Acid Deposition Lake Barkevatn in Norway used to

Acid Deposition • Lake Barkevatn in Norway used to have healthy stocks of trout and perch. • As a result of acid rain, the trout stock died out in the mid-1970 s and the perch stock at the beginning of the 1980 s.


• Unpolluted rain is itself slightly acidic with a p. H level of about 5. 6. • Results from the dissolution of CO 2 (an acidic oxide) in water droplets CO 2 (g) + H 2 O(l) H 2 CO 3 (aq) H+ (aq) + HCO 3 -(aq) CO 2 (g) + H 2 O(l) H+ (aq) + HCO 3 - (aq)

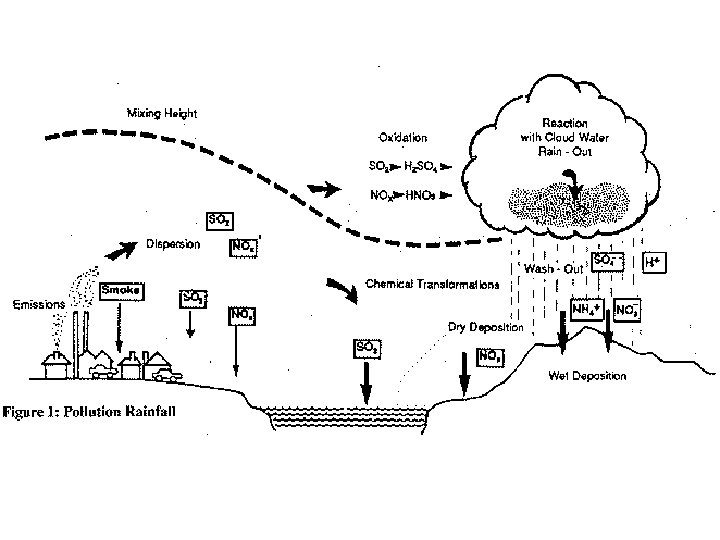
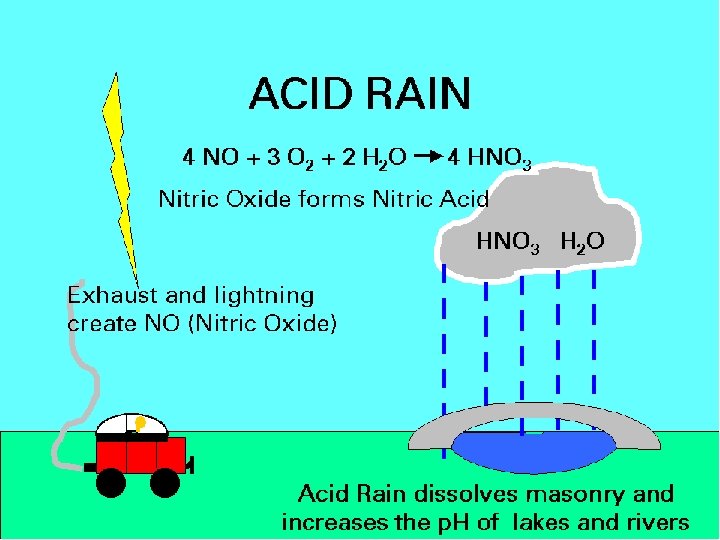
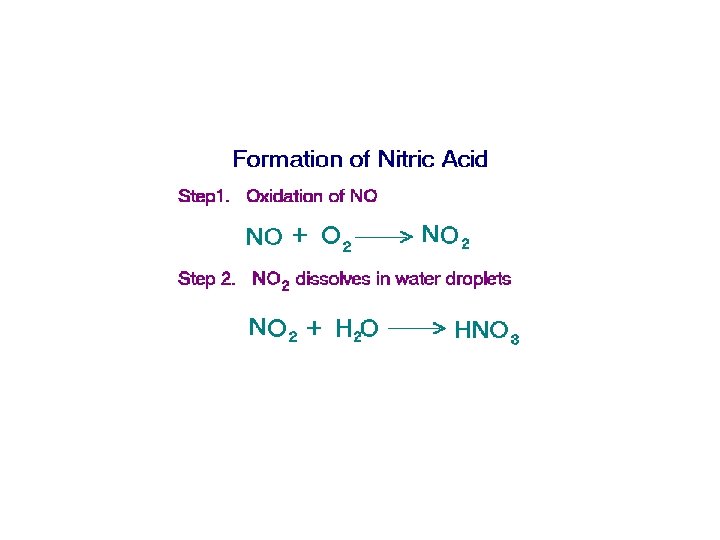
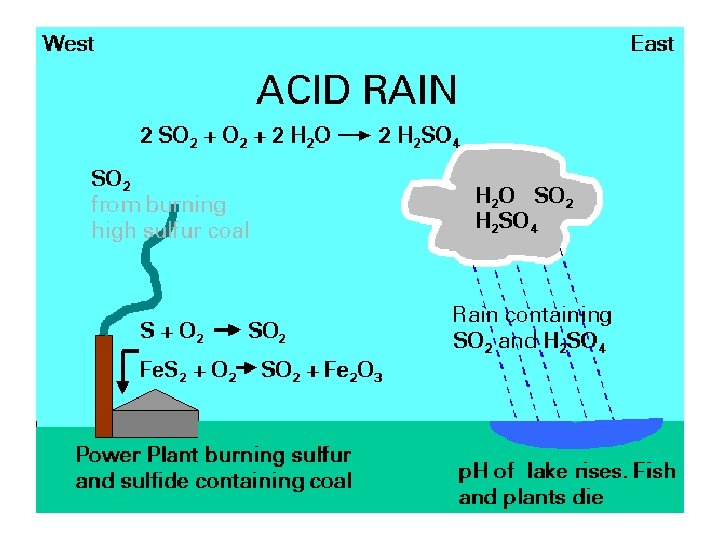
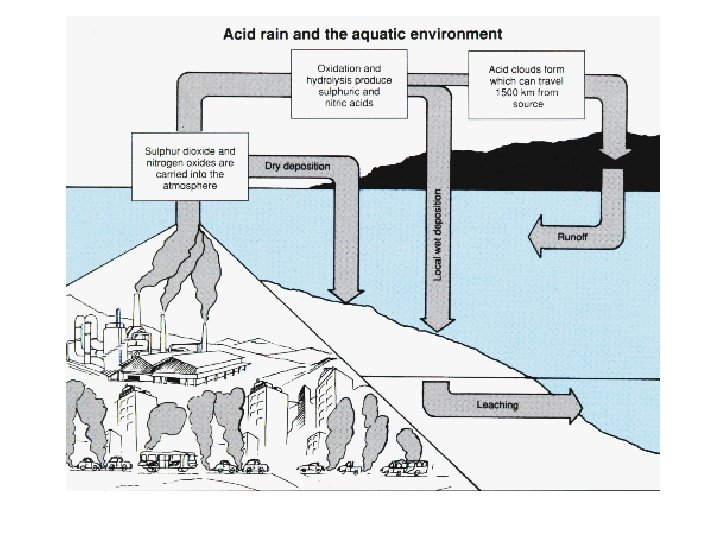
Acid Deposition • The deposition of acids from the atmosphere in precipitation that falls as rain, sleet, snow, and particulates. • Fog & dew can also be acidic. • Mainly results from nitrogen oxides and sulfur oxides.

