ACID BASE PHYSIOLOGY PROFESSOR ASHRAF HUSAIN DEPT OF

ACID - BASE PHYSIOLOGY PROFESSOR. ASHRAF HUSAIN DEPT. OF PHYSIOLOGY COLLEGE OF MEDICINE KING SAUD UNIVERSITY RIYADH. Modified by The Legendary Genius

Acid Base Physiology • Definition: p. H is defined as potential of H+ Ion concentration in body fluid. The amount of H+ ion concentration is so low in the body hence it is expressed as –ve logarithm to base of the H+ ion concentration in m. Eq/lit. p. H = log 1/ [H+ ] = - log [H+ ]

The Acid-Base Balance • Balance of H conc. In ECF. • To Achieve Homeostasis. • Balance Between : The H Intake or Production The H Removal

NORMAL VALUE • Arterial blood = 7. 35 – 7. 45 Can be explained as follows; Normal value of H+ ion conc. is about 40 n. Eq/lit. 40 n. Eq/lit = 0. 00000004 Eq/lit. Therefore p. H = - log [0. 00000004] = 7. 4

Acid – Base Imbalance • Acidosis = Decrease in arterial PH ( <7. 35 ) Due to excess H+ • Alkalosis = Elevation in Arterial PH ( >7. 45) Due to excess base.

p. H and H+ ion concentration p. H • • 6. 0 7. 0 8. 0 9. 0 • • H+ ion in nmol/lit 1000 10 1. 0 Note : one point drop in p. H results in a ten fold decrease in H+ ion conc.

Scale of p. H measurement • The p. H scale is between 0 – 14. Zero onwards below 7 is acidic. After 7 upto 14 the solution is alkaline. At 7 (neutral e. g. water) where the amount of o H+ and Hydroxyl ion are equal at 23

Examples of p. H zero, one & two. p. H 0 = 1 lit. of HCl contains 1 gm of H+ = 10 o p. H 1 = 10 lit. of N/10 HCl contains 1 gm of H+ Therefore 1 lit. will contain 1/10 gm of H+ =10 -1 p. H 2 = 100 lit. of N/100 HCl contains 1 gm of H+ Therefore 1 lit. will contain 1/100 gm of H+ =10 -2 And so on. . It will be noted that a solution of p. H 2 does not have twice the H+ ion conc. at a p. H of 4 rather it has 100 times H+ ion conc.

• Since p. H is inversely related to H+ ion conc. so a low p. H corresponds to high H+ ion conc. (Acidic) and a high p. H corresponds to low H+ ion conc. (Alkaline) Acidic 0 Neutral 7 Alkaline 14
![The Negative logarithmic relationship between [H+] and p. H The Negative logarithmic relationship between [H+] and p. H](http://slidetodoc.com/presentation_image/57806fe2fdeecf4970cf7a5bf6997d4f/image-10.jpg)
The Negative logarithmic relationship between [H+] and p. H

About H+ H+ are derived from hydrogen atoms. Hydrogen atom has proton a positive electrical charge with a negative charge electron which is revolving around it. The hydrogen ion is formed when negative ion (electron) is lost and it contains only proton. The H+ is the smallest ionic particle, highly reactive with a very short life and unable to survive on its own.

A very tight control is needed for normal metabolic functions. eg. Enzymatic activity, blood clotting and neuromuscular activity. Hydrogen ions are the toxic end product of metabolism and they adversely affect all physical and biochemical cellular process in our body.

Acid • Molecules containing H atoms that can release (donate) H ions in solutions. • Example , HCL. • Strong acids : - Completely dissociate : (HCL , H 2 SO 4 ) • Weak acid : - Partially dissociate : ( H 2 CO 3)

Base • An Ion that accept a H ion. • An example of a base is the Bicarbonate ( HCO 3 ).

Buffers • Substances that Neutralize acids or bases. • Chemical Reactions which Reduce the effect of adding acid or base to a solution PH.

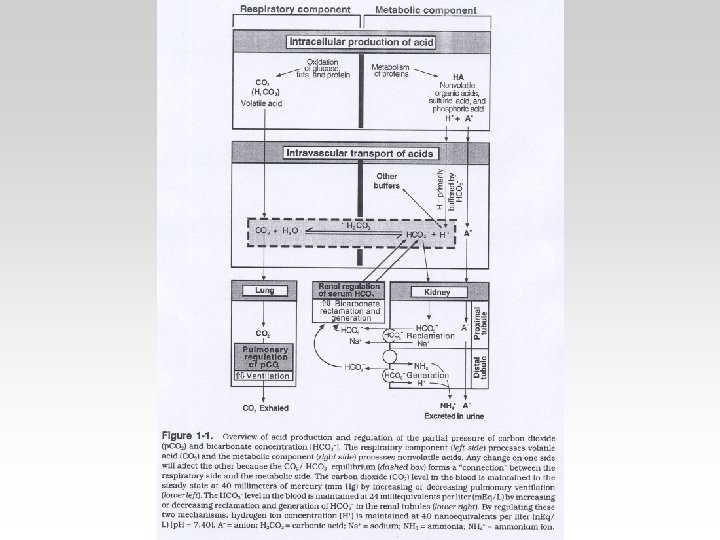
How the body defends against fluctuations in PH • Three Systems in the body : 1) Buffers in blood. 2) Respiration through the lungs. 3) Excretion by the kidney.

Blood Buffer • These buffer systems serve as a first line of defense against changes in the acidbase balance : - HCO 3(Regulated by Renal and Respiratory). - Protein - Phosphate - Hemoglobin

Protein • Acidic and Basic Amino acid in plasma and cell protein act as buffers. • HB is an important buffer , can’t be regulated physiological.

