Acid Rain BISC 312 Onkar Bains Simon Fraser

Acid Rain BISC 312 Onkar Bains Simon Fraser University

What is acid rain? n n “Acid rain” is a broad term used to describe several ways that acids fall out of the atmosphere A more precise term is acid deposition, which has two parts: wet and dry • Wet deposition refers to acidic rain, fog, and snow • Dry deposition refers to acidic gases and particles

What is acid rain? n The father of acid rain research is an Englishman from Manchester named Dr. Robert Angus Smith (1817 -1884) • World’s first climatologist to identify acid rain and its dangers • Active campaigner for the prohibition of the use of fuels that produced smoke • Published a number of books, such as Acid Rain: The Beginnings of Chemical Climatology, which were invaluable in raising awareness and educating others about dangers of smoky fuels and acid rain

Sources of acid rain n Primary sources: • Sulphur dioxide (SO 2) emissions • Nitrous oxides (NOx) emissions

SO 2/NOx emissions n Mostly comes from large combustion power plants (majority of which burn coal) • When fossil fuels are burned: sulfur dioxide (SO 2) and nitrogen oxides (NOx) are emitted • Coal accounts for most U. S. sulfur dioxide (SO 2) emissions and a large portion of NOx emissions n n Sulfur is present in coal as an impurity, and it reacts with air when the coal is burned to form SO 2 In contrast, NOx is directly formed when any fossil fuel is burned From: www. geocities. com/ whatsacidrain/pictures. html

SO 2/NOx emissions n Other sources include: • Combustion of fuels in motor vehicles • Residential and commercial furnaces • Agricultural waste burning • Forest fires • Volcanic action—primarily SO 2 • Lightning—NOx
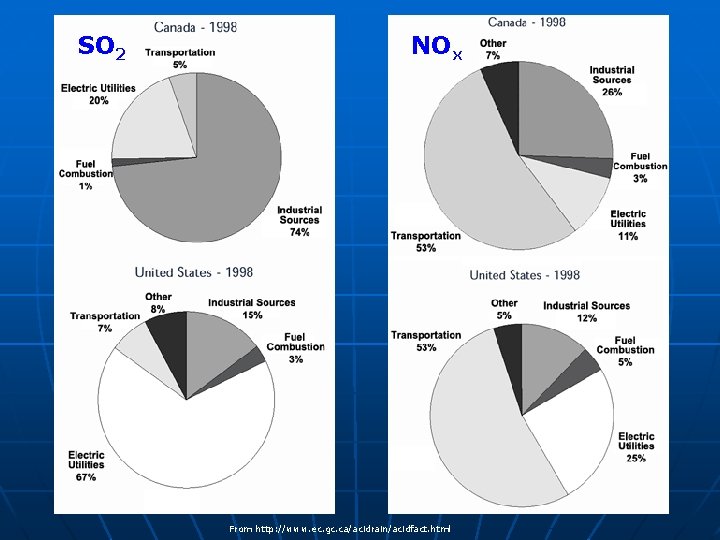
SO 2 NOx From http: //www. ec. gc. ca/acidrain/acidfact. html
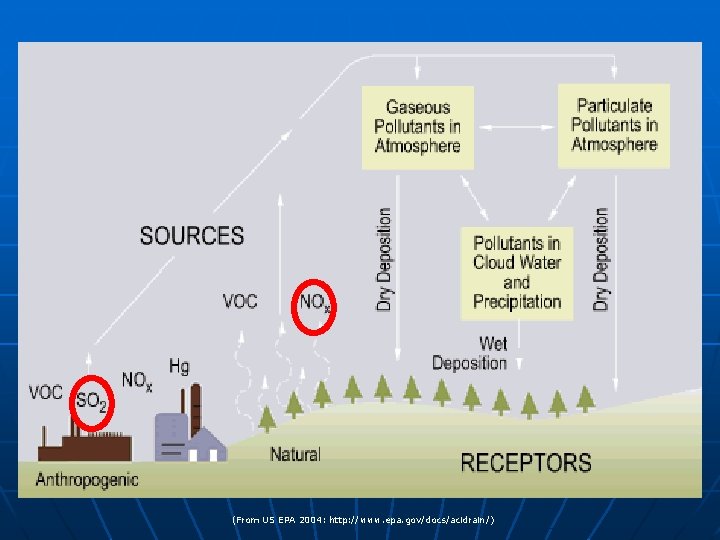
(From US EPA 2004: http: //www. epa. gov/docs/acidrain/)

