Acid Base Electrolytes Regulation for BALANCE Fluid Compartments

Acid, Base, Electrolytes Regulation for BALANCE
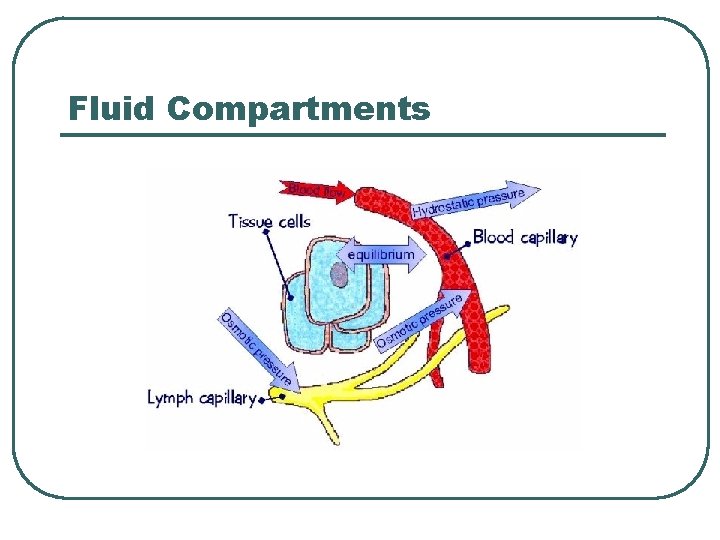
Fluid Compartments
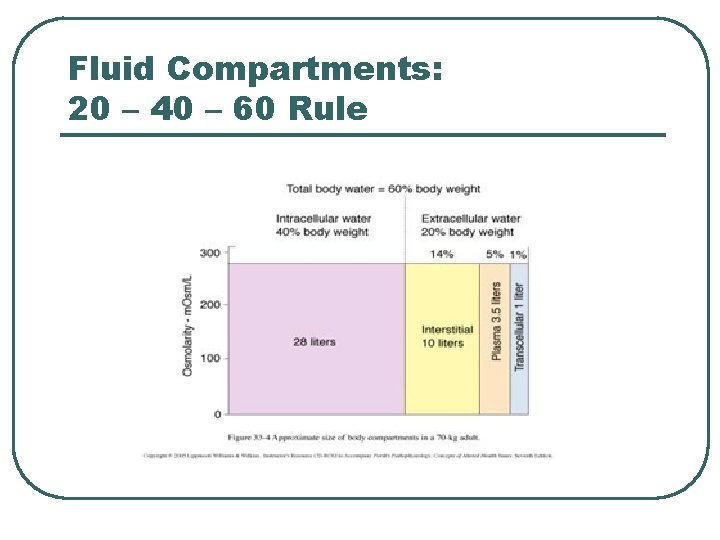
Fluid Compartments: 20 – 40 – 60 Rule
Fluid Movement

Water and Electrolyte Balance l l Input = output Hormones • • • Na+ / K+ • • • Renin Aldosterone ANP Reproductive Hormones GCC Ca++ / Mg++ • • Calcitonin PTH H 2 O • ADH l Anions follows passively • • Cl. HCO 3 -
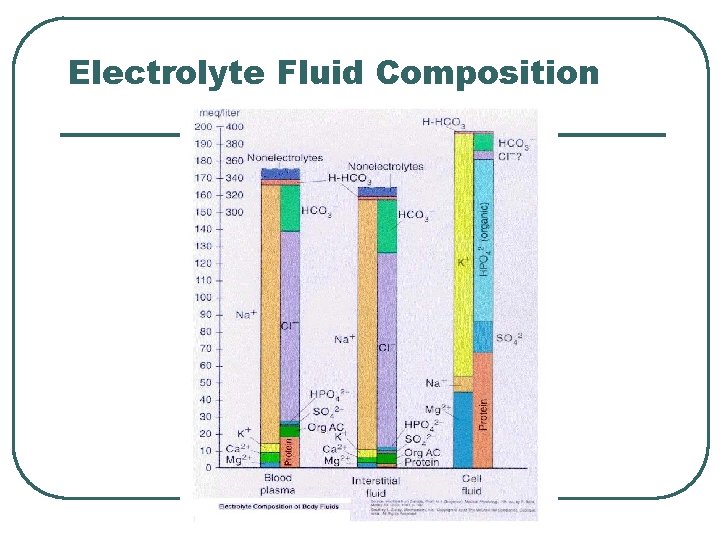
Electrolyte Fluid Composition
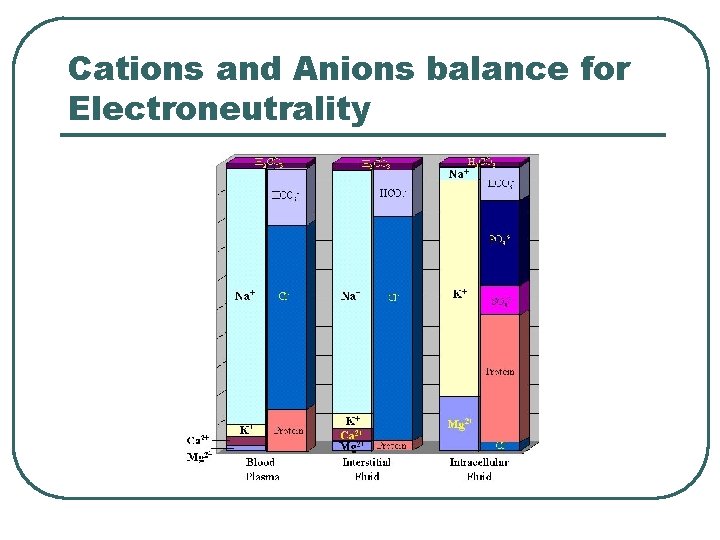
Cations and Anions balance for Electroneutrality

