Acid base disturbances Understanding Anion Gaps and Compensation

Acid base disturbances: Understanding Anion Gaps and Compensation Dr. Ankit Mehta DM Pediatric Critical Care Consultant Pediatric Intensivist Ahmedabad

p. H: is it so important ? • “ Life is a struggle, not against sin, not against money power, not against malicious animal magnetism, but against hydrogen ion”---H. L Menken • p. H is negative log of H+ ion concentration • 25% change in p. H adversely affects cells function (p. H <7. 30 or p. H >7. 52)

Effects of p. H Changes in hydrogen ions alter virtually all protein and membrane functions: • Cells enzymatic function is best @ physiological p. H • Potassium homeostasis • Regulate immune cells functions • Neurons and muscles (cardiac, skeletal) excitability (low p. H decreased excitability and high p. H increases excitability)

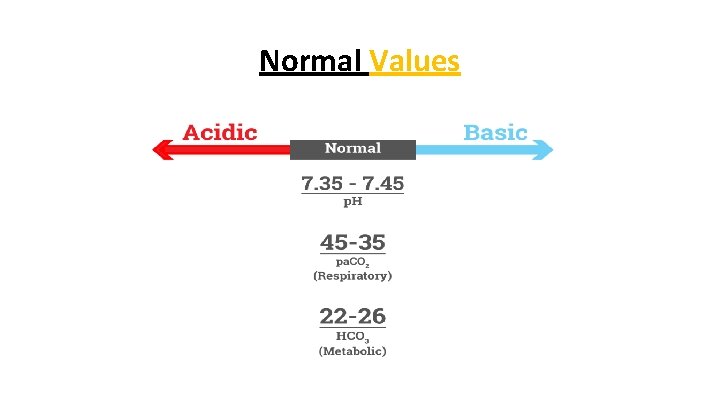
Analyze the p. H • Normal blood p. H is 7. 4 ± 0. 05 • Range 7. 35 to 7. 45 • p. H below 7. 35 is acidic • p. H above 7. 45 is alkalotic • Falls between 7. 35 to 7. 45 label what side of 7. 4 it falls on • Between 7. 35 to 7. 4 it is normal/acidic
Normal Values

Analyze the CO 2 • Normal p. CO 2 levels are 35 to 45 mm Hg • Below 35 is alkalotic • Above 45 is acidic
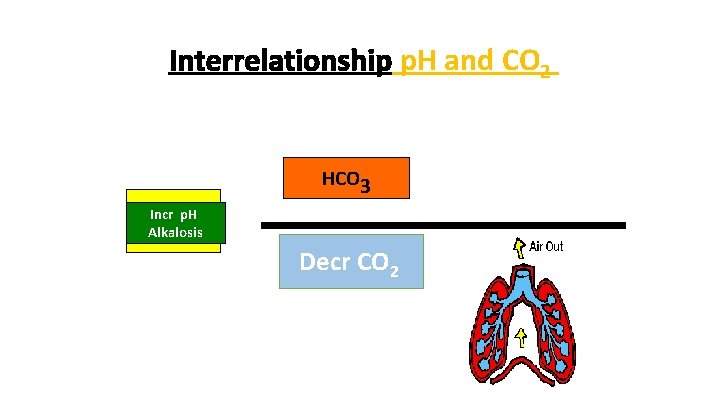
Interrelationship p. H and CO 2 HCO 3 p. H < 7. 4 Incr p. H Ph – 7. 4 Acidosis Alkalosis = Incr. CO CO 2 Decr 2