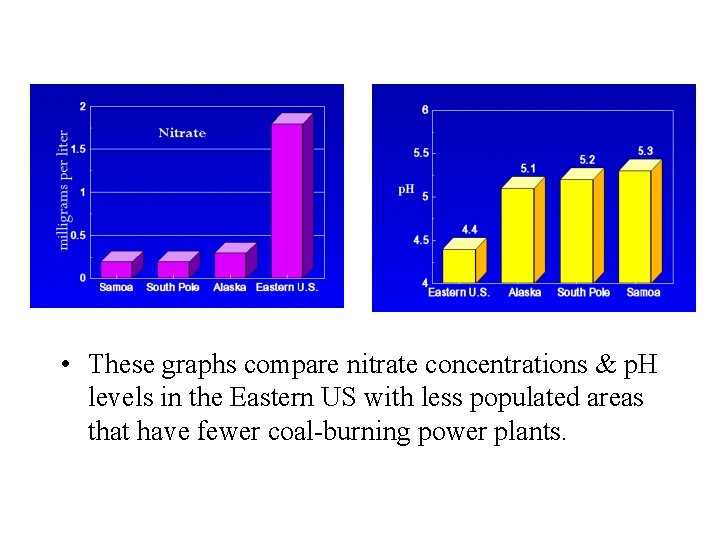
• These graphs compare nitrate concentrations & p. H levels in the Eastern US with less populated areas that have fewer coal-burning power plants.