Acid Base Terms l Define • • p. H Acid • Base • • Salt Buffer • Strong • Weak • Volatile : CO 2 from CH 20 and Fat Metabolism • Nonvolatile: H 2 SO 4, H 2 PO 4 from protein metabolism • Strong • Weak
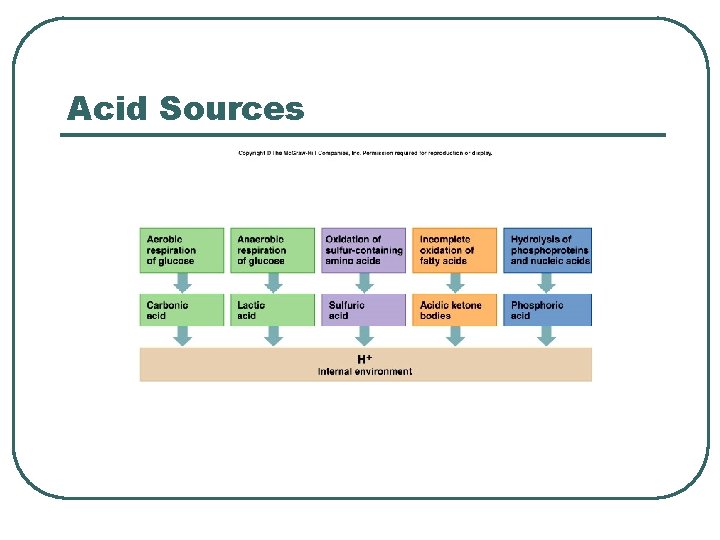
Acid Sources
![p. H l l l Define • • p. H = log (1/[H+]) p. p. H l l l Define • • p. H = log (1/[H+]) p.](http://slidetodoc.com/presentation_image_h/9a70e72ae830fc36e94bee449d0d525e/image-11.jpg)
p. H l l l Define • • p. H = log (1/[H+]) p. H = -log [H 3 O+] • H 2 O + H 2 O H 3 O+ + OH- Water Dissociation Scale Blood values • • Venous Arterial • • Acidemia Alkalemia Abnormal Values
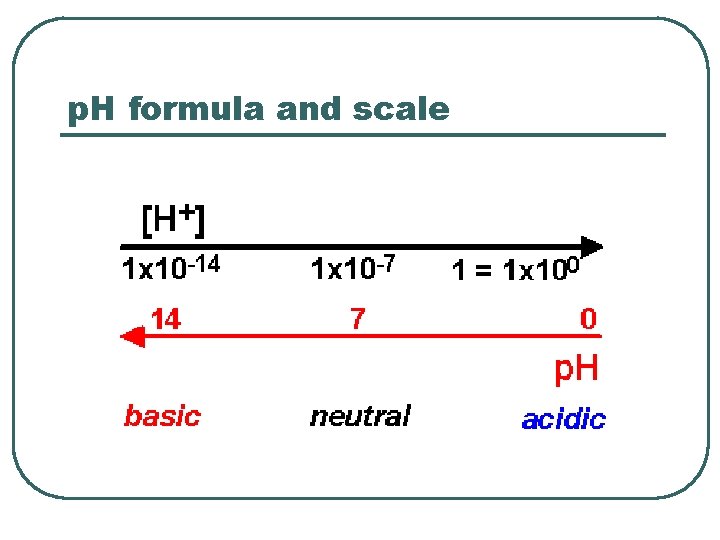
p. H formula and scale
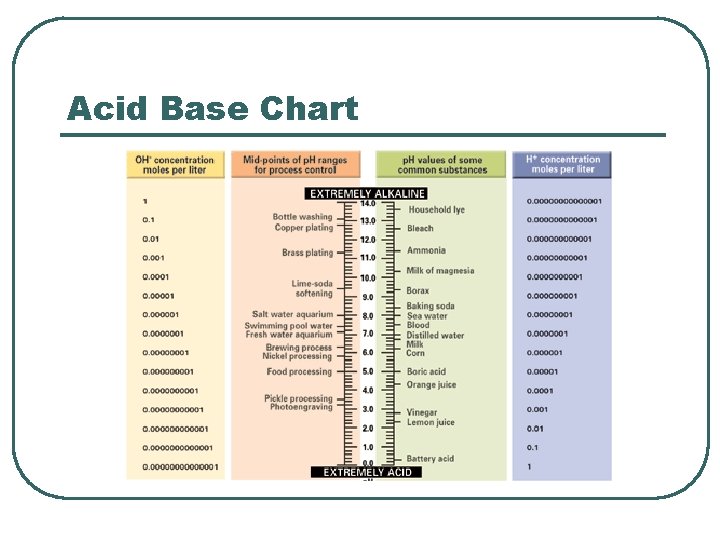
Acid Base Chart
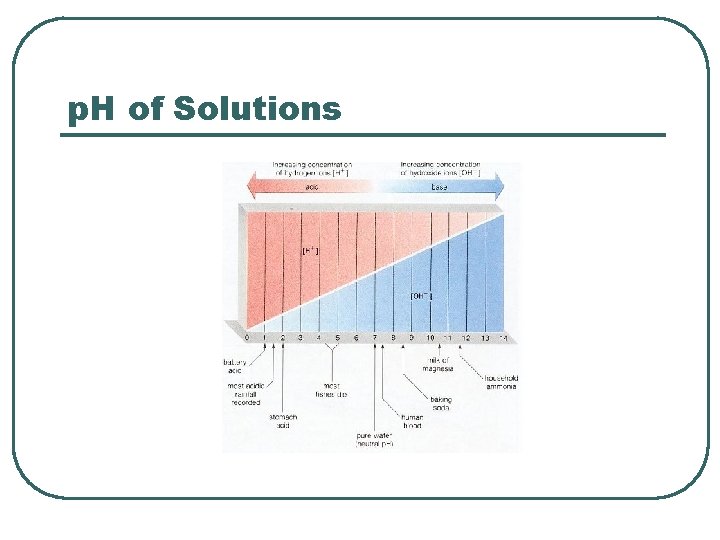
p. H of Solutions

Acid Base Regulation for Balance l l Systems • • • Chemical Buffer Systems Respiratory System Renal • • • Seconds to Minutes to Hours to Days / Weeks • • • + / - 0. 1 changes result in respiratory rate changes + / - 0. 2 to 0. 3 changes result in CV and Nervous changes + / - 0. 4 to 0. 5 changes result in death Time Strength Problems (reference 7. 4 as normal average):

Chemical Buffer Systems l l Define 3 types • Name of System • Buffer formula or name of chemical • Location • Effectiveness [p. Ka buffer = p. H location] • Why important
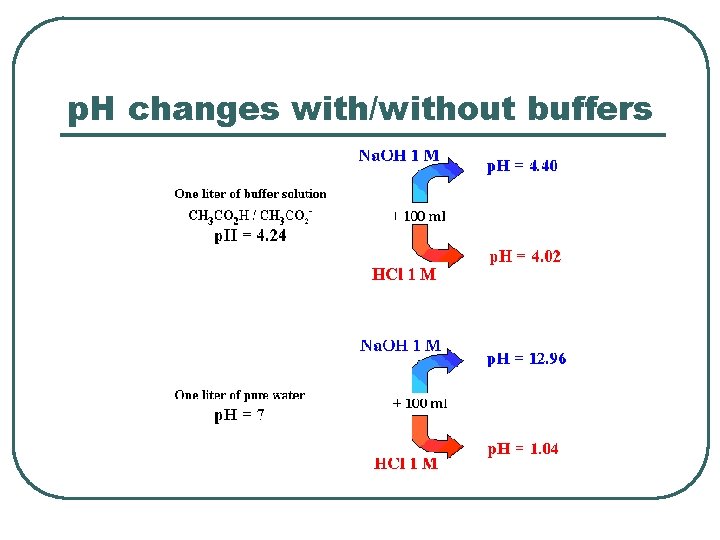
p. H changes with/without buffers
![Buffer Effectiveness HA [H+] + [A-] p. Ka = -log. Ka Buffer Effectiveness HA [H+] + [A-] p. Ka = -log. Ka](http://slidetodoc.com/presentation_image_h/9a70e72ae830fc36e94bee449d0d525e/image-18.jpg)
Buffer Effectiveness HA [H+] + [A-] p. Ka = -log. Ka
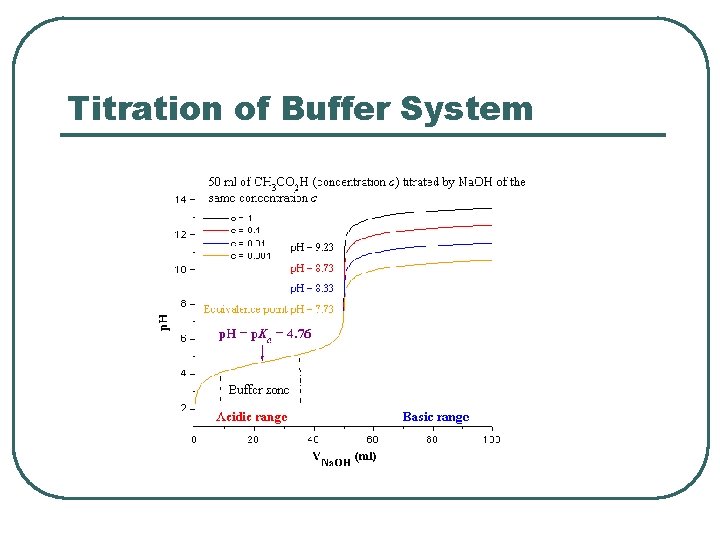
Titration of Buffer System
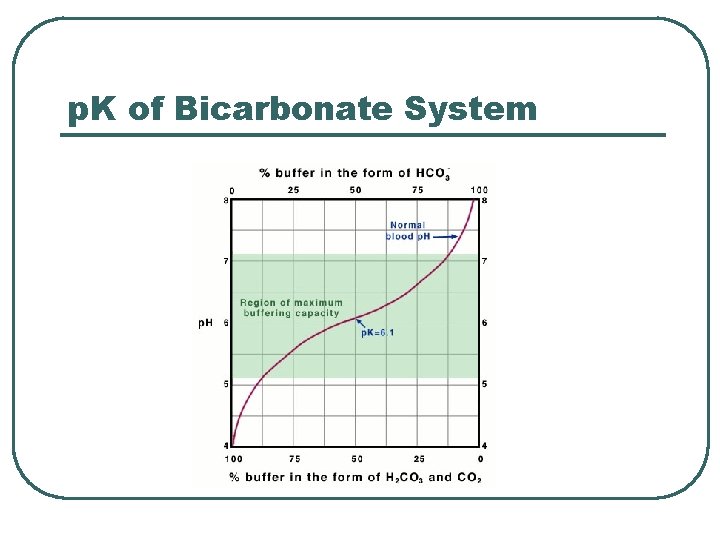
p. K of Bicarbonate System
![Formulas l l l K = [H+] [HCO 3 -] / [H 2 CO Formulas l l l K = [H+] [HCO 3 -] / [H 2 CO](http://slidetodoc.com/presentation_image_h/9a70e72ae830fc36e94bee449d0d525e/image-21.jpg)
Formulas l l l K = [H+] [HCO 3 -] / [H 2 CO 3] p. H = log (1 / [H+]) Henderson-Hasselbach Equation: CO 2 is directly related to H 2 CO 3, as CO 2 + H 20 H 2 CO 3; can substitute this equivalent [amount x solubility coefficient] in the above equation (0. 03 X p. CO 2)