How do we measure acid rain? n Acid rain is measured using a scale called "p. H. " The lower a substance's p. H, the more acidic it is: • Pure water is neutral with a p. H of 7 • Pure rain has a p. H of 5. 6, due to the carbon dioxide naturally present within the atmosphere. This CO 2 interacts with water to form carbonic acid in water • Automobile battery acid has a p. H of 1 • Vinegar has a p. H of 3 • As of the year 2000, the most acidic rain falling in the US has a p. H of about 4. 3. (US EPA 2004)

What contributes to acidity? n Inorganic acids that contribute to the acidity of rain: • H 2 SO 4: sulfuric acid is generated from anthropogenic SO 2 emissions and natural sulfur emissions. The latter includes, hydrogen sulfide (H 2 S) and dimethyl sulfide (DMS), which are oxidized to sulfur dioxide. • HNO 3: nitric acid is derived from both anthropogenic and natural NOx emissions.

What contributes to acidity? • HCl: hydrochloric acid has significant natural (volcanoes) and anthropogenic sources. Always secondary in importance to sulfuric and nitric acid in precipitation • H 2 CO 3: carbonic acid is negligible in acid rain. Source is CO 2, mostly natural, but has increased due to fossil fuel burning • H 2 SO 3: sulfurous acid makes a negligible contribution to acidic precipitation

What contributes to acidity? n Organic acids that contribute to the acidity of rain: • HCOOH: formic acid, the most common organic acid found in precipitation, is a by-product of the oxidation of more complex hydrocarbons • CH 3 COOH: acetic acid is generated as a by-product of organic photochemical decomposition in air • C 3 H 7 COOH: butyric acid is not very common in precipitation • C 5 H 11 COOH: caproic acid is not found in very large amounts in precipitation

General adverse effects n n n From www. geocities. com/ whatsacidrain/pictures. html Damage to lakes and to fish stocks arising from acid rain is well documented Damage to forests, plants and limestone buildings Acid rain may have a secondary effect on health by releasing heavy metals into drinking water

Effects—aquatic organisms n Acid rain causes a cascade of effects that: • harm or kill individual aquatic organisms • reduce population numbers • completely eliminate species from a water body • decrease biodiversity n n Freshwater shrimps, snails, mussels are the most quickly affected by acidification followed by fish such as minnows and salmon The roe and fry (eggs and young) of the fish are the worst affected • Acidity of water can cause deformity in young fish and can prevent eggs from hatching properly
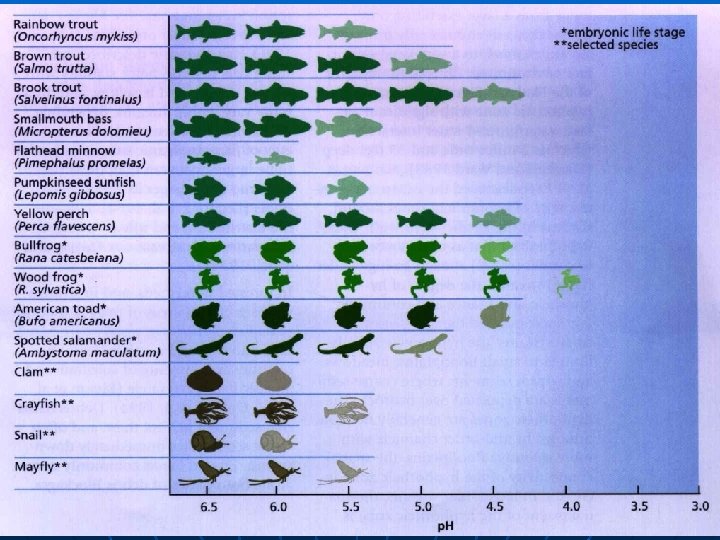