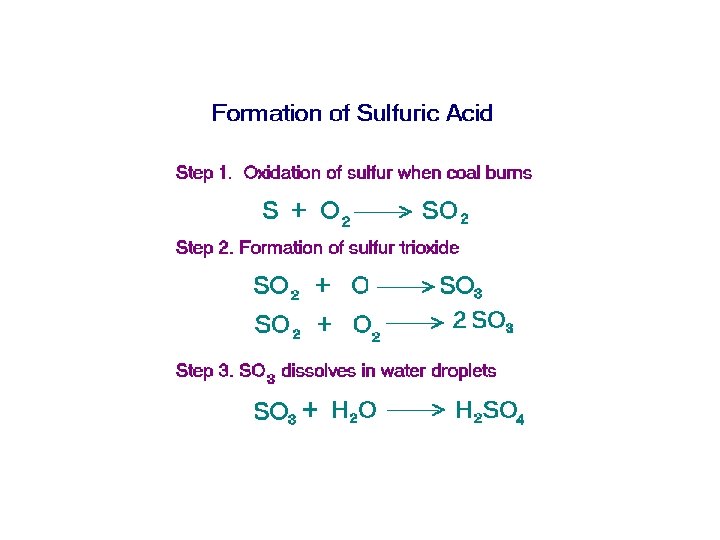
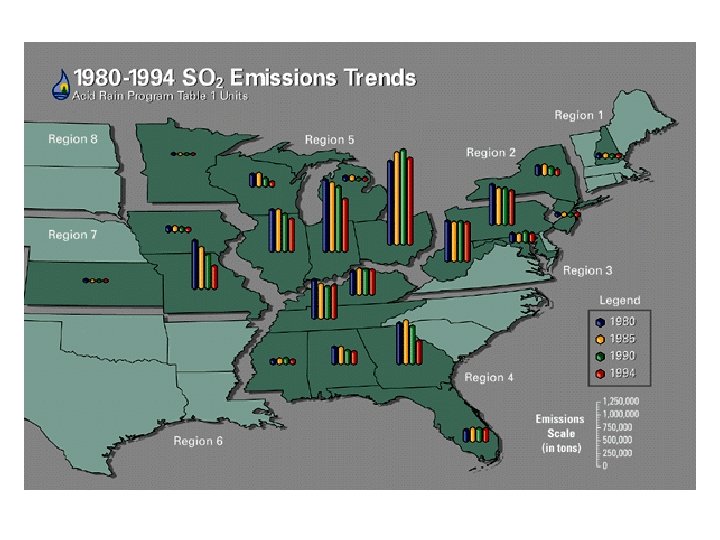
• Coal contains sulfur in the form of the mineral pyrite Fe. S 2 4 Fe. S 2 + 11 O 2 2 Fe 2 O 3 + 8 SO 2 • A 1000 megawatt coal-fired electricity generating plant burns ~700 tons of coal per hr • @4% S • 56 tons SO 2 generated per hour • 490, 560 tons SO 2 per year

Other Sources of Sulfur • Volcanoes • Microbes & plants • Smelters

Smelters • Nonferrous smelting facilities are large point sources (individual sites emitting large amounts of a pollutant) of SO 2 • Many useful metals such as copper & nickel occur in nature as sulfide ores. • In these smelters, ores are reduced to free metals.

The Smelting Process • First step in the process is to remove the sulfur from the sulfide (roasting).