Bicarbonate Chemical Buffer l l H 2 CO 3, HCO 3 Plasma buffer p. K = 6. 1 Important: • Can measure components • Can adjust concentration / ratio of components • Recalculate p. H of buffer system in ECF using Henderson-Hasselbach • • p. CO 2 = 40 mm. Hg HCO 3 - = 24 m. M • • HCO 3 - @ kidneys CO 2 @ lungs • p. H = 6. 1 + log(24 / 0. 03 x 40) • p. H = 6. 1 + log (20/1) • p. H = 7. 4
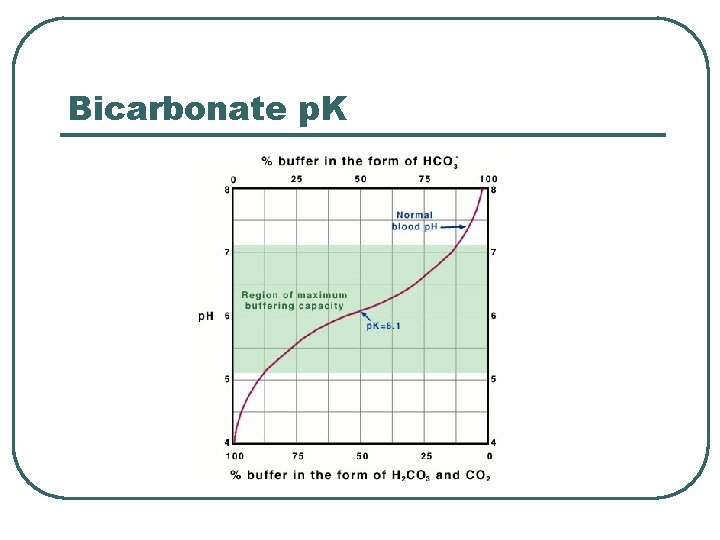
Bicarbonate p. K
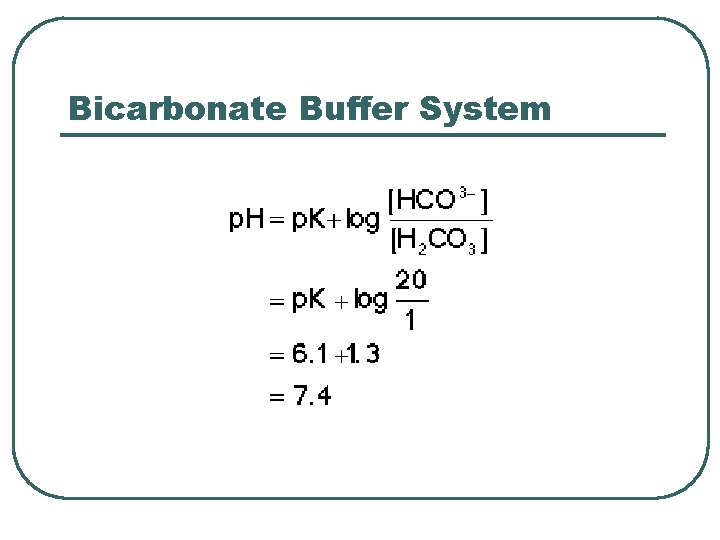
Bicarbonate Buffer System

Phosphate Chemical Buffer l l H 2 PO 4 -, HPO 4= ICF, Urine p. K = 6. 8 Important • Intracellular buffer • ICF p. H = ~ 6. 5 – 6. 8 • Renal Tubular Fluids • Urine p. H ranges 6. 0 – 7. 0

