Acid Equilibrium and p H Srensen 1 AcidBase

Acid Equilibrium and p. H Sørensen 1

Acid/Base Definitions q Arrhenius Model q Acids produce hydrogen ions in aqueous solutions q Bases produce hydroxide ions in aqueous solutions q Bronsted-Lowry Model q Acids are proton donors q Bases are proton acceptors q Lewis Acid Model q Acids are electron pair acceptors q Bases are electron pair donors 2

Acid Dissociation Constant HA Acid H+ + AProton Conjugate base Alternately, H+ may be written in its hydrated form, H 3 O+ (hydronium ion). HA(aq) + H 2 O(l) H 3 O+(aq) + A-(aq) acid base conj. Acid conj. base 3

Practice: write the simple dissociation reaction for the following acids • HCl • HC 2 H 3 O 2 • NH 4+ • C 6 H 5 NH 3+ • [Al(H 2 O)6]3+ 4

Dissociation of Strong Acids Strong acids are assumed to dissociate completely in solution. Large Ka or small Ka? Reactant favored or product favored? 5
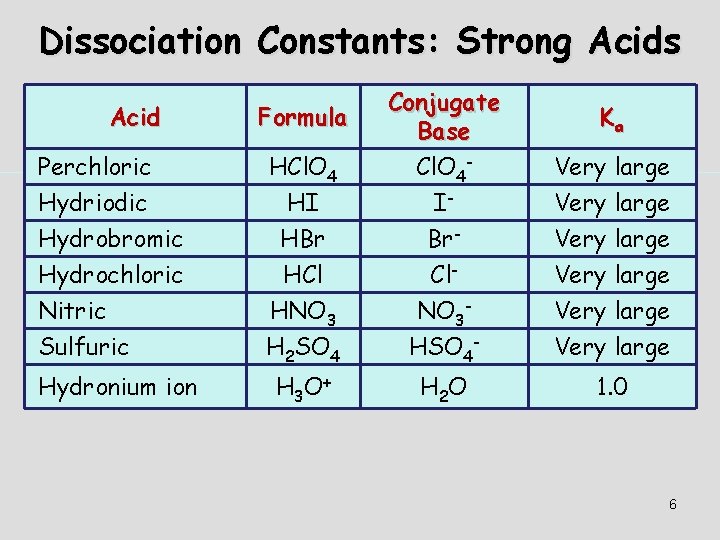
Dissociation Constants: Strong Acids Acid Formula Perchloric Hydriodic HCl. O 4 HI Conjugate Base Cl. O 4 - I- Hydrobromic Hydrochloric Nitric Sulfuric HBr HCl HNO 3 H 2 SO 4 Br- Cl- NO 3 - HSO 4 - Very large Hydronium ion H 3 O+ H 2 O 1. 0 Ka Very large 6
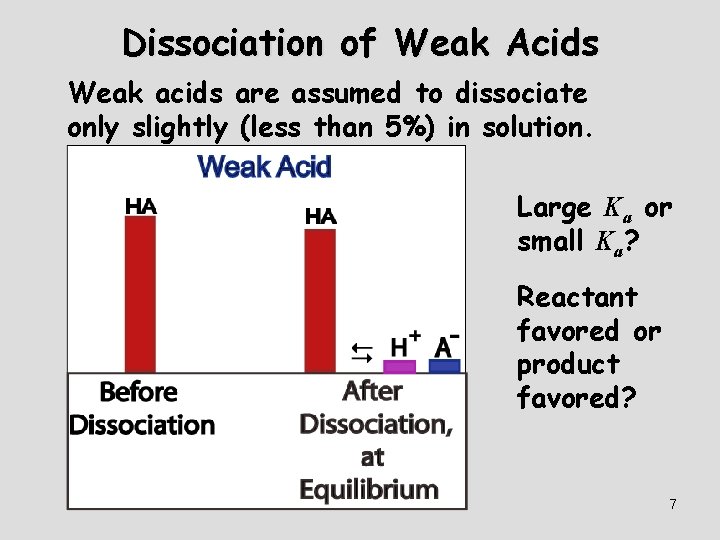
Dissociation of Weak Acids Weak acids are assumed to dissociate only slightly (less than 5%) in solution. Large Ka or small Ka? Reactant favored or product favored? 7