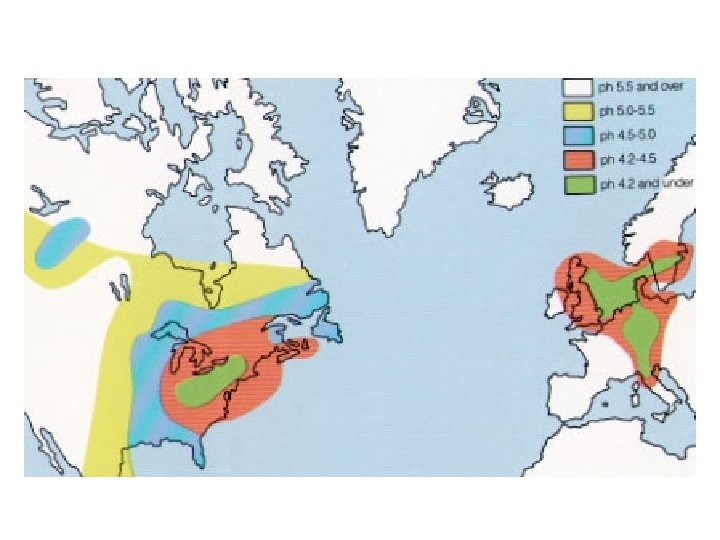
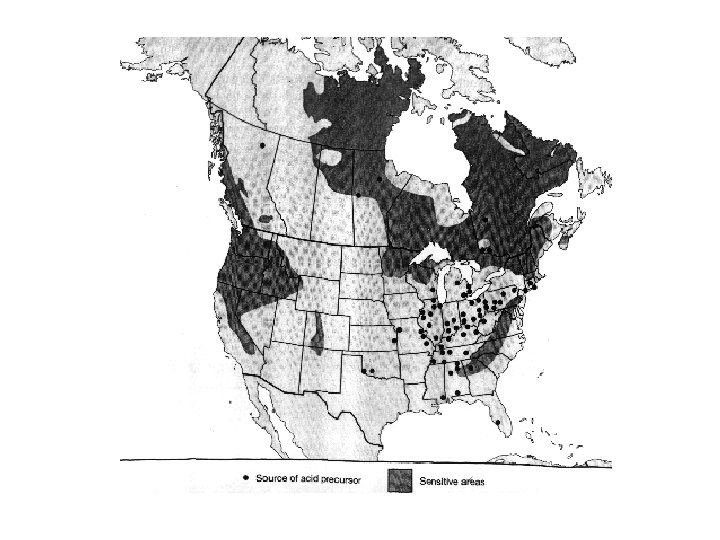
Acid Rain Is Not a New Phenomenon • First recognized by Robert Angus Smith in the mid-1800 s in Great Britain • In the 1950 s Swedish scientists began to observe p. H values of precipitation lower than 5. 