Phosphate & Intracellular Buffers • Both Intra and Extra cellular phosphate act as a buffer. But its role is minor compared to HB or HCO 3. • Intracellular buffers are needed because H doesn’t cross Plasma Membrane. • Intracellular PH is more acidic. (7. 2)
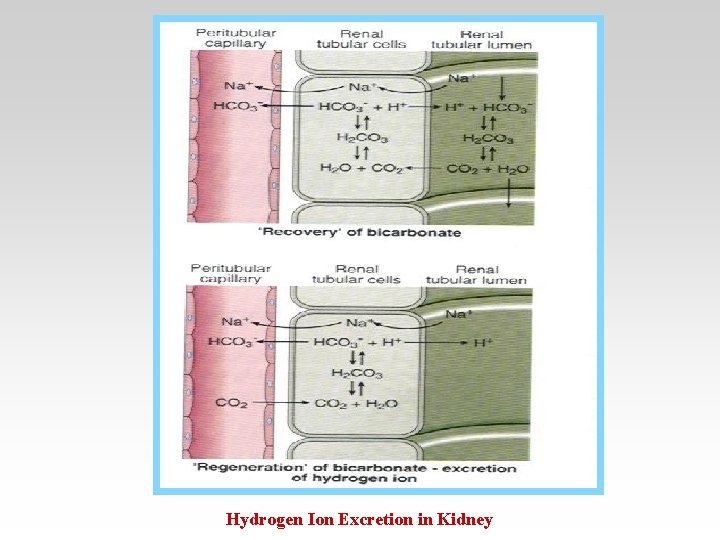
Hydrogen Ion Excretion in Kidney
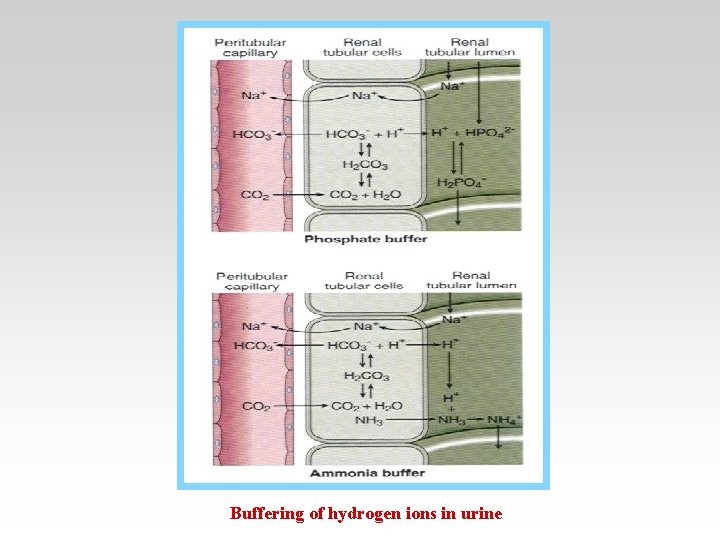
Buffering of hydrogen ions in urine
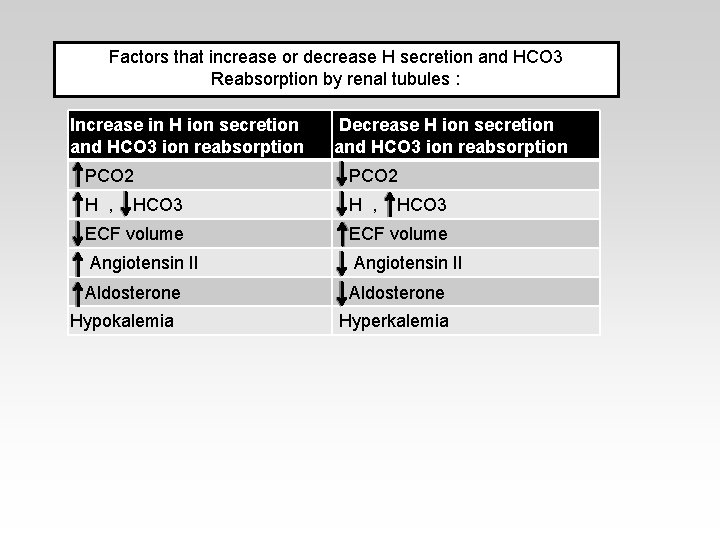
Factors that increase or decrease H secretion and HCO 3 Reabsorption by renal tubules : Increase in H ion secretion and HCO 3 ion reabsorption Decrease H ion secretion and HCO 3 ion reabsorption PCO 2 H , HCO 3 ECF volume Angiotensin II Aldosterone Hypokalemia Hyperkalemia

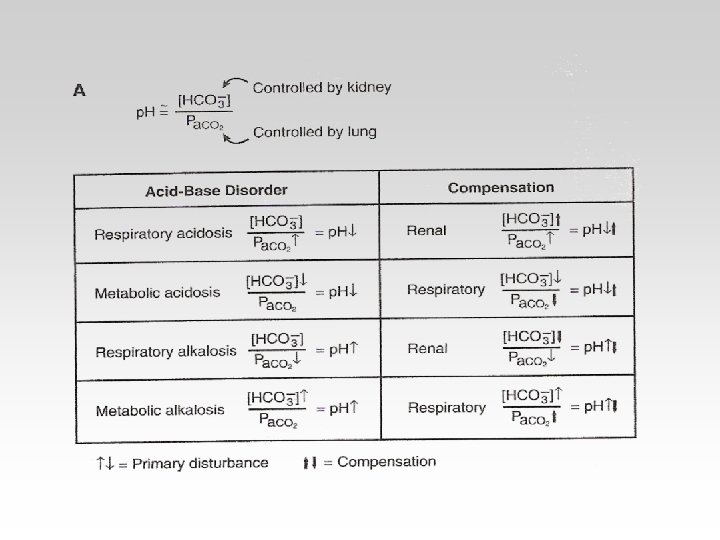
Respiratoy Regulation of PH • Maintaining Normal PH by maintaining constant PCO 2. • Normal gas Exchange and ventilation. • Controlled by chemoreceptors. • PCO 2 PH

Renal Regulation • Tubular Mechanisms of H+ • Tubular Reabsorption of HCO 3.