Effects—aquatic organisms n Acidity of the water does not just affect species directly, it also causes toxic substances like aluminium to be released into water from soil, harming fish and other aquatic animals • Example—using fish n n Metals like Al and Cu irritate gills of fish In response fish create a film of mucus in gills to stop this irritation until the irritant is gone However Al and Cu does not go away and fish continues to build up more mucus to counteract irritants Eventually there is so much mucus that it clogs the gills fish can no longer breath death


Effects—trees and plants n Acid rain can effect trees in several different ways, it may: • dissolve and wash away nutrients and minerals in soil which help trees to grow • cause release of harmful substances such as aluminum into soil • wear away waxy protective coating of leaves, damaging them and preventing them from being able to photosynthesize properly n n A combination of these effects weakens the trees Easily attacked by diseases and insects or injured by bad weather It is not just trees that are affected by acid rain, other plants may also suffer From www. geocities. com/ whatsacidrain/pictures. html

Effects—trees and plants n Soils may neutralize some or all of the acidity of the acid rainwater • This ability is called buffering capacity, and without it, soils become more acidic n Ability of forest soils to resist, or buffer, acidity depends on the thickness as well as type of bedrock beneath forest floor • If bedrock consists of limestone, effect is not so severe (neutralizes acid) • If bedrock is granite, sandstone or basalt, there is no buffer n n Midwestern states like Nebraska and Indiana have soils that are well buffered Places in the mountainous northeast, like New York's Adirondack and Catskill Mountains, have thin soils with low buffering capacity From www. geocities. com/ whatsacidrain/pictures. html

Effects—food crops n Most farm crops are less affected by acid rain than are forests • The deep soils of many farm regions, such as those in the Midwestern U. S. , can absorb and neutralize large amounts of acid • Mountain farms are more at risk—the thin soils in these higher elevations cannot neutralize so much acid n n Food crops are not usually seriously affected because farmers frequently add fertilizers to soil in order to replace nutrients that have washed away They may also add crushed limestone to soil
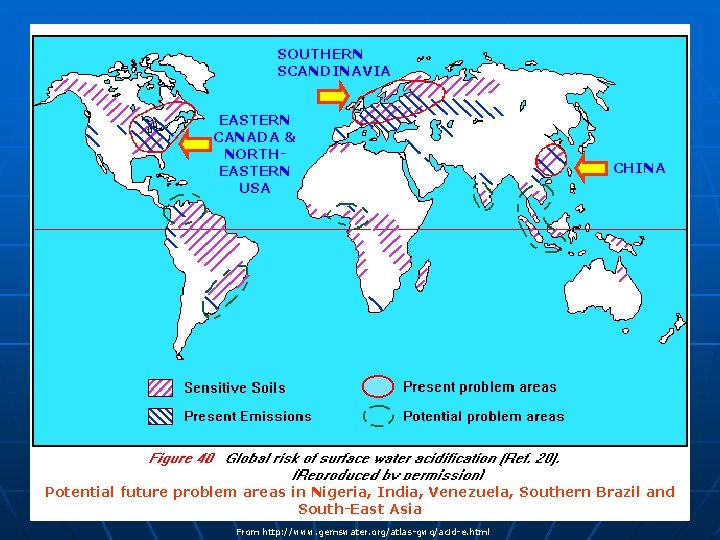
SOUTHERN SCANDINAVIA EASTERN CANADA & NORTHEASTERN USA CHINA Potential future problem areas in Nigeria, India, Venezuela, Southern Brazil and South-East Asia From http: //www. gemswater. org/atlas-gwq/acid-e. html