Protein Chemical Buffer l l Proteins • • • With Histadine: AA contain imidazole ring, p. Ka = 7. 0 R-COOH R-COO- + H+ R-NH 2 R-NH 3+ ICF (hemoglobin), ECF p. K = 7. 4 Important • • Most numerous chemicals Most powerful chemical buffer
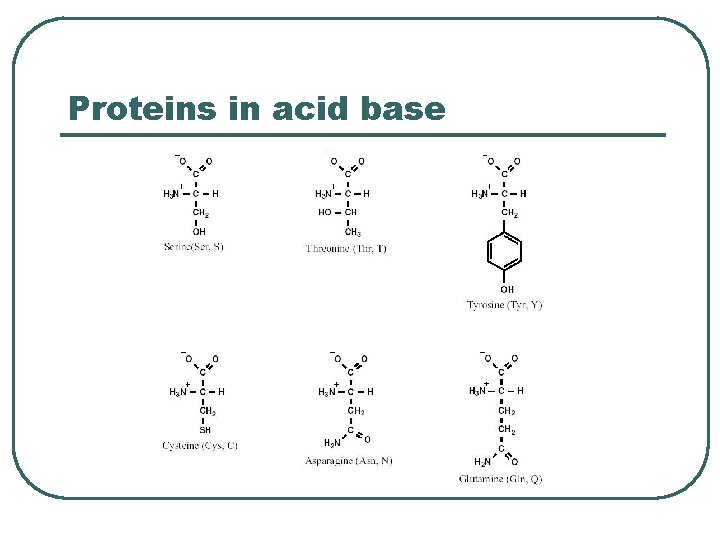
Proteins in acid base
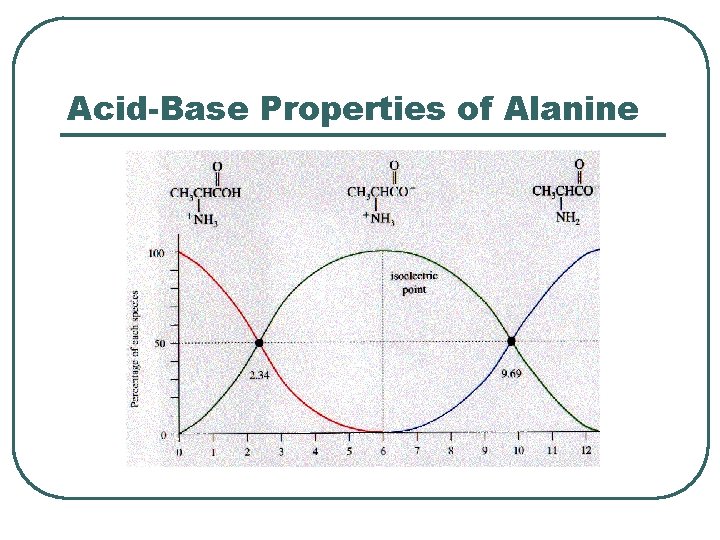
Acid-Base Properties of Alanine
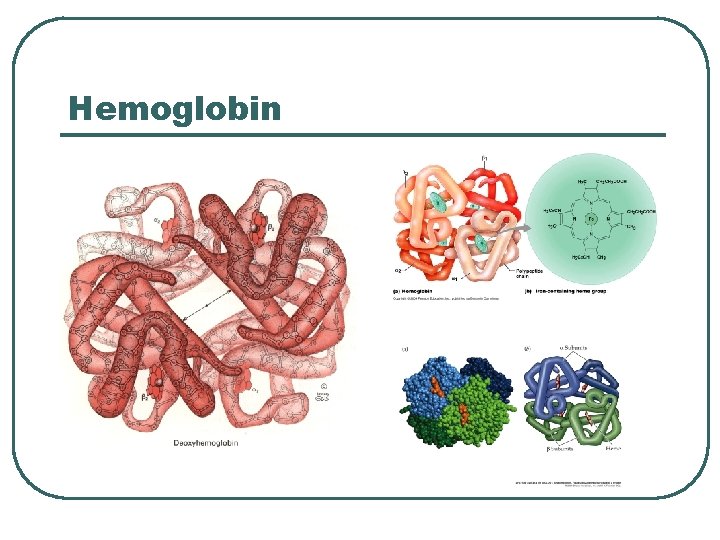
Hemoglobin
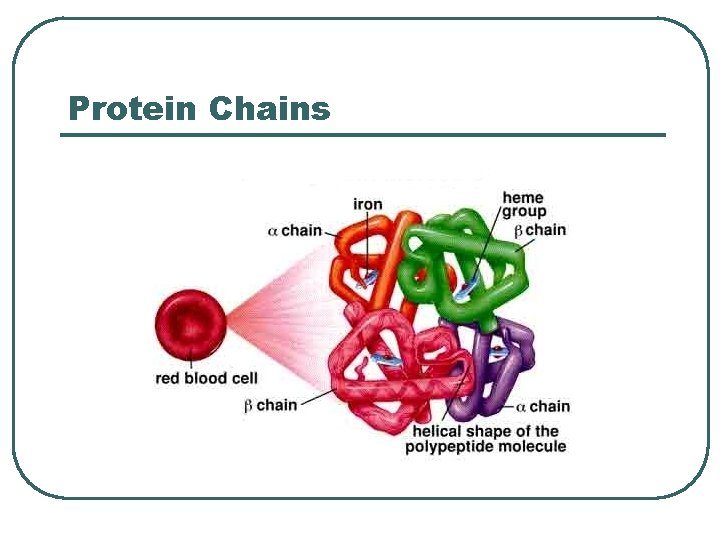
Protein Chains
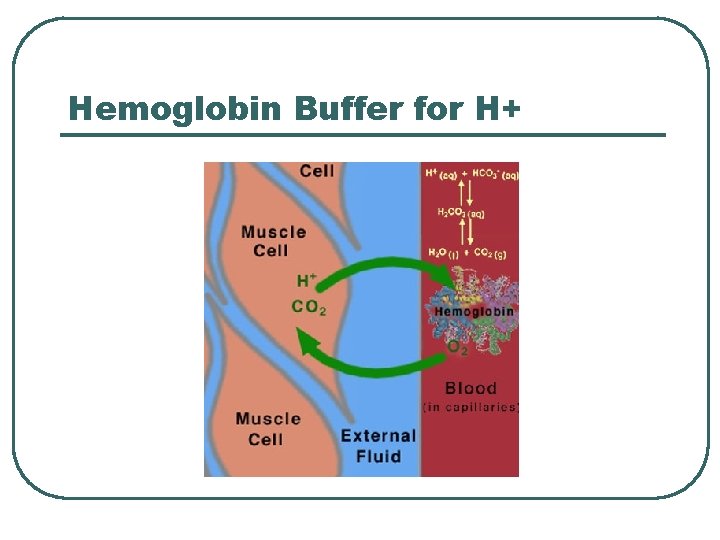
Hemoglobin Buffer for H+
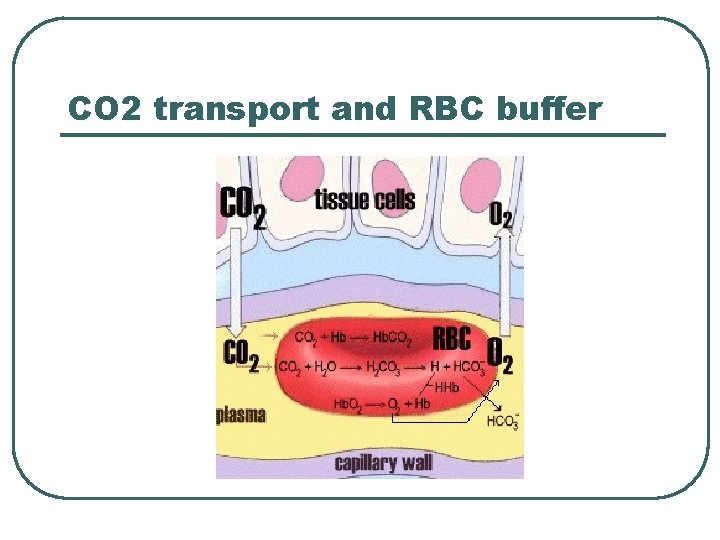
CO 2 transport and RBC buffer
Respiratory for A/B Balance l l l Occurs in minutes CO 2 only Rate changes