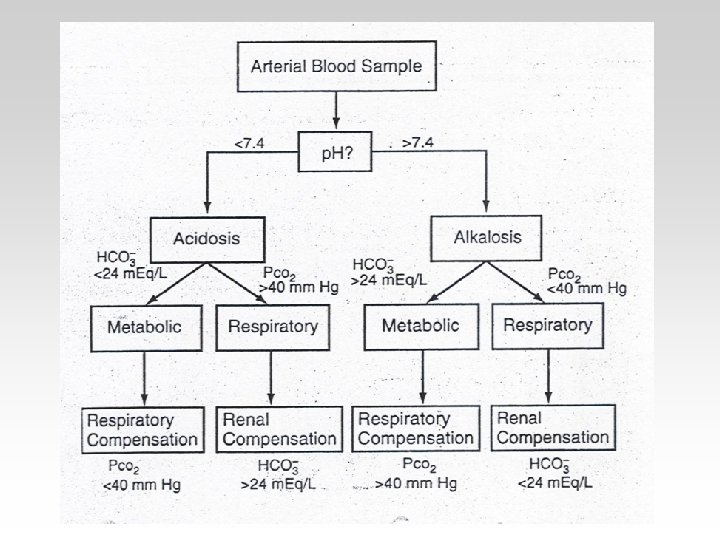
Three ways to measure acidbase balance in the body : • Blood PH • Blood PCO 2 • Blood HCO 3

Compensation -If underlying problem is metabolic : Hyperventilation and Hypoventilation mechanisms will help through Respiratory Compensation. -If the problem is Respiratory , Renal mechanisms , then Renal mechanisms will help through Metabolic Compensation.

Acidosis -Principal effect of acidosis is Depression of the CNS through the decrease in synaptic transmission. - Generelized Weakness. - Deranged CNS is the greatest thread. - severe acidosis causes : 1 - Disorientation 2 - Coma 3 - Death