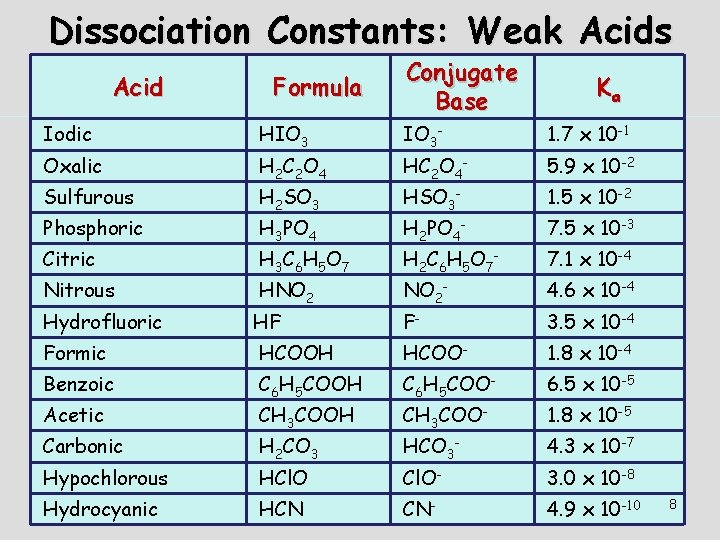
Dissociation Constants: Weak Acids Acid Iodic Oxalic Sulfurous Phosphoric Citric Nitrous Hydrofluoric Formula Conjugate Base Ka HIO 3 IO 3 - 1. 7 x 10 -1 H 2 SO 3 HSO 3 - 1. 5 x 10 -2 H 2 C 6 H 5 O 7 - 7. 1 x 10 -4 H 2 C 2 O 4 H 3 PO 4 H 3 C 6 H 5 O 7 HNO 2 HF HC 2 O 4 - 5. 9 x 10 -2 H 2 PO 4 - 7. 5 x 10 -3 NO 2 - 4. 6 x 10 -4 F- 3. 5 x 10 -4 Formic HCOOH HCOO- 1. 8 x 10 -4 Benzoic C 6 H 5 COOH C 6 H 5 COO- 6. 5 x 10 -5 H 2 CO 3 HCO 3 - 4. 3 x 10 -7 Acetic Carbonic Hypochlorous Hydrocyanic CH 3 COOH HCl. O HCN CH 3 COO- 1. 8 x 10 -5 Cl. O- 3. 0 x 10 -8 CN- 4. 9 x 10 -10 8
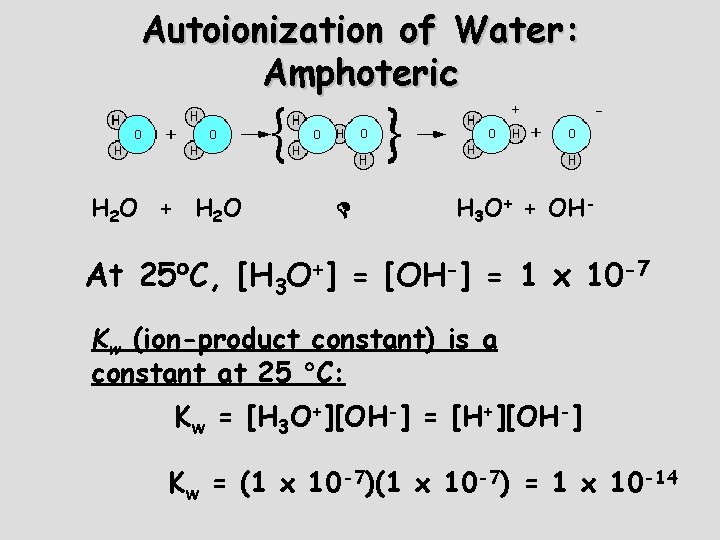
Autoionization of Water: Amphoteric H 2 O + H 2 O H 3 O+ + OH- At 25 C, [H 3 O+] = [OH-] = 1 x 10 -7 Kw (ion-product constant) is a constant at 25 C: Kw = [H 3 O+][OH-] = [H+][OH-] Kw = (1 x 10 -7) = 1 x 10 -14
![Calculating p. H, p. OH p. H = -log 10[H+] p. OH = -log Calculating p. H, p. OH p. H = -log 10[H+] p. OH = -log](http://slidetodoc.com/presentation_image_h/1a5c8ac5c3661994ee00e8b4dd10240c/image-10.jpg)
Calculating p. H, p. OH p. H = -log 10[H+] p. OH = -log 10[OH-] Relationship between p. H and p. OH p. H + p. OH = 14 Finding [H 3 O+], [OH-] from p. H, p. OH [H 3 O+] = 10 -p. H [OH-] = 10 -p. OH
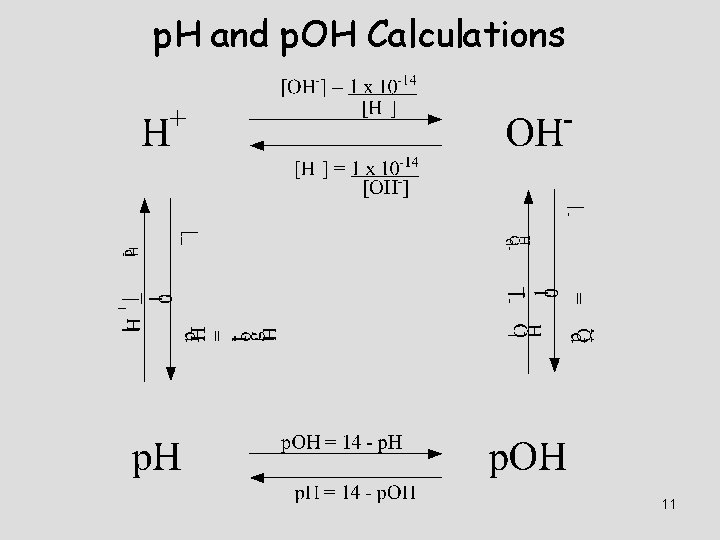
p. H and p. OH Calculations 11