Respiratory Controls for Acid /Base balance l l Volatile Acid: CO 2 p. H changes in CSF Respiratory Rate • • Pons Medulla Oblongata Chemoreceptors • • p. CO 2 p. O 2
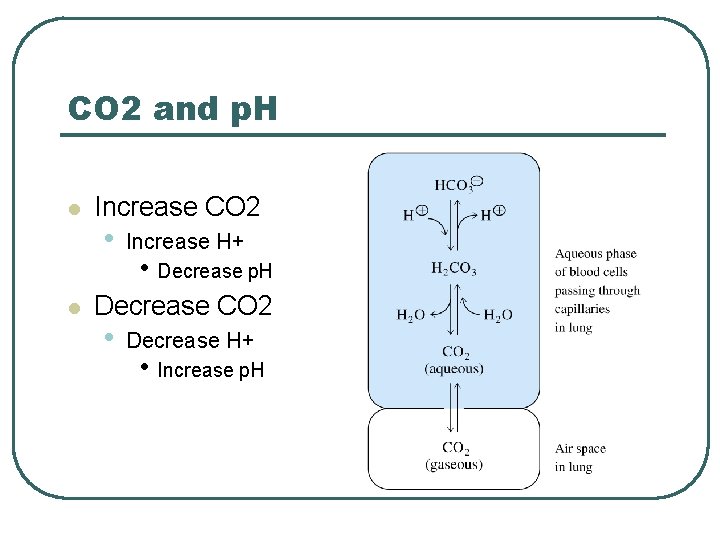
CO 2 and p. H l l Increase CO 2 • Increase H+ • Decrease p. H Decrease CO 2 • Decrease H+ • Increase p. H
Renal Control for Long Term Acid / Base Balance
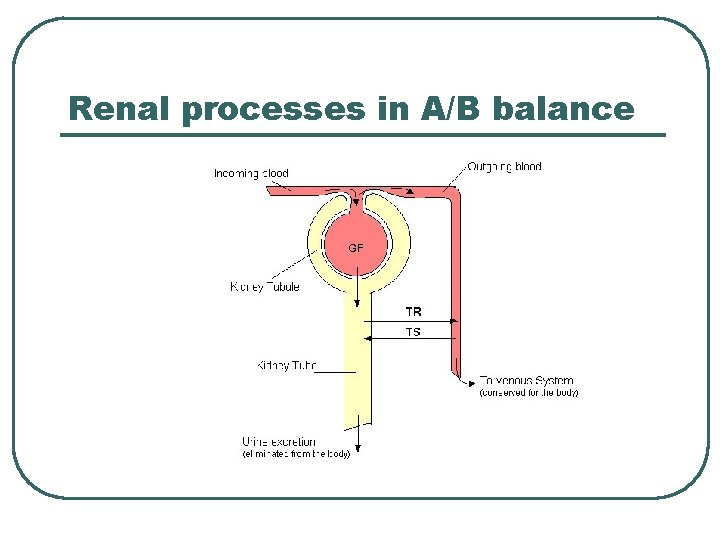
Renal processes in A/B balance
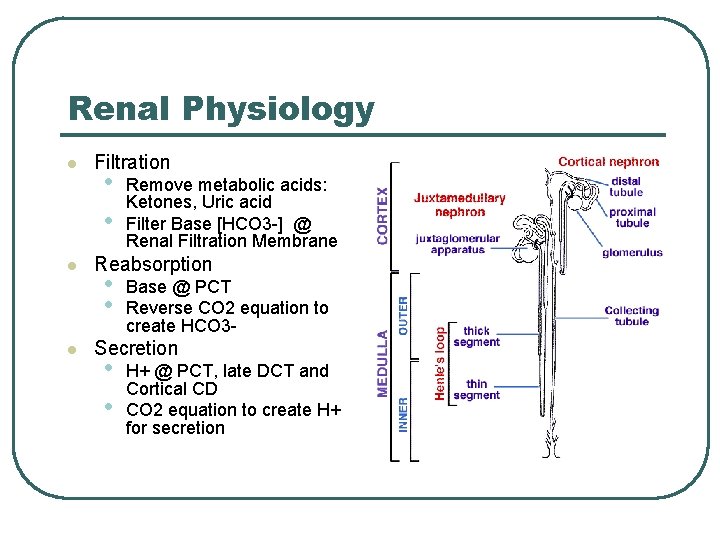
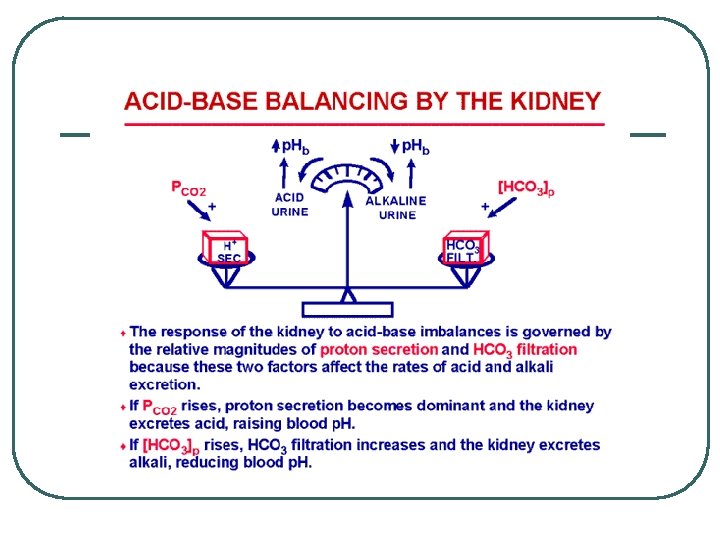
Renal Physiology l l l Filtration • • Remove metabolic acids: Ketones, Uric acid Filter Base [HCO 3 -] @ Renal Filtration Membrane Reabsorption • • Base @ PCT Reverse CO 2 equation to create HCO 3 - Secretion • • H+ @ PCT, late DCT and Cortical CD CO 2 equation to create H+ for secretion
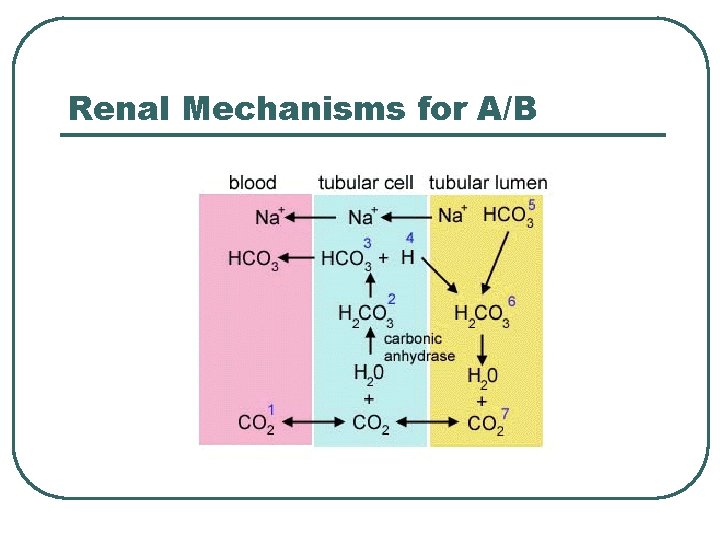
Renal Mechanisms for A/B
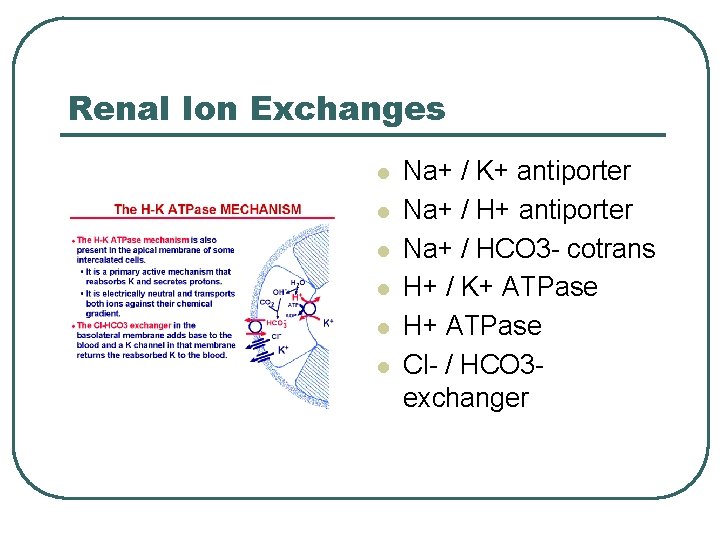
Renal Ion Exchanges l l l Na+ / K+ antiporter Na+ / HCO 3 - cotrans H+ / K+ ATPase H+ ATPase Cl- / HCO 3 exchanger
