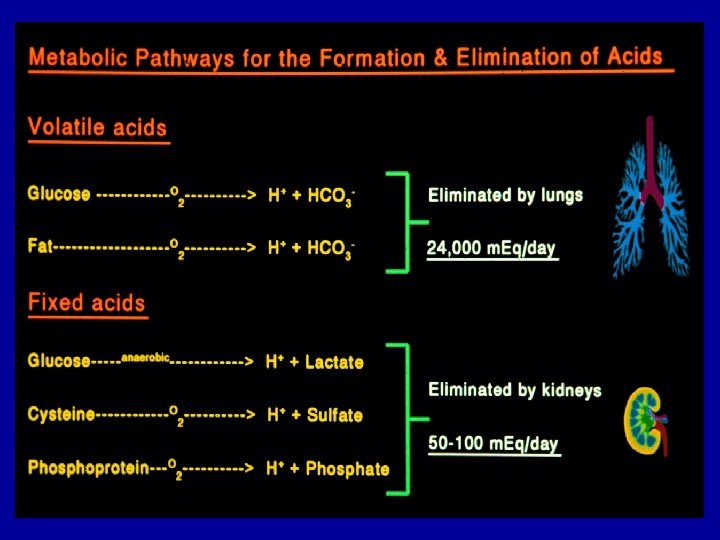
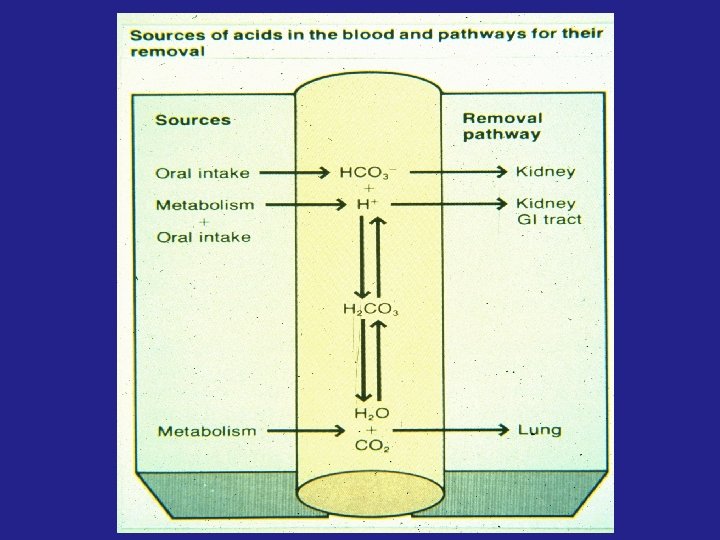
ACID BASE PHYSIOLOGY DEFINITIONS ACID can donate a


ACID - BASE PHYSIOLOGY DEFINITIONS • ACID - can donate a hydrogen ion • BASE – can accept a hydrogen ion • STRONG ACID – completely or almost completely dissociates into a hydrogen ion and its conjugate base in aqueous solution • WEAK ACID – only slightly dissociated in aqueous solution • Strong acids usually have weak conjugate bases; weak acids usually have strong conjugate bases • BUFFER – mixture of substances in aqueous solution. Usually a weak acid and its conjugate base, that can resist changes in hydrogen ion concentration when strong acids or bases are added.

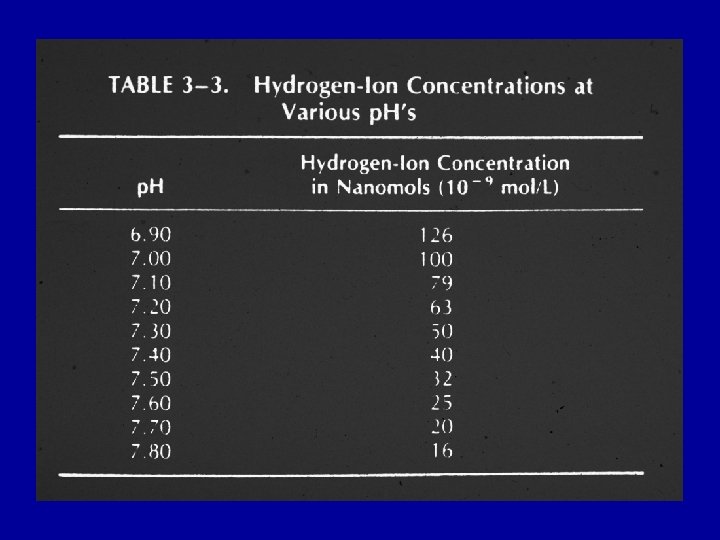
ACID - BASE PHYSIOLOGY DEFINITIONS p. H - negative log of hydrogen ion concentration ACIDOSIS - p. H < 7. 35 ALKALOSIS - p. H > 7. 45