The p. H Scale Graphic: Wikimedia Commons user Slower

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #1: Write the dissociation equation HC 2 H 3 O 2 C 2 H 3 O 2 - + H+

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #2: ICE it! I C E HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ 0. 50 - x 0 0 +x +x x x

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #3: Set up the law of mass action E HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ 0. 50 - x x x

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #4: Solve for x, which is also [H+] E HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ 0. 50 - x x x [H+] = 3. 0 x 10 -3 M

A Weak Acid Equilibrium Problem What is the p. H of a 0. 50 M solution of acetic acid, HC 2 H 3 O 2, Ka = 1. 8 x 10 -5 ? Step #5: Convert [H+] to p. H E HC 2 H 3 O 2 C 2 H 3 O 2 - + H+ 0. 50 - x x x
Your turn • P. 691 #49, 61, 63, 65 • Refer to table 14. 2 for Ka’s of common monoprotic acids on page 644
Sodium hydroxide Bases Preferred IUPAC name Sodium hydroxide Systematic name Sodium oxidanide Other names Caustic soda Lye Graphics Source: Wikipedia

Acid/Base Definitions q Arrhenius Model q q Acids produce hydrogen ions in aqueous solutions Bases produce hydroxide ions in aqueous solutions q Bronsted-Lowry Model q Acids are proton donors q Bases are proton acceptors q Lewis Acid Model q Acids are electron pair acceptors q Bases are electron pair donors

Dissociation of Strong Bases MOH(s) M+(aq) + OH-(aq) Ø Strong bases are metallic hydroxides Ø Group I hydroxides (Na. OH, KOH) are very soluble Ø Group II hydroxides (Ca, Ba, Mg, Sr) are less soluble Ø p. H of strong bases is calculated directly from the concentration of the base in solution