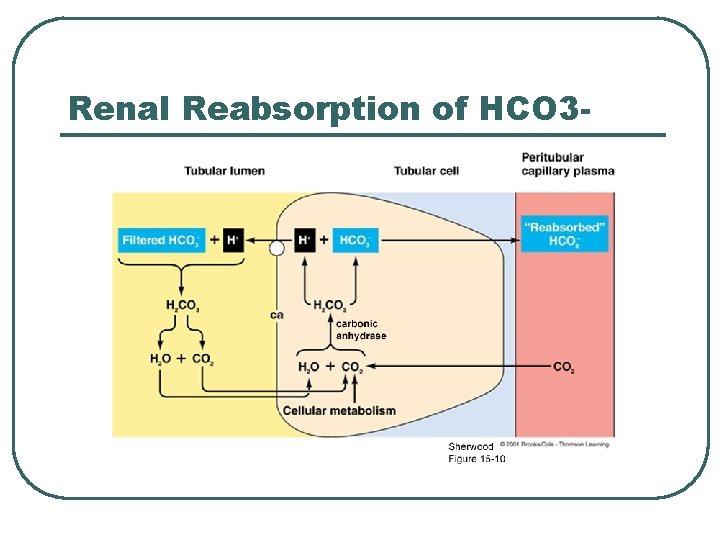
Renal Reabsorption of HCO 3 -
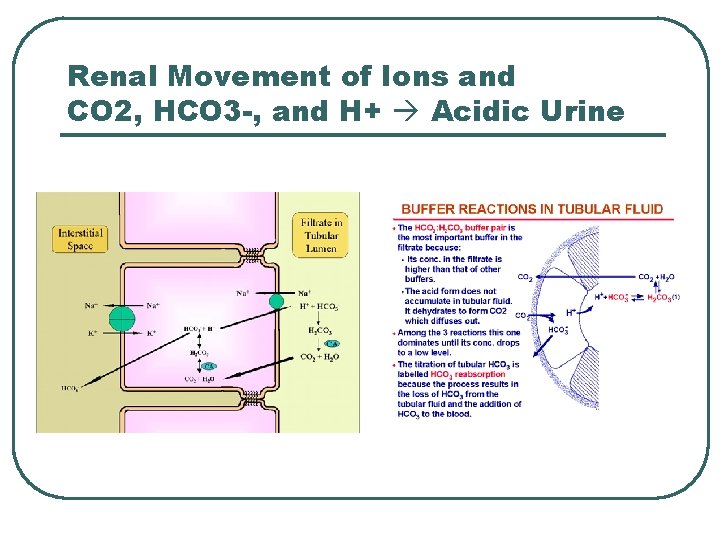
Renal Movement of Ions and CO 2, HCO 3 -, and H+ Acidic Urine
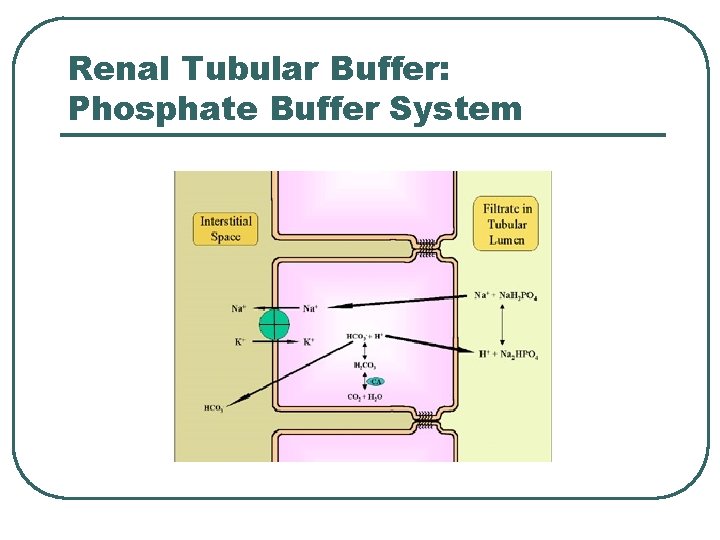
Renal Tubular Buffer: Phosphate Buffer System
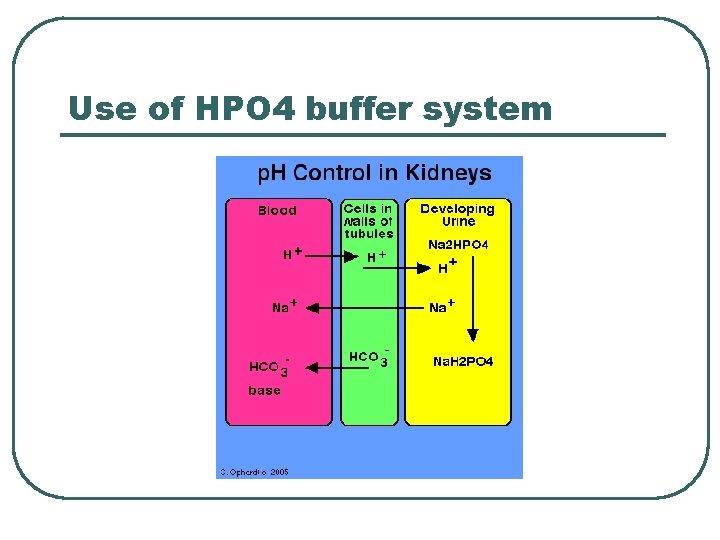
Use of HPO 4 buffer system
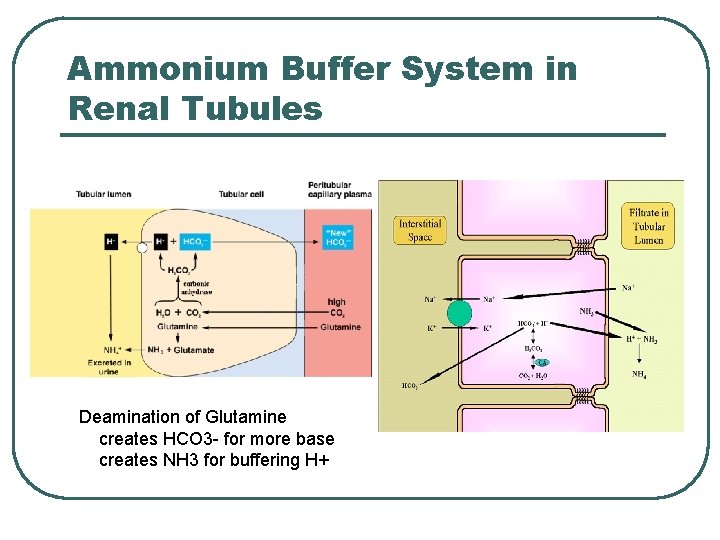
Ammonium Buffer System in Renal Tubules Deamination of Glutamine creates HCO 3 - for more base creates NH 3 for buffering H+
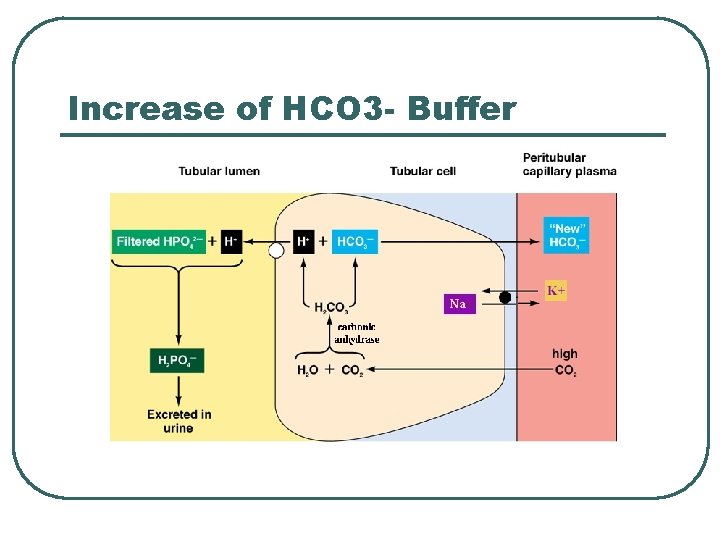
Increase of HCO 3 - Buffer
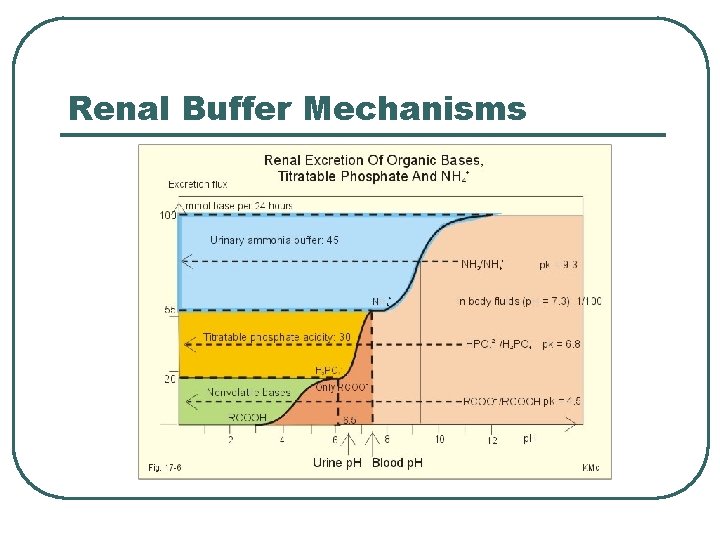
Renal Buffer Mechanisms
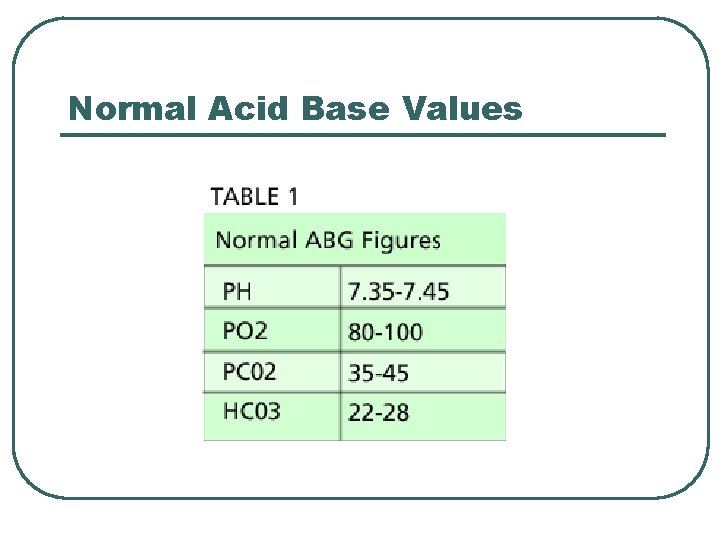
Normal Acid Base Values
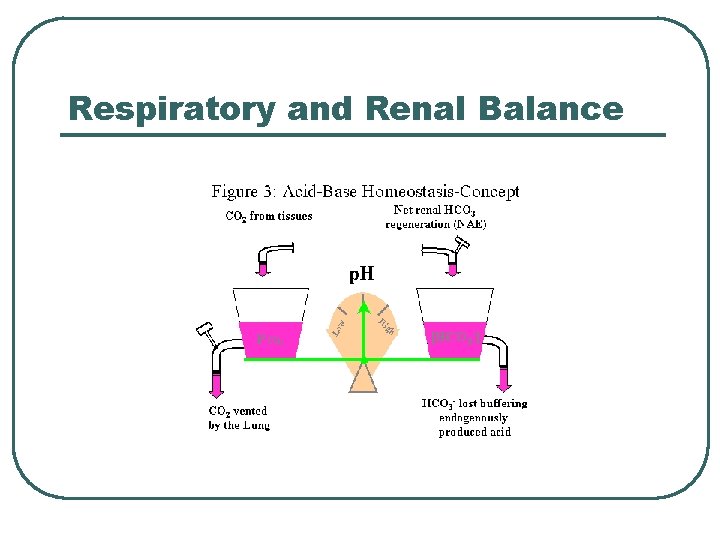
Respiratory and Renal Balance
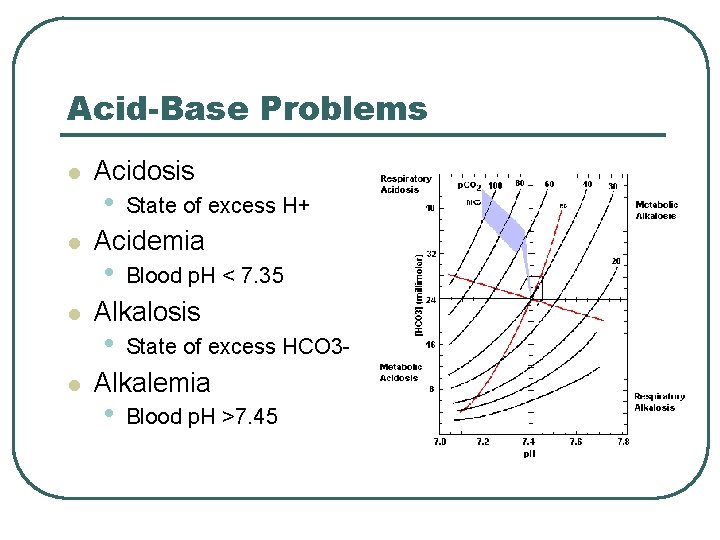
Acid-Base Problems l l Acidosis • State of excess H+ Acidemia • Blood p. H < 7. 35 Alkalosis • State of excess HCO 3 - Alkalemia • Blood p. H >7. 45