2003 Progress Report (U. S. ) n US EPA Acid Rain Program 2003 Progress Report n Over the last decade: • Ambient SO 2 and sulfate levels are down more than 40% and 30%, respectively, in the eastern U. S. • Wet sulfate deposition, which acidifies sensitive lakes, streams and forest soils, has decreased 39% in the northeastern U. S. and 17% in the southeastern U. S. • Some modest reductions in inorganic nitrogen deposition and wet nitrate concentrations have occurred in the Northeast and Mid-Atlantic regions, but other areas have not shown much improvement. • Signs of recovery in acidified lakes and streams are evident in the Adirondacks, the northern Appalachian Plateau, and the upper Midwest. These signs include lower concentrations of sulfates, nitrates, and improvements in acid neutralizing capacity.
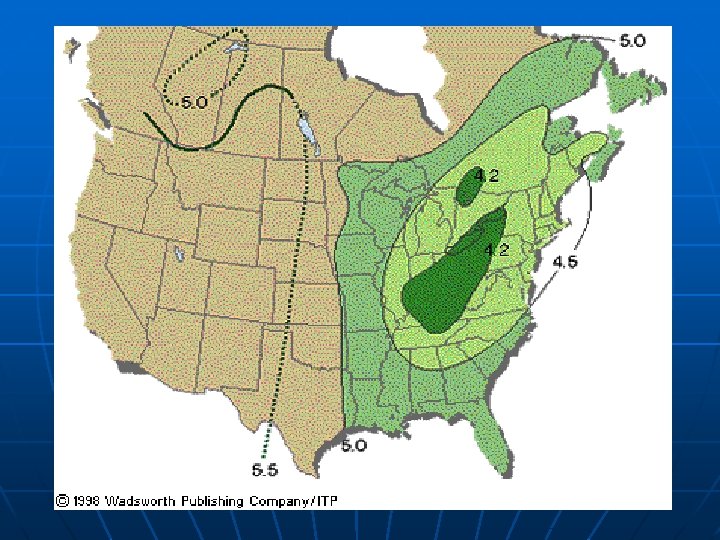

Acid rain in Canada n n Acid rain is a problem in eastern Canada because many of the water and soil systems in this region lack natural alkalinity - such as a lime base - and therefore cannot neutralize acid naturally Provinces that are part of the Canadian Precambrian Shield, like Ontario, Quebec, New Brunswick and Nova Scotia, are hardest hit In fact, more than half of Canada consists of susceptible hard rock (i. e. , granite) areas that cannot neutralize the effects of acid rain If the water and soil systems were more alkaline-as in western Canada-they could neutralize or "buffer" against acid rain naturally

Have SO 2 emission levels decreased in Canada? n n Initiated in 1985, the Eastern Canada Acid Rain program committed Canada to cap SO 2 emissions in the seven provinces from Manitoba eastward at 2. 3 million tonnes by 1994, a 40% reduction from 1980 levels By 1994, all seven provinces had achieved or exceeded their targets In 1998, the provinces, territories and the federal government signed The Canada-Wide Acid Rain Strategy for Post-2000, committing them to further actions to deal with acid rain In 1999, the most recent year for which data is available, emissions in eastern Canada totalled just 1. 61 million tonnes - 30% below the 2. 3 -million tonne cap and a 58% reduction from 1980 levels.

Have NOx emission levels decreased in Canada? n n Canadian NOX emissions have been relatively stable since the late 1980 s In 1999, total NOX emissions from anthropogenic sources were just under 2. 2 million tonnes

Controlling acid deposition n Clean up smokestacks and exhaust pipes • Using coal containing less sulfur, washing the coal, and using devices called scrubbers to chemically remove the SO 2 from the gases leaving the smokestack • Power plants can also switch fuels; for example burning natural gas creates much less SO 2 than burning coal • Catalytic converters reduce NOx emissions from cars

Controlling acid deposition n Use alternative energy sources • There are other sources of electricity besides fossil fuels. They include: hydropower, wind energy, geothermal energy, and solar energy • Also alternative energies available to power automobiles, including natural gas powered vehicles, battery-powered cars, and fuel cells n Take action as individuals • Individuals can contribute directly by conserving energy, since energy production causes the largest portion of the acid deposition problem. For example, you can: n n Turn off lights, computers, and other appliances when you're not using them Carpool, use public transportation, or better yet, walk or bicycle whenever possible
- Slides: 28