Acidbase and ABG interpretation Carlos A Camal Sanchez

Acid-base and ABG interpretation Carlos A. Camal Sanchez DNP NCLIN 411

Normal parameters: p. H = 7. 35 – 7. 45 PCO 2 = 35 – 45 mm Hg This is your acid, your respiratory component (takes 45 s to 2 min for lungs to compensate) HCO 3 = 22 – 26 m. Eq/L This is your base, your metabolic component (takes 6 to 12 hrs fully developed after a few days for kidneys to compensate)

CO 2 and HCO 3 have a reverse reaction and equilibrium CO 2 + H 2 O H 2 CO 3 H + HCO 3 This is the way kidney excrete or retain HCO 3 Or CO 2 + H 2 O H 2 CO 3 H + HCO 3 This is the way to eliminate CO 2 through the lungs
![Basic Concepts: Hydrogen Ion [H+] in extracellular fluid is determined by balance between: -Partial Basic Concepts: Hydrogen Ion [H+] in extracellular fluid is determined by balance between: -Partial](http://slidetodoc.com/presentation_image_h/88f79dda478c3e67555dc3c4d638f73a/image-4.jpg)
Basic Concepts: Hydrogen Ion [H+] in extracellular fluid is determined by balance between: -Partial pressure of carbon dioxide (PCO 2) Normal 40 mm Hg arterial blood -Concentration of Bicarbonate (HCO 3) Normal 24 m. Eq/L arterial blood

Basic Concepts: For example Hydrogen ion concentration expressed as follows in nanoequivalents per liter because represents amount, and expressd in p. H units: [H+] (n. Eq/L) = 24 x (PCO 2 / HCO 3) = 24 x (40/24) = 40 n. Eq/L
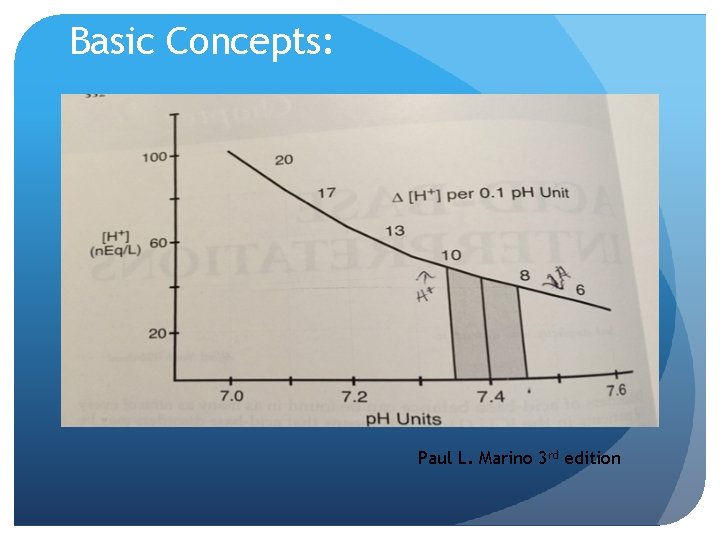
Basic Concepts: Paul L. Marino 3 rd edition
![Basic Concepts: There is a relationship between p. H and [H+] A normal [H+] Basic Concepts: There is a relationship between p. H and [H+] A normal [H+]](http://slidetodoc.com/presentation_image_h/88f79dda478c3e67555dc3c4d638f73a/image-7.jpg)
Basic Concepts: There is a relationship between p. H and [H+] A normal [H+] of 40 n. Eq/L corresponds to a p. H of 7. 40

Normal parameters: p. H = 7. 35 – 7. 45 PCO 2 = 35 – 45 mm Hg This is your acid, your respiratory component (takes 45 s to 2 min for lungs to compensate) HCO 3 = 22 – 26 m. Eq/L – This is your base, your metabolic component (takes 6 to 12 hrs fully developed after a few days for kidneys to compensate)

For example: of PCO 2 refers to respiratory acidosis of PCO 2 refers to respiratory alkalosis of HCO 3 refers to metabolic acidosis Any changes of values outside the normal range, you may use the suffix -emia -Acidemia p. H < 7. 36 -Alkalemia p. H >7. 44