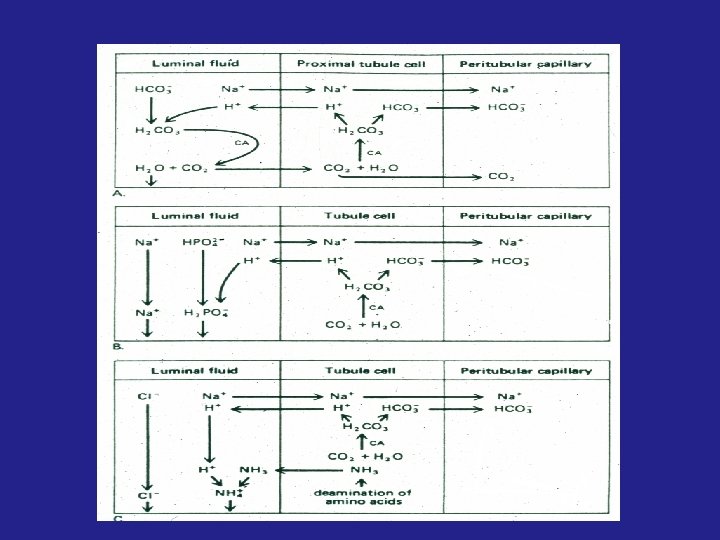
ACID - BASE PHYSIOLOGY BUFFERS OF THE BODY • ISOHYDRIC PRINCIPLE – All buffer pairs in a homogeneous solution are in equilibrium with the same hydrogen ion concentration • BICARBONATE • PHOSPHATE • BLOOD PROTEINS – especially hemoglobin • INTERSTITIAL FLUID – mainly bicarbonate, some phosphate • BONES – mainly phosphate in hydroxyapatite • INTRACELLULAR BUFFERS – intracellular proteins and organic phosphates
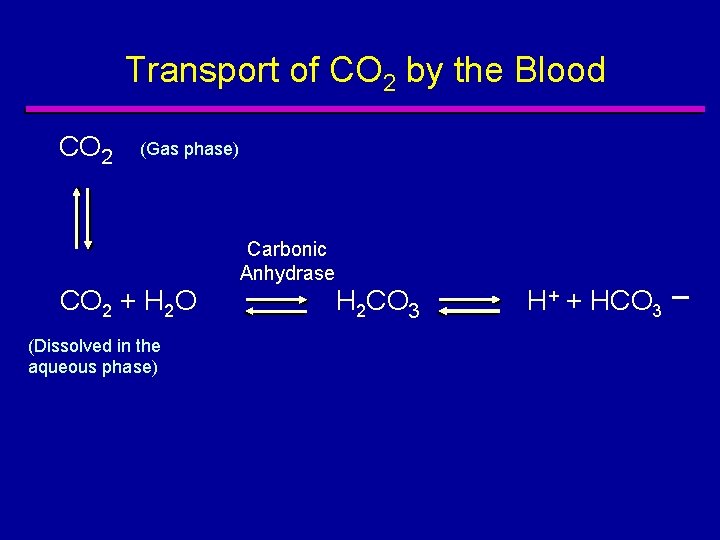
Transport of CO 2 by the Blood CO 2 (Gas phase) Carbonic Anhydrase CO 2 + H 2 O (Dissolved in the aqueous phase) H 2 CO 3 H+ + HCO 3 _