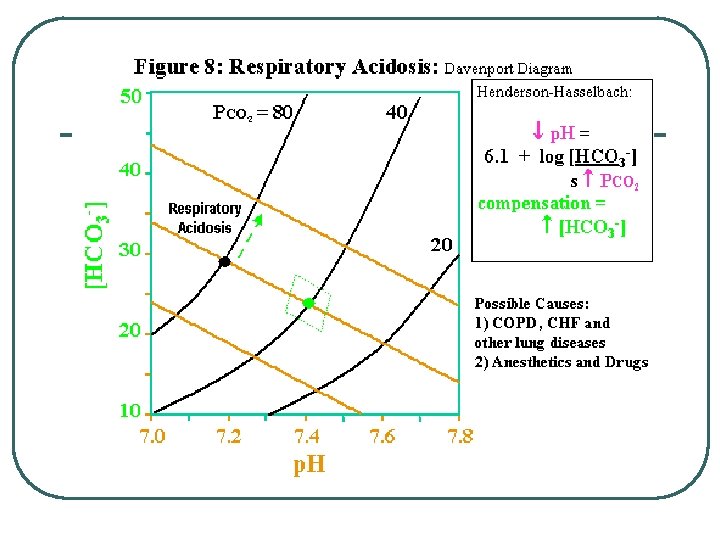
Classifying Respiratory Acid Base Problems (p. CO 2 changes) l l Respiratory Acidosis • • Respiratory Rate Decreases Any Respiratory Disease • Obstruction • Pneumonia • Gas exchange / transport problems • Respiratory Membrane • RBC / Hemoglobin Respiratory Alkalosis • Respiratory Rate Increases
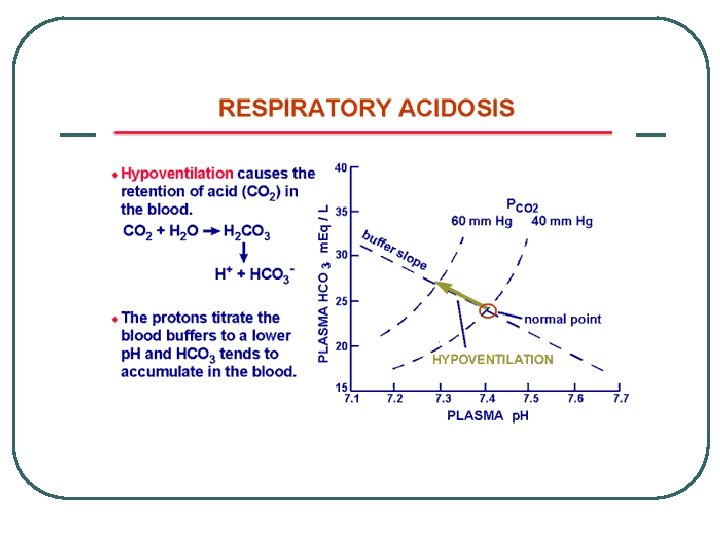