Remember that p. H requires a constant PCO 2 / HCO 3 ration of 40 n. Eq/L
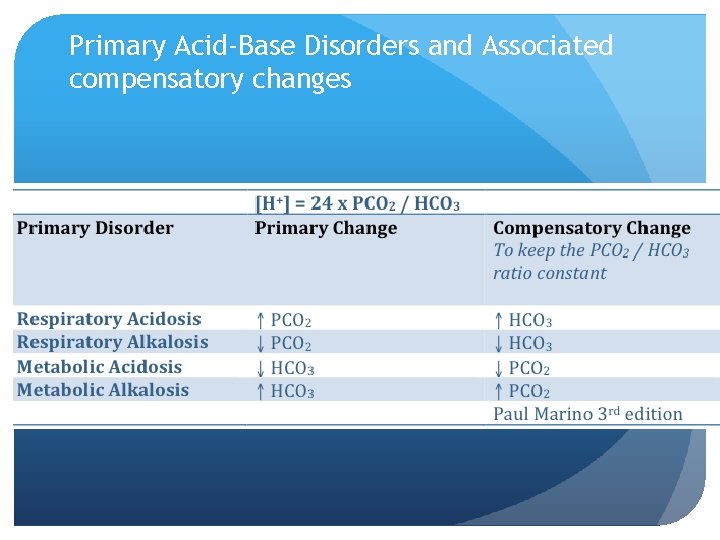
Primary Acid-Base Disorders and Associated compensatory changes

Respiratory Compensation Peripheral chemoreceptors located: In the carotid body at carotid bifurcation in the neck Metabolic acidosis stimulate the chemoreceptors - ventilation and arterial PCO 2 (Pa. CO 2) Metabolic alkalosis silences the chemoreceptors - ventilation and arterial Pa. CO 2 Respiratory Acidosis: p. H ↓ 7. 35 CO 2↑ 45 HCO 3 22 -26 normal or↑ Respiratory Alkalosis: p. H↑ 7. 45 CO 2↓ 35 HCO 3↓ 22 -26 or normal

Metabolic Compensation A primary change in Pa. CO 2 takes place in the kidneys And adjustment in HCO 3 reabsorption in the proximal tubules Pa. CO 2 (respiratory acidosis) results in HCO 3 Pa. CO 2 (respiratory alkalosis) results in decreased HCO 3

Systematic approach to Acid-Base interpretation: 1. p. H normal? p. H low acidemia, p. H high alkalemia 2. PCO 2 normal? If p. H and PCO 2 are opposite direction, therefore it is primary respiratory disorder -If both p. H and Pa. CO 2 same direction or ; therefore is primary metabolic disorder 3. HCO 3 normal? HCO 3 < 22; therefore the patient is acidotic HCO 3 >26, then patient is alkalotic 4. If Pa. CO 2 and HCO 3 opposite direction of p. H: Compensation when: - HCO 3 is opposite from p. H, as evidence of compensation from metabolic system -p. H acidotic, Pa. CO 2 acidotic, and HCO 3 alkalotic

Why Acid-Base interpretation and Nursing Consideration: Vomiting, What GI Content is loss? Acid Diarrhea, What GI content loss? HCO 3

Causes of respiratory Acidosis? Patient will not breathe D/T CNS depression Analgesia, sedatives: (naloxone to reverse) Or Head injury Brainstem stroke CNS infection Neuromuscular weakness/illness: -Myasthenia gravis -Guillain-barre -ALS Chronic lung disease: CODP, emphysema,

Causes of respiratory Alkalosis: Pain, anxiety, fever ASA toxicity Pregnancy Liver failure Mechanical ventilation

Let's Review ABGs

Questions? References: 1. Marino, Paul. The ICU Book 3 rd Edition 2. A primer on Arterial Blood Gas Analysis by Andrew M. Luks, MD https: //courses. washington. edu/med 610/abg_primer. html 3. Frendl G. , Urman Richard D. Pocket Notebook ICU, 2013
- Slides: 19