Taking Logarithms Since p. H is negative logarithm
![Henderson – Hasselbalch Equation p. H = p. K´ + log [HCO-3] p 0. Henderson – Hasselbalch Equation p. H = p. K´ + log [HCO-3] p 0.](http://slidetodoc.com/presentation_image_h/0c5bf3f0e6bb335ae74b4f2e9ef0d6ce/image-9.jpg)
Henderson – Hasselbalch Equation p. H = p. K´ + log [HCO-3] p 0. 03 x Pco 2
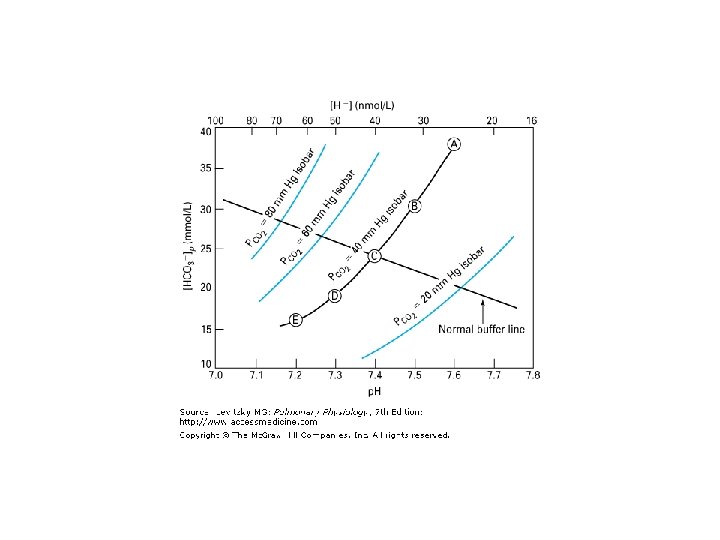
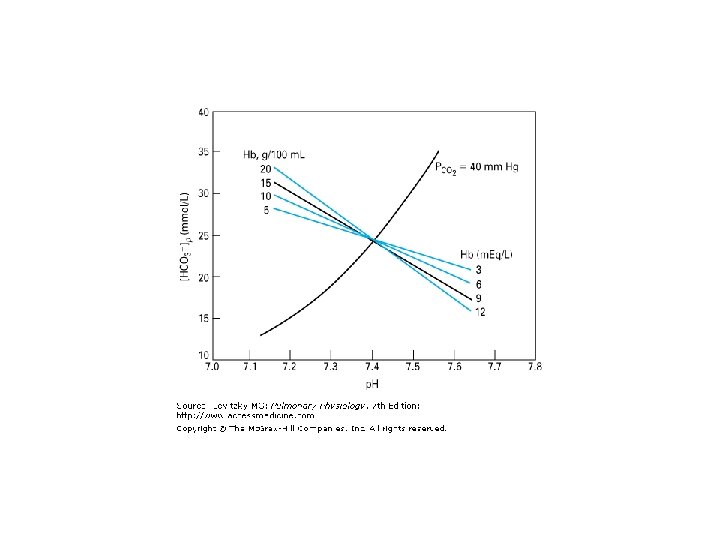
![Respiratory acidosis Metabolic alkalosis and respiratory F acidosis 40 [HCO 3 - ]p , Respiratory acidosis Metabolic alkalosis and respiratory F acidosis 40 [HCO 3 - ]p ,](http://slidetodoc.com/presentation_image_h/0c5bf3f0e6bb335ae74b4f2e9ef0d6ce/image-12.jpg)
Respiratory acidosis Metabolic alkalosis and respiratory F acidosis 40 [HCO 3 - ]p , mmol/ liter D 35 Compensated E 30 25 20 HCO 3 C Uncompensated respiratory acidosis I A Pco 2 B G 15 H 7. 0 7. 1 7. 2 7. 3 7. 4 p. H 7. 5 7. 6 7. 7 7. 8

Common Causes of Respiratory Acidosis Ø Depression of Respiratory Control Centers Ø Ø Ø Neuromuscular disorders Ø Ø Ø Ø Anesthetics Sedatives Opiates Brain injury or disease Severe hypercapnia, hypoxia Spinal cord injury Phrenic nerve injury Poliomyelitis Botulism, tetanus Myasthenia gravis Administration of curare-like drugs Diseases affecting the respiratory muscles Pulmonary diseases Ø Ø Acute asthma Pulmonary vascular disease