Alkalosis -Causes over excitability of the central and peripheral nervous systems. -Numbness - Lightheadedness It can cause : - nervousness. - muscle spasms or tetany. - convulsions - looss of consciousness - death.
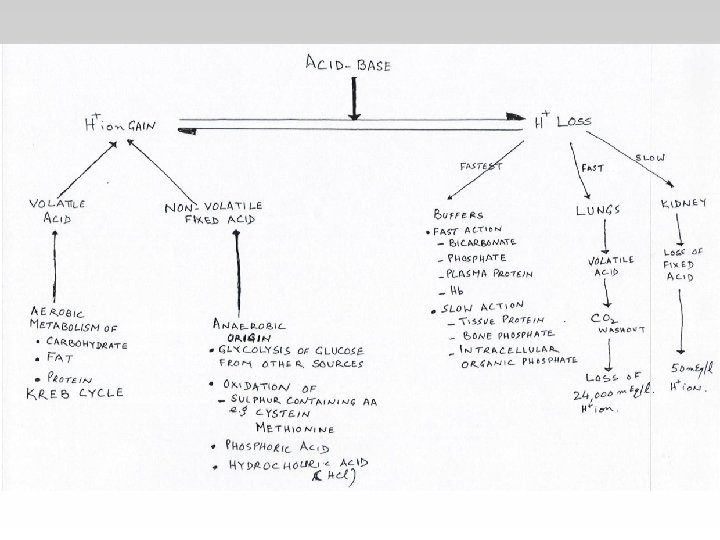
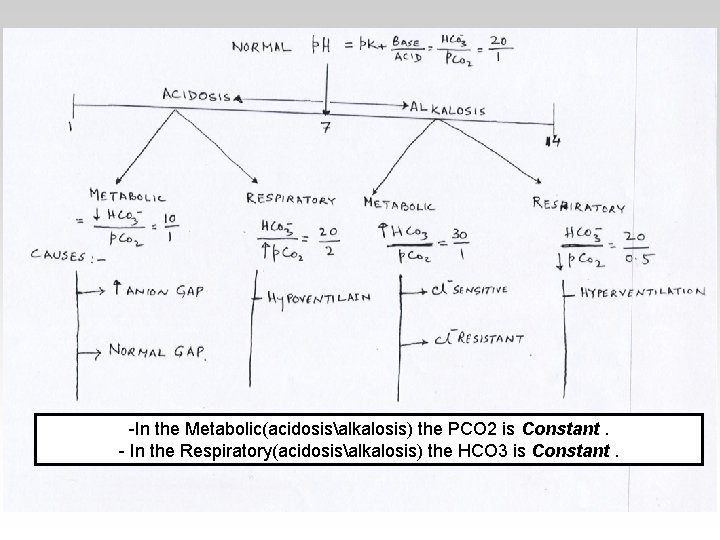
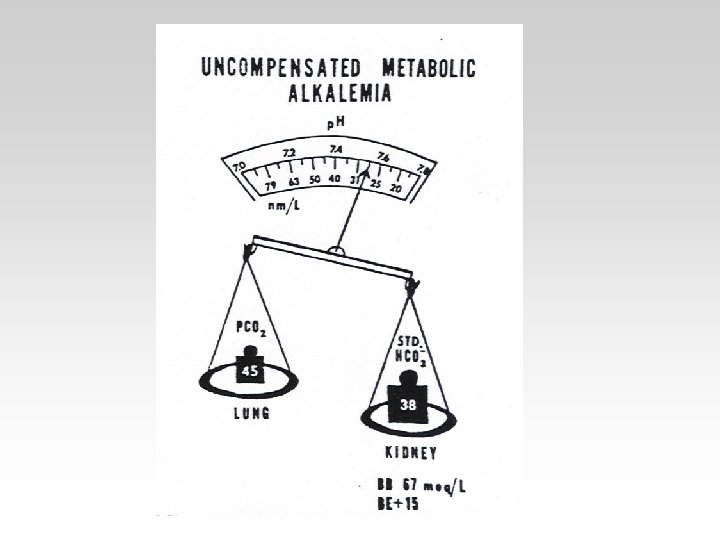
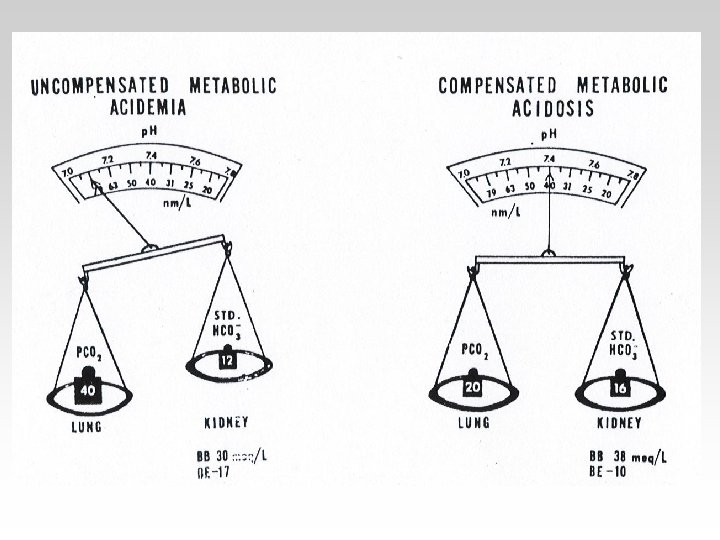
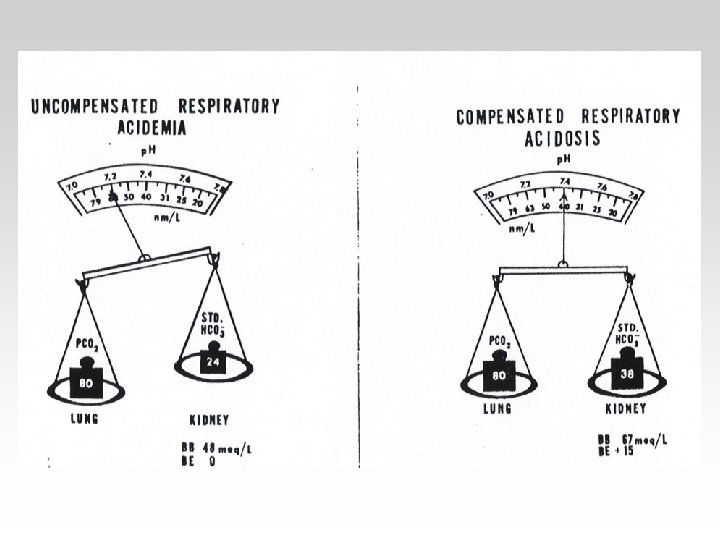
-In the Metabolic(acidosisalkalosis) the PCO 2 is Constant. - In the Respiratory(acidosisalkalosis) the HCO 3 is Constant.
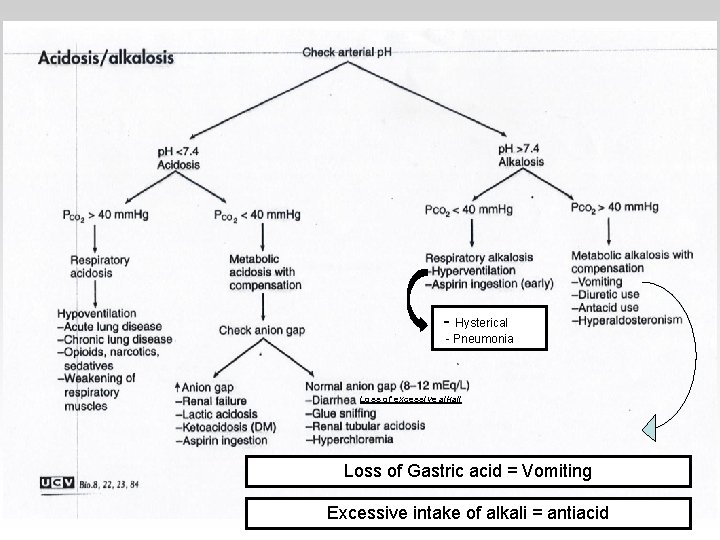
- Hysterical - Pneumonia Loss of excessive alkali Loss of Gastric acid = Vomiting Excessive intake of alkali = antiacid
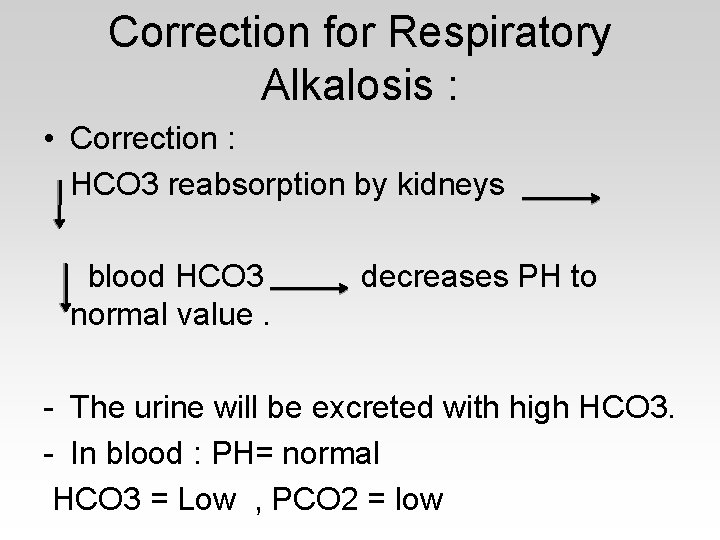
Correction for Respiratory Alkalosis : • Correction : HCO 3 reabsorption by kidneys blood HCO 3 normal value. decreases PH to - The urine will be excreted with high HCO 3. - In blood : PH= normal HCO 3 = Low , PCO 2 = low
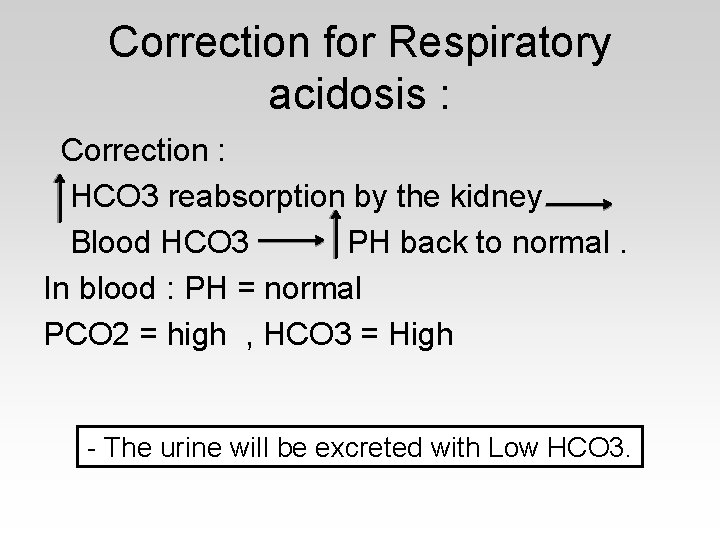
Correction for Respiratory acidosis : Correction : HCO 3 reabsorption by the kidney Blood HCO 3 PH back to normal. In blood : PH = normal PCO 2 = high , HCO 3 = High - The urine will be excreted with Low HCO 3.

