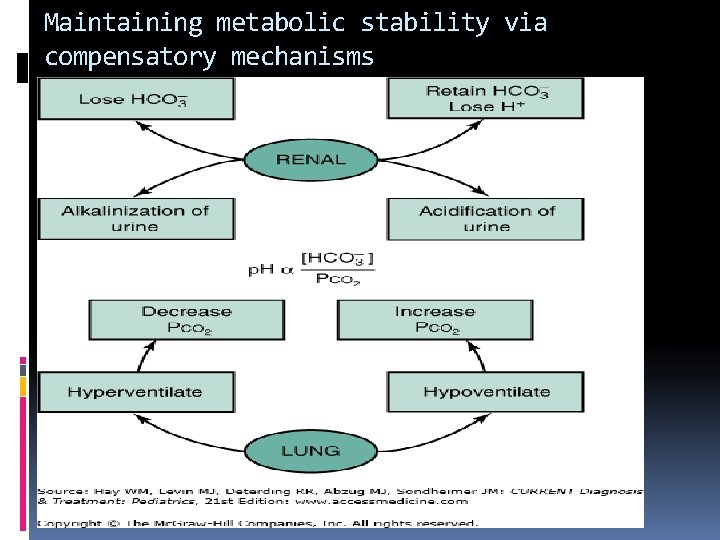
Maintaining metabolic stability via compensatory mechanisms

Acid-Base Disturbances When evaluating a disturbance in acid-base balance, the systemic p. H, partial carbon dioxide pressure (PCO 2), serum HCO 3 , and anion gap must be considered. The anion gap, Na+ − (Cl− + HCO 3), is an expression of the unmeasured anions in the plasma and is normally 12 ± 4 m. Eq/L. An increase above normal suggests the presence of an unmeasured anion, such as occurs in diabetic ketoacidosis, lactic acidosis, salicylate intoxication, and so on.

base excess (or deficit) this expression of acid-base balance is influenced by the renal response to respiratory disorders and cannot be interpreted independently (as in a compensated respiratory acidosis, wherein the base excess may be quite large).

Metabolic Acidosis a primary decrease in serum HCO 3 and systemic p. H due to the loss of HCO 3 from the kidneys or gastrointestinal tract, the addition of an acid (from external sources or via altered metabolic processes), the rapid dilution of the ECF with non-bicarbonate– containing solution (usually normal saline).

When HCO 3 is lost through the kidneys or gastrointestinal tract, Cl− must be reabsorbed with Na+ disproportionately, resulting in a hyperchloremic acidosis with a normal anion gap. Thus, a normal anion gap acidosis in the absence of diarrhea or other bicarbonate-rich gastrointestinal losses suggests the possibility of renal tubular acidosis and should be evaluated appropriately.

In contrast, acidosis that results from addition of an unmeasured acid is associated with a widened anion gap. Examples are diabetic ketoacidosis, lactic acidosis, starvation, uremia, toxin ingestion (salicylates, ethylene glycol, or methanol), and certain inborn errors of organic or amino acid metabolism. Dehydration may also result in a widened anion gap acidosis as a result of inadequate tissue perfusion, decreased O 2 delivery, and subsequent lactic and keto acid production. Respiratory compensation is accomplished through an increase in minute ventilation and a decrease in PCO 2. The patient’s history, physical findings, and laboratory features should lead to the appropriate diagnosis.

The ingestion of unknown toxins or the possibility of an inborn error of metabolism must be considered in children without an obvious cause for a widened anion gap acidosis. Ethylene glycol (eg, antifreeze) is particularly worrisome because of its sweet taste and accounts for a significant number of toxin ingestions. Salicylate intoxication has a stimulatory effect on the respiratory center of the CNS; thus, patients may initially present with respiratory alkalosis or mixed respiratory alkalosis and widened anion gap acidosis.

Most types of metabolic acidosis will resolve with correction of the underlying disorder, improved renal perfusion, and acid excretion. IV Na. HCO 3 administration may be considered in the setting of metabolic acidosis when the p. H < 7. 0 or the HCO 3 < 5 m. Eq/L, but only if adequate ventilation is ensured. The dose (in milliequivalents) of Na. HCO 3 may be calculated as: and given as a continuous infusion over 1 hour. The effect of Na. HCO 3 in lowering serum potassium and ionized calcium concentrations must also be considered and monitored.

Metabolic Alkalosis A primary increase in HCO 3 and p. H resulting from a loss of strong acid or gain of buffer base. The loss of gastric juice via nasogastric suction or vomiting. Cl−-responsive alkalosis, characterized by a low urinary [Cl−] (< 20 m. Eq/L) indicative of a volume-contracted state that will be responsive to the provision of adequate Cl− salt (usually in the form of normal saline). Cystic fibrosis may also be associated with a Cl−-responsive alkalosis due to the high losses of Na. Cl through the sweat, Congenital Cl−-losing diarrhea is a rare cause of Cl−-responsive metabolic alkalosis.
![Chloride-resistant alkaloses are characterized by a urinary [Cl−] greater than 20 m. Eq/L Chloride-resistant alkaloses are characterized by a urinary [Cl−] greater than 20 m. Eq/L](http://slidetodoc.com/presentation_image_h2/004d03429c2b767039aead699a81ea29/image-14.jpg)
Chloride-resistant alkaloses are characterized by a urinary [Cl−] greater than 20 m. Eq/L and include: Bartter syndrome, Cushing syndrome, and primary hyperaldosteronism, conditions associated with primary increases in urinary [Cl−], or volume-expanded states lacking stimuli for renal Cl− reabsorption.
![Urinary [Cl−] and serum [K+] Thus, the urinary [Cl−] is helpful in distinguishing the Urinary [Cl−] and serum [K+] Thus, the urinary [Cl−] is helpful in distinguishing the](http://slidetodoc.com/presentation_image_h2/004d03429c2b767039aead699a81ea29/image-15.jpg)
Urinary [Cl−] and serum [K+] Thus, the urinary [Cl−] is helpful in distinguishing the nature of a metabolic alkalosis. The serum [K+] is also low in these settings (hypokalemic metabolic alkalosis) owing to a combination of: increased mineralocorticoid activity associated with volume contraction, the shift of K+ to the ICF compartment, preferential reabsorption of Na+ rather than K+ to preserve intravascular volume, primary mineralocorticoid excess.