Common Causes of Respiratory Acidosis Ø Chest wall restriction Ø Ø Ø Lung restriction Ø Ø Pulmonary fibrosis Sarcoidosis Pneumothorax Pulmonary parenchymal diseases Ø Ø Ø Kyphoscoliosis Extreme obesity Pneumonia Pulmonary edema Airway obstruction Ø Ø Chronic obstructive pulmonary disease Upper airway obstruction
![Respiratory alkalosis F 40 [HCO 3 - ]p , mmol/ liter D HCO 3 Respiratory alkalosis F 40 [HCO 3 - ]p , mmol/ liter D HCO 3](http://slidetodoc.com/presentation_image_h/0c5bf3f0e6bb335ae74b4f2e9ef0d6ce/image-15.jpg)
Respiratory alkalosis F 40 [HCO 3 - ]p , mmol/ liter D HCO 3 - 35 E Pco 2 30 C 25 A Uncompensated respiratory B alkalosis I 20 G 15 Metabolic acidosis and respiratory alkalosis 7. 0 7. 1 7. 2 7. 3 7. 4 p. H H 7. 5 Compensated 7. 6 7. 7 7. 8

Common Causes of Respiratory Alkalosis Ø Ø Ø Central Nervous System Ø Anxiety Ø Hyperventilation Ø Inflammation (encephalitis, meningitis) Ø Cerebrovascular disease Ø Tumors Drugs or hormones Ø Salicylates Ø Progesterone Pulmonary diseases Ø Acute asthma Ø Pulmonary vascular disease (pulmonary embolism)

Common Causes of Respiratory Alkalosis Ø Ø Ø Bacteremias, fever Overventilation with mechanical ventilators Hypoxia, high altitude
![Metabolic acidosis F [HCO 3 - ]p , mmol/ liter 40 D 35 E Metabolic acidosis F [HCO 3 - ]p , mmol/ liter 40 D 35 E](http://slidetodoc.com/presentation_image_h/0c5bf3f0e6bb335ae74b4f2e9ef0d6ce/image-18.jpg)
Metabolic acidosis F [HCO 3 - ]p , mmol/ liter 40 D 35 E 30 HCO 3 - C 25 A Metabolic I acidosis 20 Uncompensated 15 metabolic acidosis 7. 0 7. 1 7. 2 Pco 2 B G Compensated H 7. 3 7. 4 p. H 7. 5 Metabolic acidosis and respiratory alkalosis 7. 6 7. 7 7. 8

Common Causes of Metabolic Acidosis Ø Ingested drugs or toxic substances Ø Ø Ø Loss of bicarbonate ions Ø Ø Methanol Alcohol Salicylates Ammonium Chloride Ethylene glycol Diarrhea Pancreatic Fistulas Renal dysfunction Inability to excrete hydrogen ions Ø Renal dysfunction

Common Causes of Metabolic Acidosis Ø Lactic acidosis Ø Ø Ø Hypoxemia Anemia, carbon monoxide Shock (hypovolemic, cardiogenic, septic, etc. ) Severe exercise Acute respiratory distress syndrome (ARDS) Ketoacidosis Ø Ø Ø Diabetes mellitus Alcoholism Starvation
![Metabolic alkalosis and respiratory F acidosis 40 Compensated [HCO 3 - ]p , mmol/ Metabolic alkalosis and respiratory F acidosis 40 Compensated [HCO 3 - ]p , mmol/](http://slidetodoc.com/presentation_image_h/0c5bf3f0e6bb335ae74b4f2e9ef0d6ce/image-21.jpg)
Metabolic alkalosis and respiratory F acidosis 40 Compensated [HCO 3 - ]p , mmol/ liter D 35 E Metabolic alkalosis 30 HCO 3 - C 25 Pco 2 A 20 I B G 15 H 7. 0 7. 1 7. 2 7. 3 7. 4 p. H 7. 5 7. 6 7. 7 7. 8

Common Causes of Metabolic Alkalosis Ø Loss of hydrogen ions Ø Ø Ø Vomiting Gastric fistulas Diuretic therapy Treatment with or overproduction of mineralocorticoids Ingestion or administration of excess bicarbonate Ø Ø Intravenous bicarbonate Ingestion of bicarbonate or other bases (e. g. antacids)