Correction of Metabolic Alkalosis • Depress ventilation Blood PCO 2 PH is back to normal. In blood : PH=~ Normal HCO 3 = high PCO 2 = high

Correction for Metabolic Acidosis • Correction : - Stimulate ventilation ( Hyperventilation ) PCO 2 PH Back to normal. In blood : PH =~ normal HCO 3 = Low PCO 2 = Low
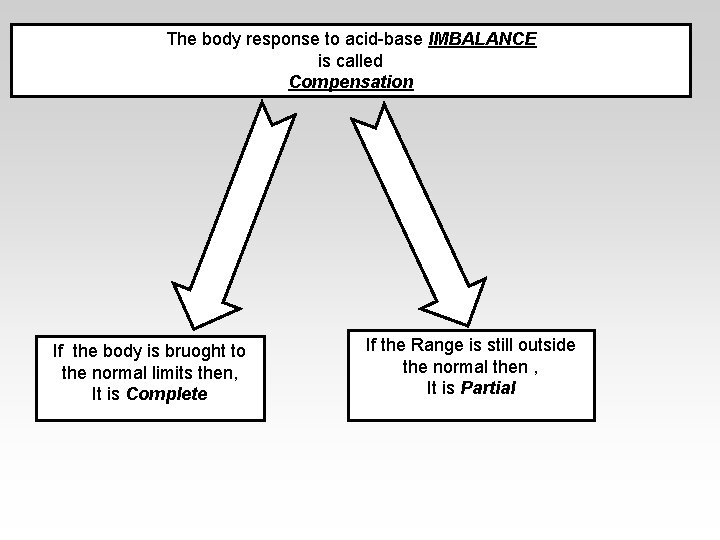
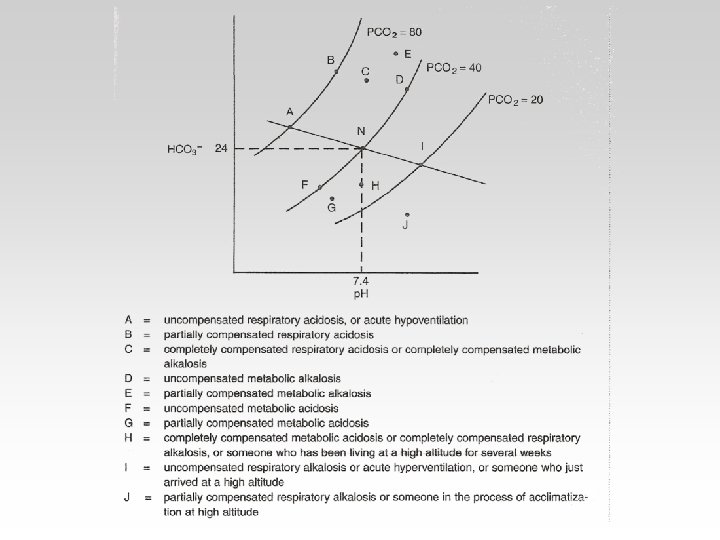
The body response to acid-base IMBALANCE is called Compensation If the body is bruoght to the normal limits then, It is Complete If the Range is still outside the normal then , It is Partial