2. • In the 1960 s low p. H values were discovered in the north Eastern U. S.
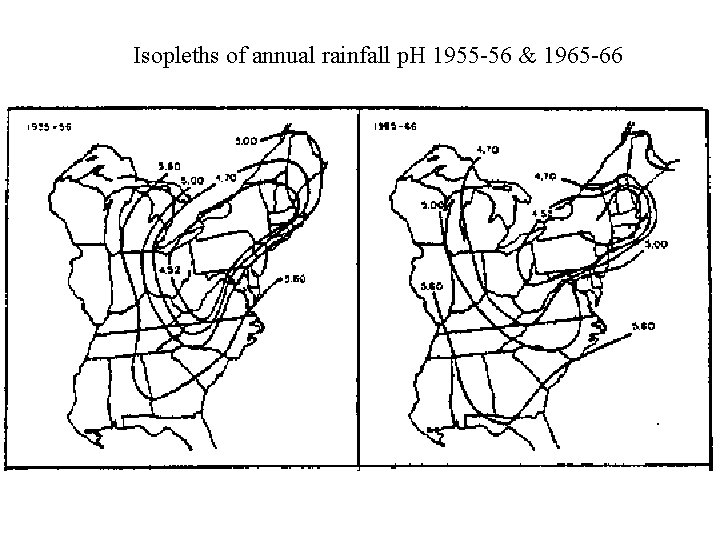
Isopleths of annual rainfall p. H 1955 -56 & 1965 -66
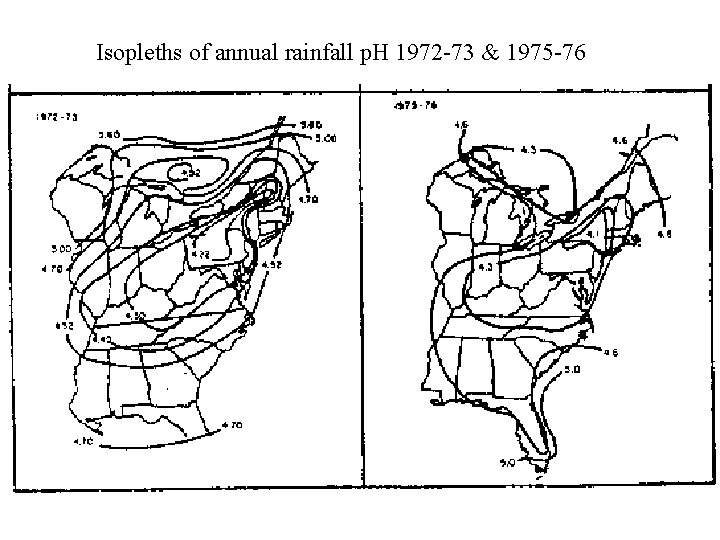
Isopleths of annual rainfall p. H 1972 -73 & 1975 -76
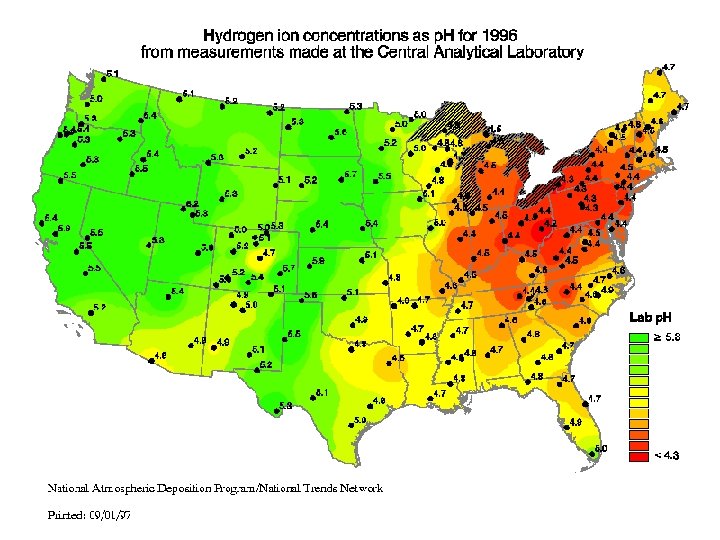