ACID - BASE PHYSIOLOGY DEFINITIONS BASE EXCESS OR DEFICIT = m. Eq of acid or base needed to titrate 1 liter of blood to a p. H of 7. 4 at 37ºC if the Pco 2 were 40 torr ANION GAP = [Na+] - ([Cl-] + [HCO 3 -]) Normally 12 ± 4 m. Eq / L If the anion gap is greater than 16 m. Eq / L : Lactic acidosis, ketoacidosis or organic anions Renal retention of sulfate, phosphate, or urate Decreased [K+], [Ca++], and/ or [Mg++]

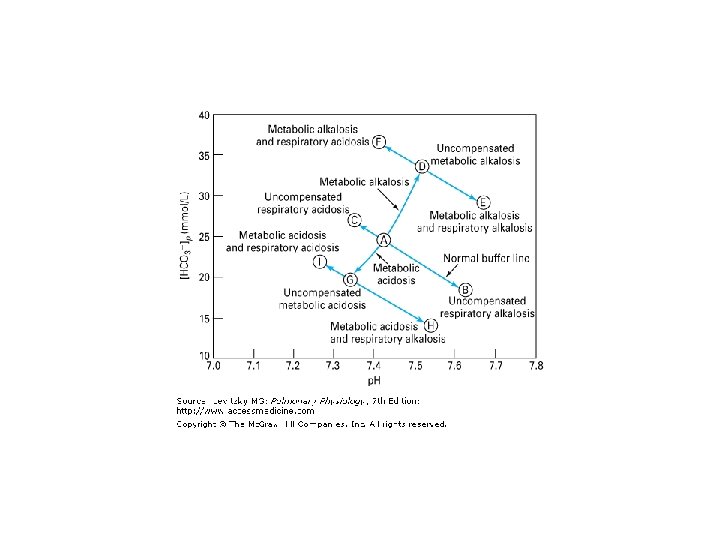
Acid- Base Disturbances p. H Uncompensated respiratory acidosis Uncompensated respiratory alkalosis Uncompensated metabolic acidosis Uncompensated metabolic alkalosis Partially compensated respiratory acidosis Partially compensated respiratory alkalosis Partially compensated metabolic acidosis Partially compensated metabolic alkalosis Respiratory and metabolic acidosis Respiratory and metabolic alkalosis Pco 2 _ HCO 3
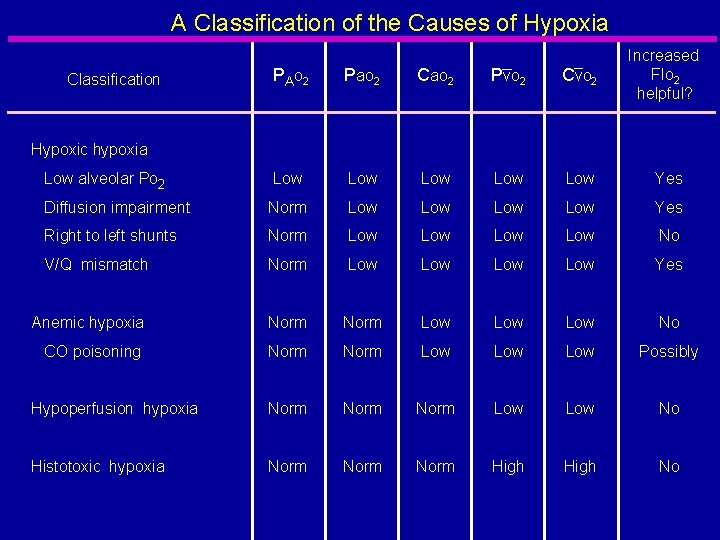
A Classification of the Causes of Hypoxia Increased FIo 2 helpful? PAo 2 Pao 2 Cao 2 Pvo 2 Cvo 2 Low Low Low Yes Diffusion impairment Norm Low Low Yes Right to left shunts Norm Low Low No V/Q mismatch Norm Low Low Yes Anemic hypoxia Norm Low Low No CO poisoning Norm Low Low Possibly Hypoperfusion hypoxia Norm Low No Histotoxic hypoxia Norm High No Classification Hypoxic hypoxia Low alveolar Po 2
- Slides: 28