1. A 44 -year – old woman with a long history of indigestion begins to vomit at home. She becomes unwell after 4 days and is admitted to hospital because of marked muscle weakness. Her arterial blood gases are [H+] 28 nmol/l, Pa. CO 2 6. 5 k. Pa, [HCO 3 - 40 mmol/l, Pa. O 210. 3 k. Pa, plasma potassium 2. 1 mmol/l. Q. What is the likely diagnosis and what treatment is required?

• Metabolic alkalosis is less common than metabolic acidosis. It is characterized by an increase in plasma bicarbonate, a fall in [H+], and a small compensatory rise in Pa. CO 2.

2. A 32 – year-old man is admitted in a very ill state. He had been drinking with friends, but had not been seen for some hours. On admission he is barely conscious and breathing heavily. His arterial blood gases are; [H+] 98 nmol/l, Pa. CO 2 2. 7 k. Pa, [HCO 3 - 6 mmol/l, Pa. O 2 13 k. Pa. Q. What is likely diagnosis, how should it be confirmed, and what treatment is required?

• Metabolic acidosis is characterized by a reduction in plasma bicarbonate and a rise in [H+]. The Pa. CO 2 is reduced secondarily by hyperventilation, which mitigates the rise in [H+].

• 3. a 56 -year-old man, who has smoked heavily for many years, develops a worsening cough with purulent sputum, and is admitted to hospital because of difficulty in breathing. He is drowsy and cyanosed. His arterial blood gases are [H+] 65 nmol/l, Pa. CO 2 9. 5 k. Pa, [HCO 3 - 28 mmol/l, Pa. O 26. 2 k. Pa, Q. What is likely diagnosis and what treatment is required?

• Respiratory acidosis arises when effective alveolar ventilation fails to keep pace with the rate of CO 2 production. As a result Pa. CO 2, blood [HCO 3 -] and [H+] all rise.

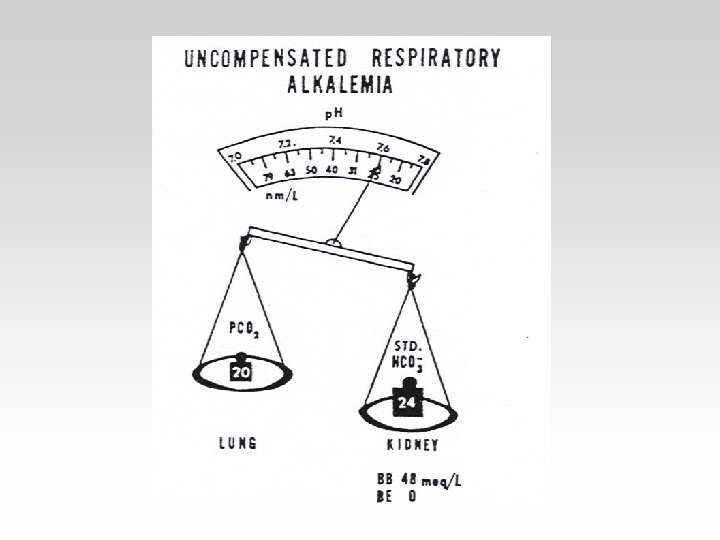
• 4. a 13 -year-school boy is brought the casualty department, having become acutely unwell in the headmaster’s office. He is alert and agitated, the respiratory rate is 35/min, and he complains of tingling in his hands. His arterial blood gases are; [H+] 29 nmol/l, Pa. CO 2 2. 8 k. Pa, [HCO 3 - 22 mmol/l, Pa. O 2 16 k. Pa. Q. What is the likely diagnosis and what treatment is required?

• Respiratory alkalosis occurs when there is excessive loss of CO 2 due to overventilation of the lungs. Pa. CO 2 and [H+] fall. The low Pa. CO 2 results in reduced renal Na+/H+ exchange.