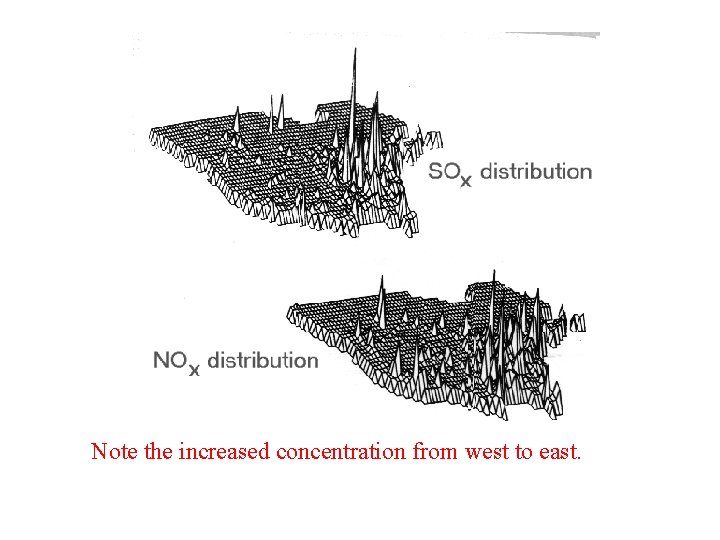
Note the increased concentration from west to east.
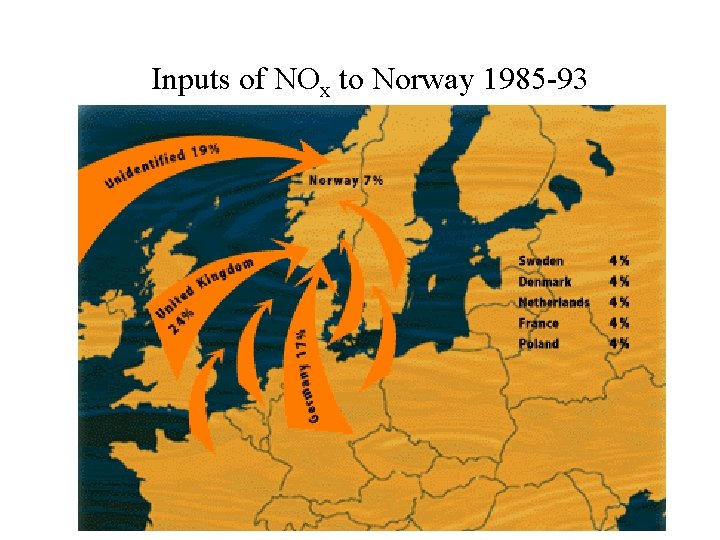
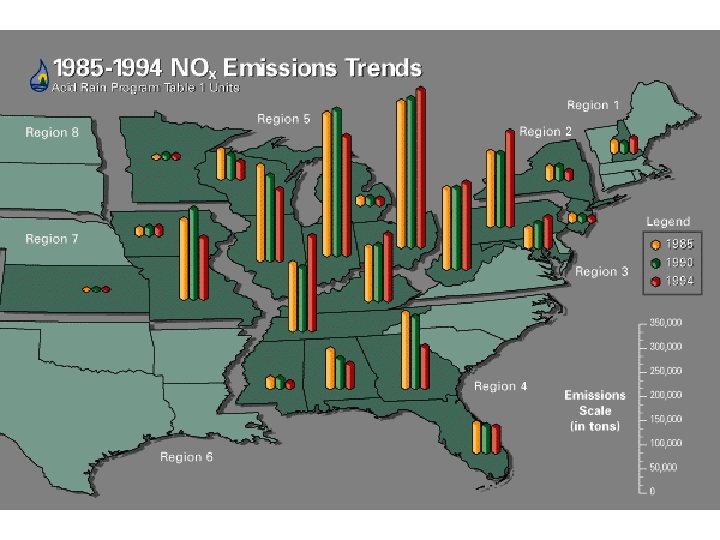
Inputs of NOx to Norway 1985 -93
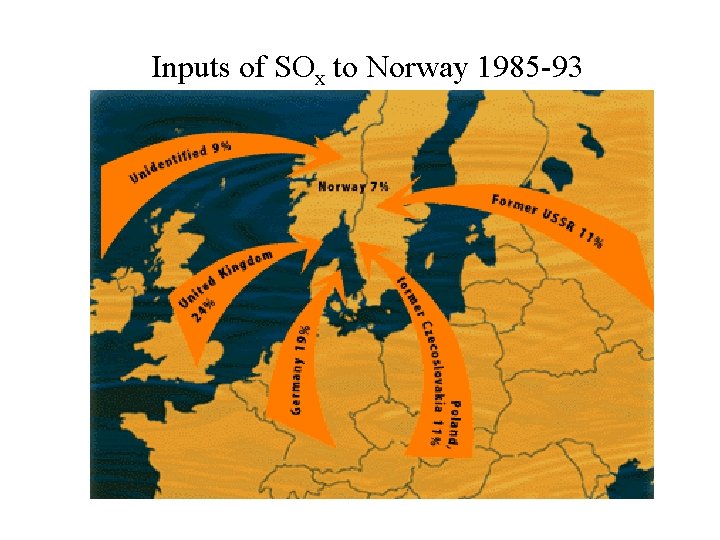
Inputs of SOx to Norway 1985 -93