Respiratory Acidosis develops when alveolar ventilation is decreased, increasing PCO 2 and lowering systemic p. H. The kidneys compensate for respiratory acidosis by increasing HCO 3 reabsorption, a process that takes several days to fully manifest. air hunger with retractions and the use of accessory respiratory muscles

Respiratory acidoses occur in: upper or lower airway obstruction, ventilation-perfusion disturbances, CNS depression, neuromuscular defects. • Hypercapnia is not as detrimental as the hypoxia that usually accompanies these disorders. • The goal of therapy is to correct or compensate for the underlying pathologic process to improve alveolar ventilation. • Bicarbonate therapy is not indicated in a pure respiratory acidosis, because it will worsen the acidosis by shifting the equilibrium of the carbonic acid–bicarbonate buffer system to increase PCO 2.

Respiratory Alkalosis Occurs when hyperventilation results in a decrease in PCO 2 and an increase in systemic p. H. Depending on the acuity of the respiratory alkalosis, there may be an associated compensatory loss of bicarbonate by the kidneys manifested as a low serum bicarbonate level and a normal anion gap that may be misinterpreted as a normal anion gap acidosis if all acid-base parameters are not considered. Patients may experience tingling, paresthesias, dizziness, palpitations, syncope, or even tetany and seizures due to the associated decrease in ionized calcium. Causes of respiratory alkalosis include psychobehavioral disturbances, CNS irritation from meningitis or encephalitis, salicylate intoxication, and iatrogenic over ventilation in patients who are intubated and mechanically ventilated. Therapy is directed toward the causal process. Rebreathing into a paper bag will decrease the severity of symptoms in acute hyperventilation.

Diagnosis A systematic evaluation of an arterial blood gas sample, combined with the clinical history, can usually explain the patient's acidbase disturbance. Assessment of an arterial or venous or capillary blood gas sample requires knowledge of normal values (Table 2). In most cases, this is accomplished via a 3 step process (Fig. -2):

1 - Determine whether acidemia or alkalemia is present. 2 - Determine a cause of the acidemia or alkalemia. 3 - Determine whether a mixed disorder is present. p. H [HCO 3−] PCO 2 7. 35 -7. 45 20 -28 m. Eq/L 35 -45 mm Hg

References Adrogue JA, Madias NE: Management of life-threatening acid–base disorders: Parts 1 and 2. N Engl J Med 1998; 338: 26, 107. [Pub. Med: 9414329] Avner ED: Clinical disorders of water metabolism: Hyponatremia and hypernatremia. Pediatr Ann 1995; 24: 23. [Pub. Med: 7715960] Choong K et al: Hypotonic versus isotonic maintenance fluids after surgery for children: A randomized controlled trial. Pediatrics 2011; 128: 980– 983. [Pub. Med: 22007013] Fall PJ: A stepwise approach to acid-base disorders. Practical patient evaluation for metabolic acidosis and other conditions. Postgraduate Med 2000; 107: 249. [Pub. Med: 10728149] Finberg L et al: Water and Electrolytes in Pediatrics: Physiology, Pathophysiology and Treatment, 2 nd ed. WB Saunders, 1993. Friedman A: Fluid and electrolyte therapy: A primer. Pediatr Nephrol 2009; 7: 1189. [Pub. Med: 19444484] Gunnerson KJ, Kellum JA: Acid-base and electrolyte analysis in critically ill patients: Are we ready for the new millennium? Curr Opin Crit Care 2003; 9: 468. [Pub. Med: 14639065] Hellerstein S: Fluids and electrolytes: Physiology. Pediatr Rev 1993; 14: 70. [Pub. Med: 8493184] Jospe N, Forbes G: Fluids and electrolytes: Clinical aspects. Pediatr Rev 1996; 17: 395. [Pub. Med: 8937172]. Kappy MS, Ganong CA: Cerebral salt wasting in children: The role of atrial natriuretic hormone. Adv Pediatr 1996; 43: 271. [Pub. Med: 8937172] Liebelt EL: Clinical and laboratory evaluation and management of children with vomiting, diarrhea, and dehydration. Curr Opin Pediatr 1998; 10: 461. [Pub. Med: 9818241] Mc. Donald RA: Disorders of potassium balance. Pediatr Ann 1995; 24: 31. [Pub. Med: 7715961] Moritz AL, Ayus JC: Prevention of hospital-acquired hyponatremia: A case for using isotonic saline. Pediatrics 2003; 111: 227. [Pub. Med:
- Slides: 21