Reaction of Weak Bases with Water The base reacts with water, producing its conjugate acid and hydroxide ion: CH 3 NH 2 + H 2 O CH 3 NH 3+ + OH- Kb = 4. 38 x 10 -4
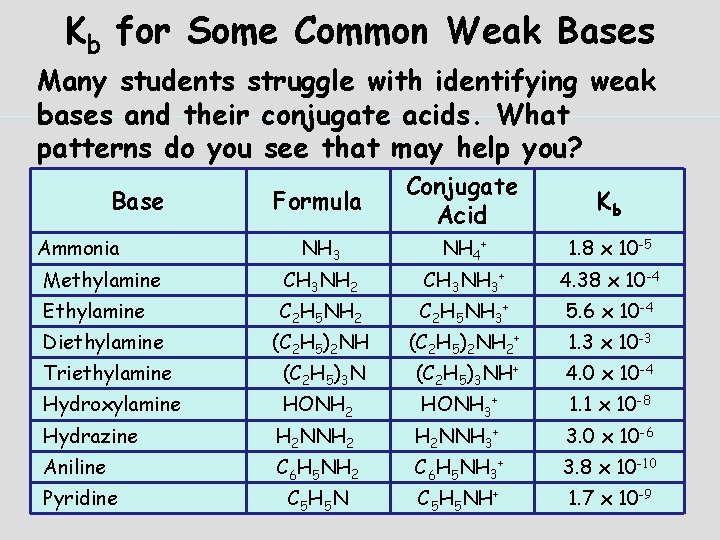
Kb for Some Common Weak Bases Many students struggle with identifying weak bases and their conjugate acids. What patterns do you see that may help you? Formula Conjugate Acid Kb NH 3 NH 4+ 1. 8 x 10 -5 Methylamine CH 3 NH 2 CH 3 NH 3+ 4. 38 x 10 -4 Ethylamine C 2 H 5 NH 2 C 2 H 5 NH 3+ 5. 6 x 10 -4 Diethylamine (C 2 H 5)2 NH 2+ 1. 3 x 10 -3 Triethylamine (C 2 H 5)3 NH+ 4. 0 x 10 -4 Hydroxylamine HONH 2 HONH 3+ 1. 1 x 10 -8 Base Ammonia Hydrazine H 2 NNH 2 H 2 NNH 3+ 3. 0 x 10 -6 Aniline C 6 H 5 NH 2 C 6 H 5 NH 3+ 3. 8 x 10 -10 Pyridine C 5 H 5 NH+ 1. 7 x 10 -9

Reaction of Weak Bases with Water The generic reaction for a base reacting with water, producing its conjugate acid and hydroxide ion: B + H 2 O BH+ + OH- (Yes, all weak bases do this – DO NOT endeavor to make this complicated!)

A Weak Base Equilibrium Problem What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #1: Write the equation for the reaction NH 3 + H 2 O NH 4+ + OH-
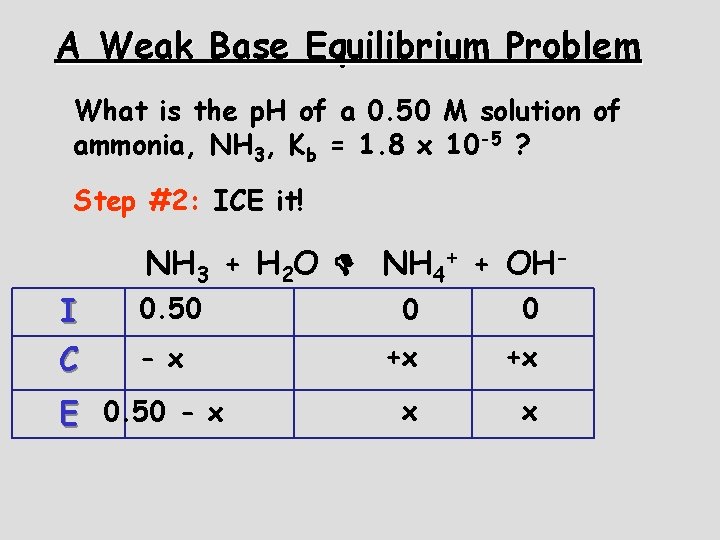
A Weak Base Equilibrium Problem What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #2: ICE it! I C NH 3 + H 2 O NH 4+ + OH- 0. 50 - x E 0. 50 - x 0 0 +x +x x x

A Weak Base Equilibrium Problem What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #3: Set up the law of mass action E NH 3 + H 2 O NH 4+ + OH 0. 50 - x x x

A Weak Base Equilibrium Problem What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #4: Solve for x, which is also [OH-] E NH 3 + H 2 O NH 4+ + OH 0. 50 - x x x [OH-] = 3. 0 x 10 -3 M

A Weak Base Equilibrium Problem What is the p. H of a 0. 50 M solution of ammonia, NH 3, Kb = 1. 8 x 10 -5 ? Step #5: Convert [OH-] to p. H NH 3 + H 2 O NH 4+ + OH- E 0. 50 - x x x
Reference Table
- Slides: 31