Effects of Acid Rain


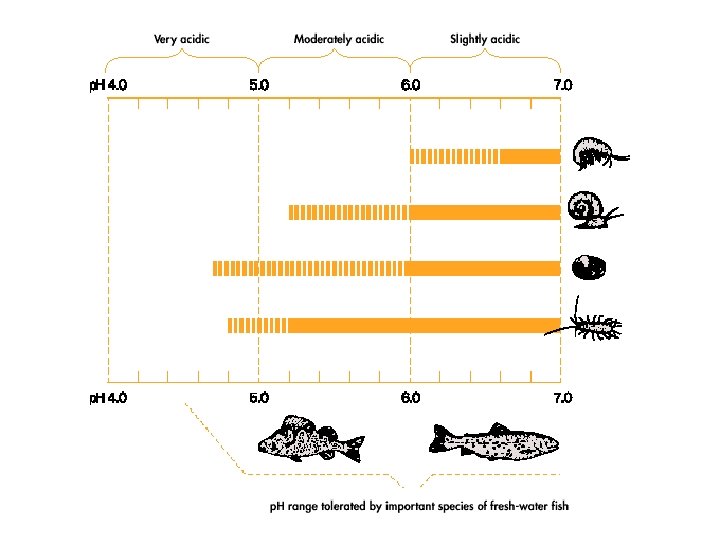
Effects on Aquatic Life • In late 1960 s – fish populations were found to be declining in lakes in the Adirondack Mountains. – Fish populations were declining in lakes in Sweden & Norway – Salamander populations were declining rapidly in the northeastern U. S.

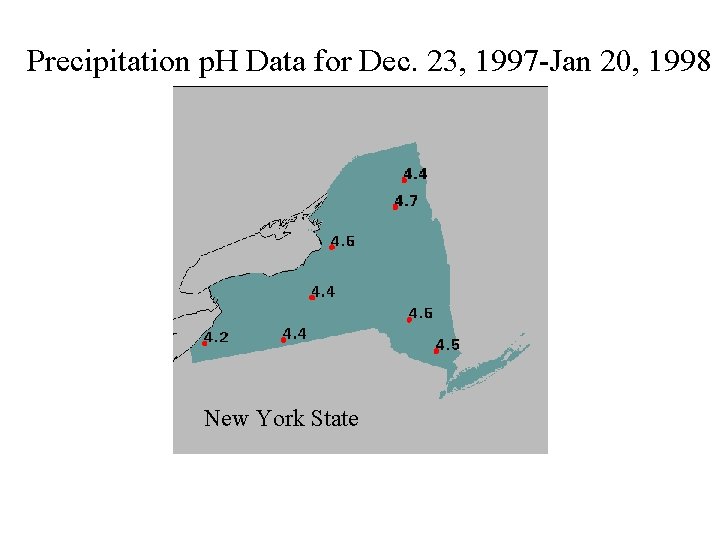
Precipitation p. H Data for Dec. 23, 1997 -Jan 20, 1998 New York State

Effects on Aquatic Life • By 1970 s in Adirondack Mountains – more than 50% of lakes >600 meters had p. H<5 – 90% of lakes contained no fish • By 1990 s in Sweden – 14, 000 lakes unable to support lake organisms sensitive to acidic conditions – 2, 200 lakes nearly lifeless

Neutralization & Acid-sensitivity • Different rivers, lakes & soils are not equally affected by or sensitive to acid deposition. • Natural water bodies may contain substances that buffer the p. H. • The buffering capacity is mainly determined by the amount of bicarbonate (HCO 3 -) and carbonate (CO 32 -) in the water.

Natural Buffers • If soils and the underlying rock are rich in calcium carbonate (limestone), then the deposited acids are partially neutralized. Ca. CO 3 + H 2 SO 4 Ca 2+ +SO 42 - + HCO 3 - + H+ • This leads to a delay in the acidification of these aquatic systems.