Classifying Metabolic Acid Base Balance Problems (H+/ HCO 3 -) l l l Systems • • Renal Endocrine GI Cardiovascular / Fluid administration • • Retain Acid Lose Base • • Retain Base Lose Acid Metabolic Acidosis Metabolic Alkalosis

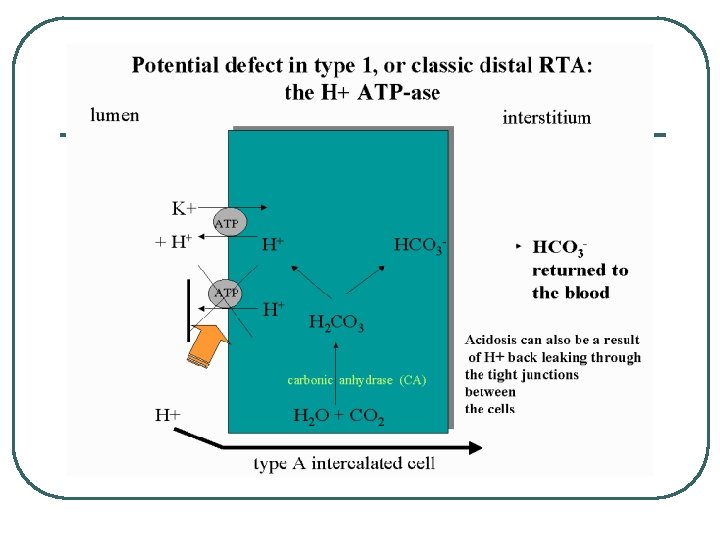
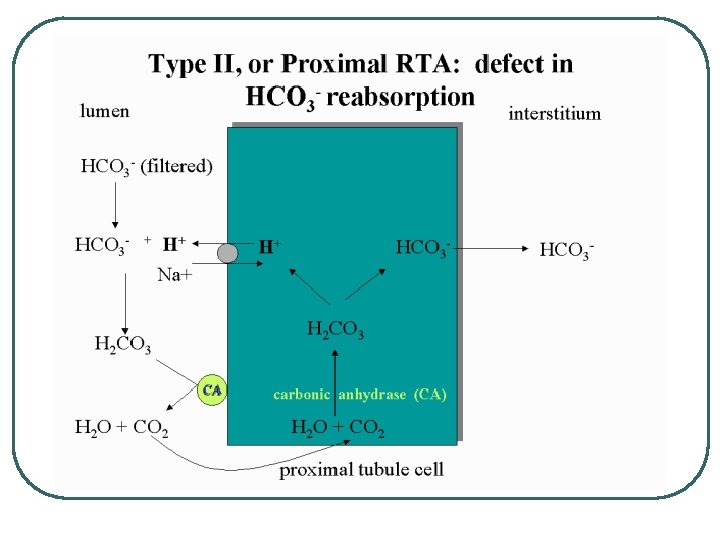
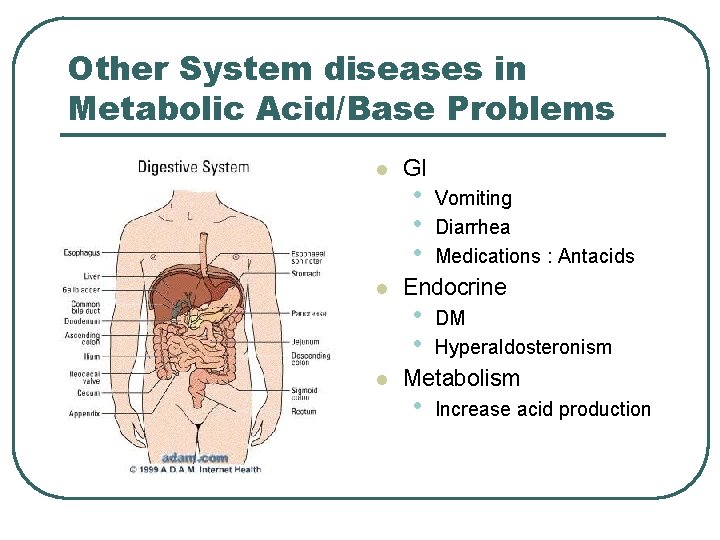
Other System diseases in Metabolic Acid/Base Problems l l l GI • • • Vomiting Diarrhea Medications : Antacids Endocrine • • DM Hyperaldosteronism Metabolism • Increase acid production
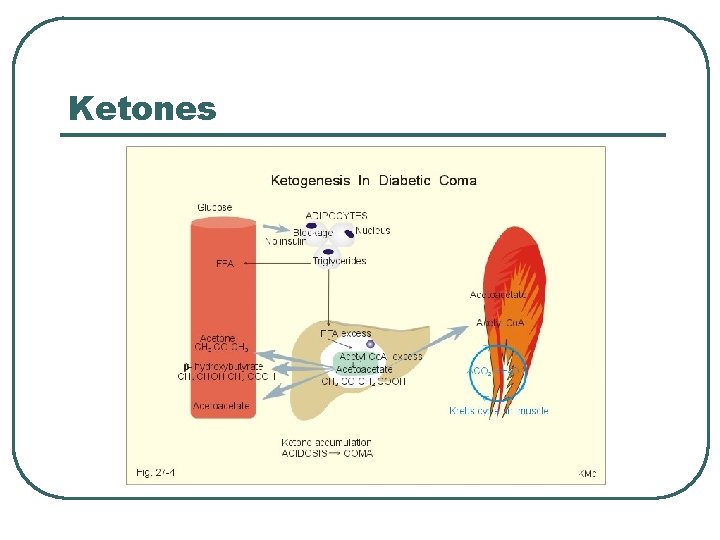
Ketones
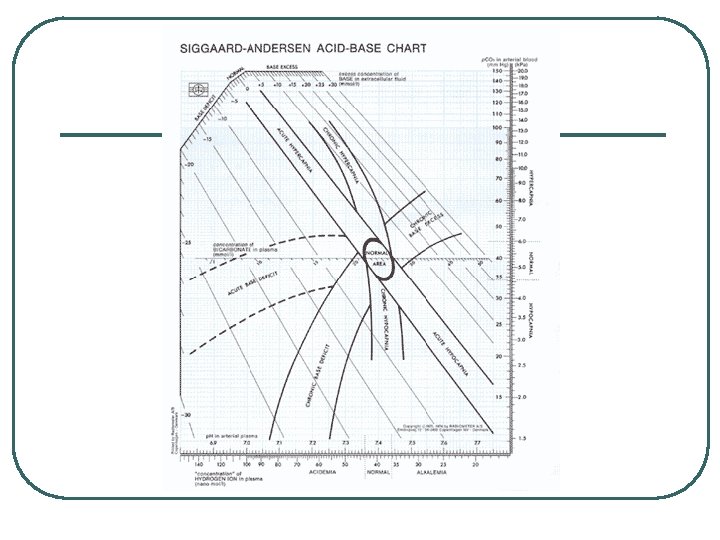

ECF Cations, Anions, and Anion Gap l Anion Gap • Difference between major plasma cations and major plasma anions AG = ([Na+] + [K+]) – ([Cl-] + [HCO 3 -]) Normal AG = 12 +/- 4 Check in metabolic Acidosis to help identify non-measured acids
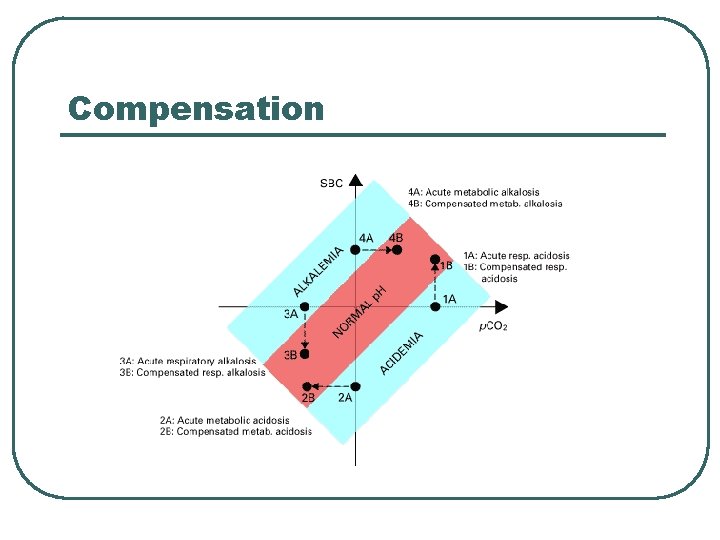
Compensation

Adjustments for Acid/Base Balance l Imbalance • • Respiratory Acidosis • Compensation • Increase renal acid excretion, Incr HCO 3 - • Decrease renal acid excretion, decr HCO 3 - • Hyperventilate to lower p. CO 2 • Hypoventilate to increase p. CO 2 Incr p. CO 2 Respiratory Alkalosis • l Decr p. CO 2 Metabolic Acidosis • • Decr HCO 3 Incr H+ Metabolic Alkalosis • • Incr HCO 3 Decr H+
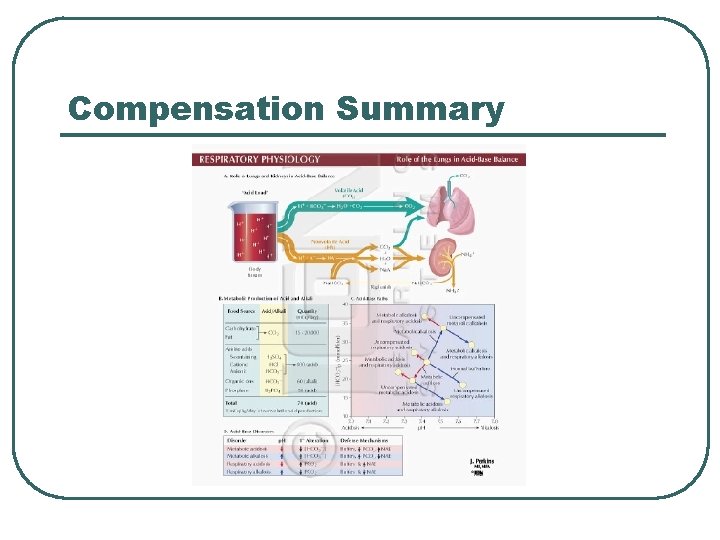
Compensation Summary

Summary for A/B Balance
- Slides: 66