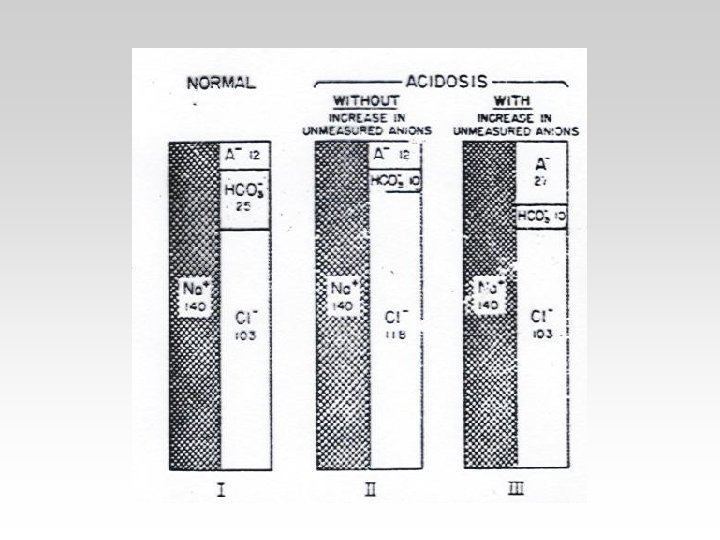
REASONS FOR METABOLIC ACIDOSIS AND ALKALOSIS
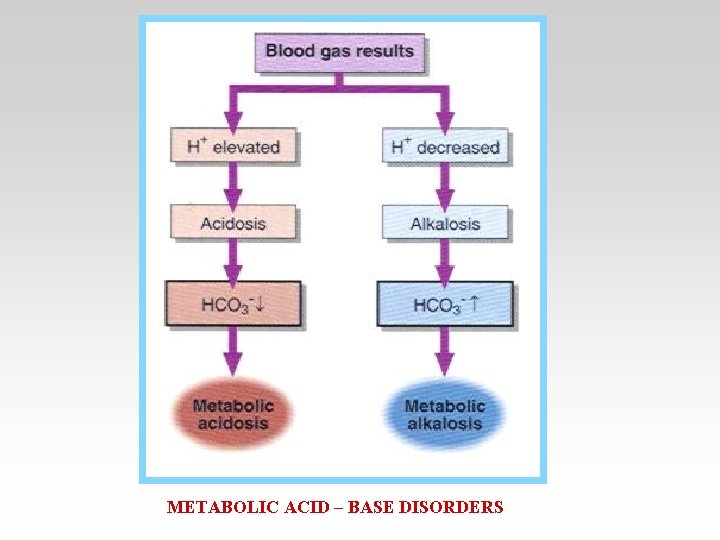
METABOLIC ACID – BASE DISORDERS
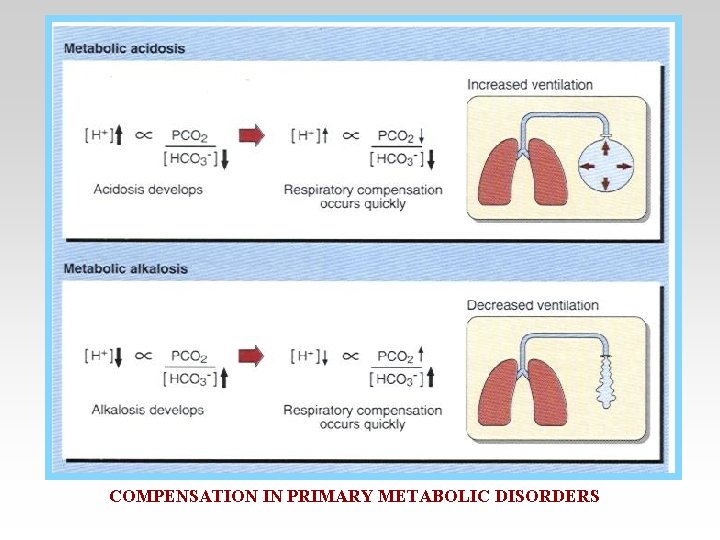
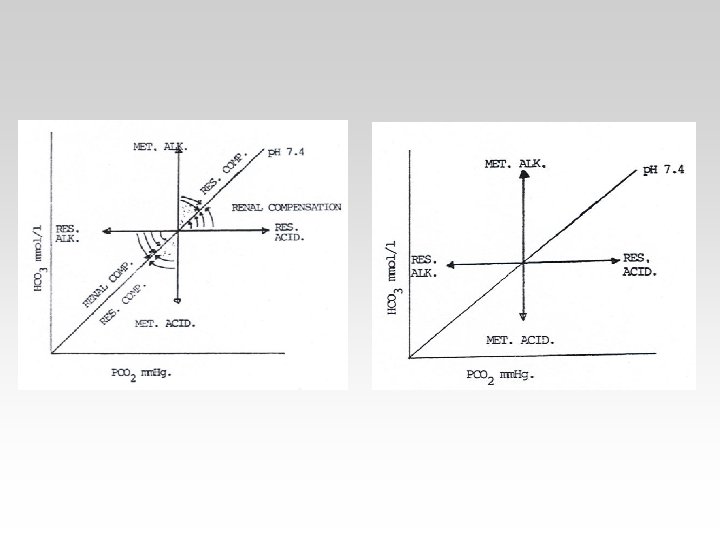
COMPENSATION IN PRIMARY METABOLIC DISORDERS
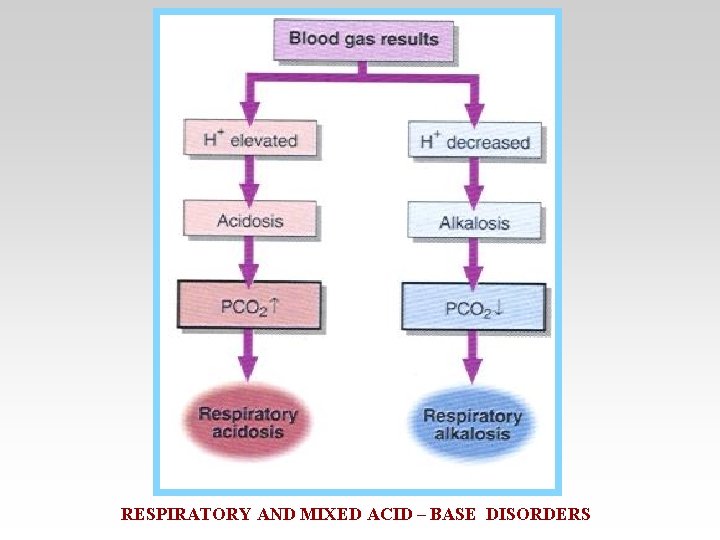
RESPIRATORY AND MIXED ACID – BASE DISORDERS