Natural Buffers • Natural waters underlain by rocks containing little Ca. CO 3 are more susceptible to acidification. – Adirondacks – Fennoscandinavian region • Rocks rich in silicate minerals are abundant in these areas.

Human Intervention • Sweden & Norway are carrying out a program to neutralize the acid in lakes by adding lime to the lakes. • Stimulates the natural process involving the reaction of acid with carbonate. • An expensive temporary cure. • Analogous to taking antacid tablets to neutralize stomach acid!

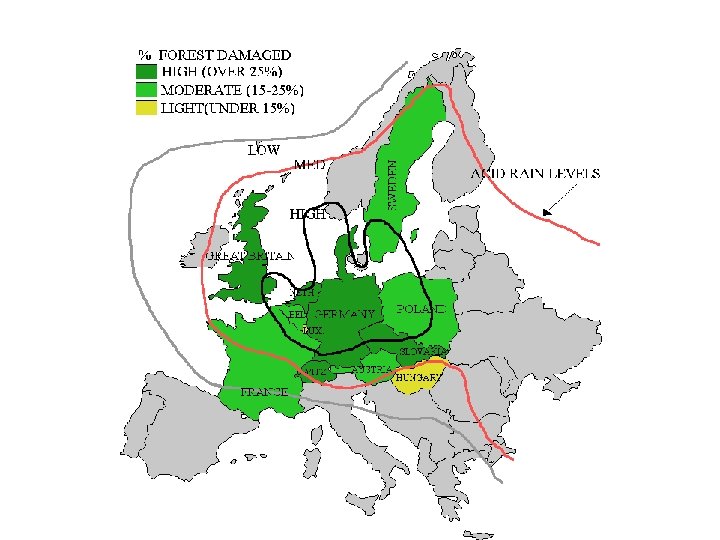
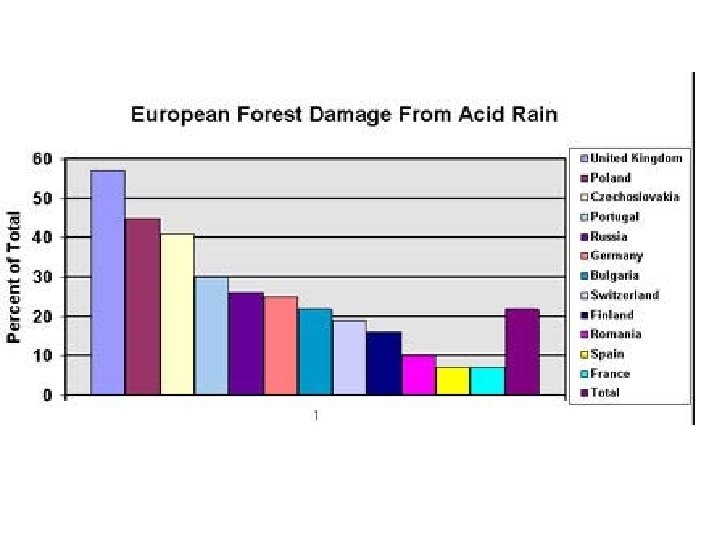
Effects on Forest Ecosystems • Forest damage has been extensive in Europe. • In Germany, the 75% of the fir trees were found to be affected in 1983.

Declining Forests • The crown density of the spruce at right is normal. • The crown density of tree at left is lower

Declining Forests • Leaves and needles can be damaged directly • Trees damaged indirectly by loss of nutrients from the soil via leaching. – In very acidic conditions, Al becomes soluble & is released from soil – at high concentration it is toxic & damages roots

Destruction of Our History • Both SO 2 and acid rain react with buildings, statues & monuments made of limestone & marble. • Calcium carbonate is the chemical composition of these materials.

Reactions of Ca. CO 3 • Calcium carbonate dissolves in acid rain Ca. CO 3(s) + H+(aq) Ca 2+ (aq) + HCO 3 - (aq) • Calcium carbonate reacts with SO 2 to form gypsum (Ca. SO 4. 2 H 2 O) which is also water soluble.

German sandstone statue from 1702 photographed in 1908 (left) & in 1969 (right).

Gargoyles
- Slides: 48