WHY AN INCREASED PCO 2 CAUSES AN ACIDOSIS
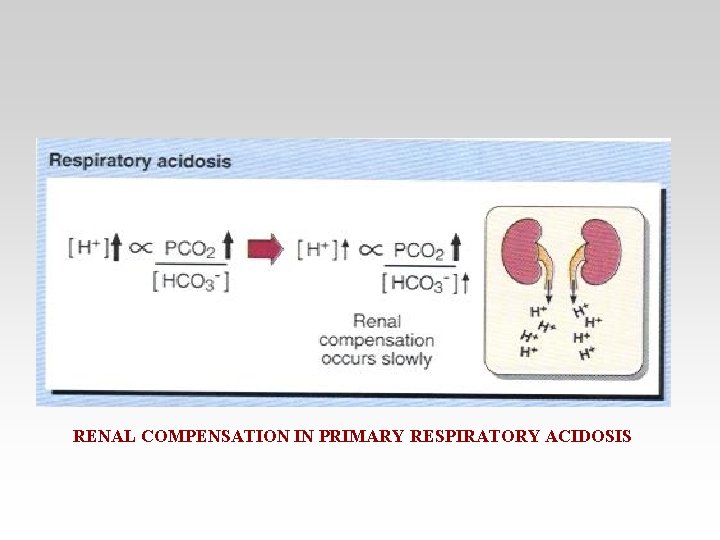
RENAL COMPENSATION IN PRIMARY RESPIRATORY ACIDOSIS
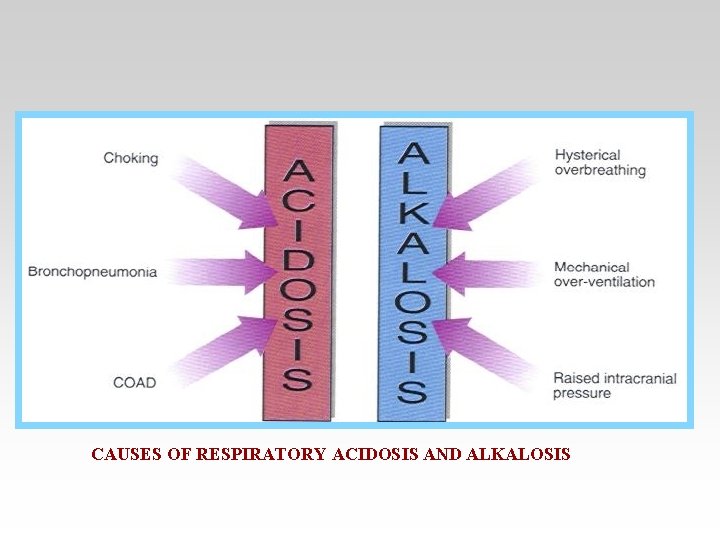
CAUSES OF RESPIRATORY ACIDOSIS AND ALKALOSIS
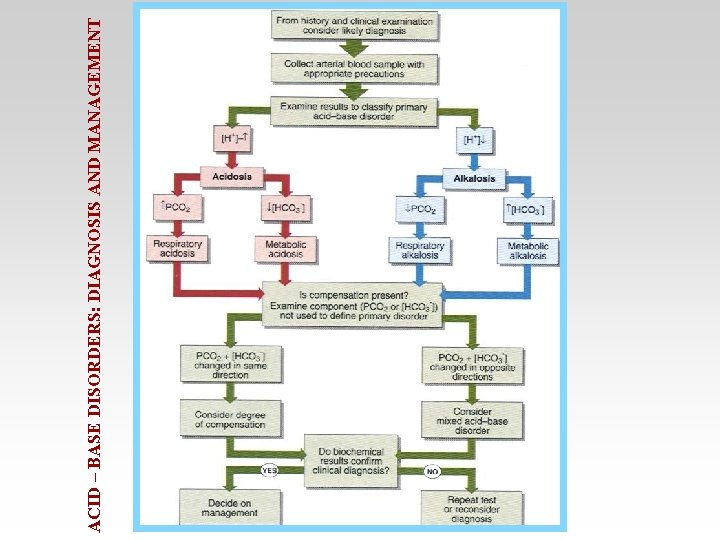
ACID – BASE DISORDERS: DIAGNOSIS AND MANAGEMENT
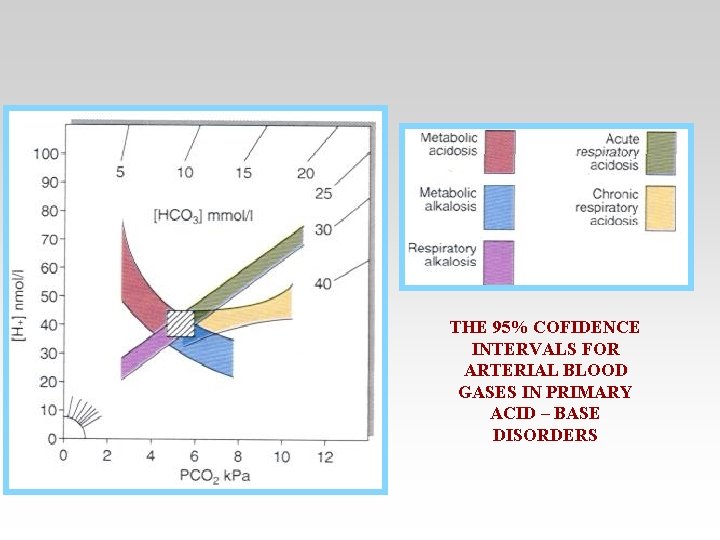
THE 95% COFIDENCE INTERVALS FOR ARTERIAL BLOOD GASES IN PRIMARY ACID – BASE DISORDERS












THANK YOU
- Slides: 76