Analyze the HCO 3 • Normal HCO 3 level is 22 -26 m. Eq/l • If HCO 3 level is below 22, the patient is acidotic • If HCO 3 level is above 26, the patient is alkalotic
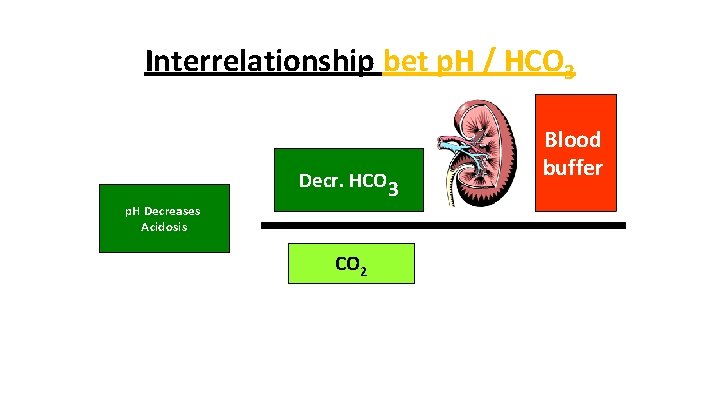
Interrelationship bet p. H / HCO 3 Ph increases p. H Decreases PhØ– 7. 4 Acidosis ØAlkalosis HCO Decr. HCO Incr HCO 3 33 = CO 2 Blood buffer

Match the C 02 or the HCO 3 with the p. H • If the p. H is acidotic and the CO 2 is acidotic v Acid base disturbance is caused by respiratory system , call it respiratory acidosis • If the p. H is alkalotic and the HCO 3 is alkalotic v Acid base disturbance caused by renal system, call it metabolic alkalosis
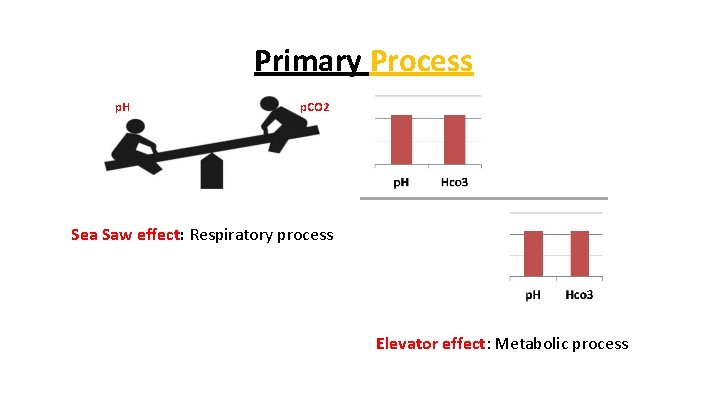
Primary Process p. H p. CO 2 Sea Saw effect: Respiratory process Elevator effect: Metabolic process

How to look for compensation ? • Does either the CO 2 or HCO 3 go in the opposite direction of the p. H • If p. H is acidotic, CO 2 is acidotic and HCO 3 is alkalotic v So HCO 3 is opposite the p. H , evidence of compensation from the metabolic system

Commandments of Compensation # 1 • Lung responsible for respiratory compensation • kidney responsible for metabolic compensation • kidney will absorb or excrete HCO 3 with change in p. CO 2 in same way • Lung will also wash out or retain CO 2 with change in HCO 3 in same way

Commandments of Compensation # 2 • Tries to normalize the p. H (7. 35 -7. 45) • Never over compensate or normalize p. H (7. 40) • If p. H normal think of mixed disorder • Respiratory compensation start within minutes, maximum 12 -24 hours • In metabolic disorder p. CO 2 >65 mm. Hg is not possible

Commandments of Compensation # 3 • Metabolic compensation v Start within hours (HCO 3 absorption) v Peak 3 -5 days (NH 3 secretion) • Maximal increase in HCO 3 v During acute compensation is 31– 32 meq/L v Maximal increase of approximately 45 meq/L in chronic respiratory acidosis

Commandments of Compensation # 4 Primary disorders Compensatory response Metabolic Acidosis Increased minute ventilation Metabolic Alkalosis Decreased minute ventilation Respiratory Acidosis Increased Renal reabsorption of HCO 3 in proximal tubules Increased Renal excretion of H+ in distal tubules Respiratory Alkalosis Decreased Renal reabsorption of HCO 3 in proximal tubules Decreased Renal excretion of H+ in distal tubules
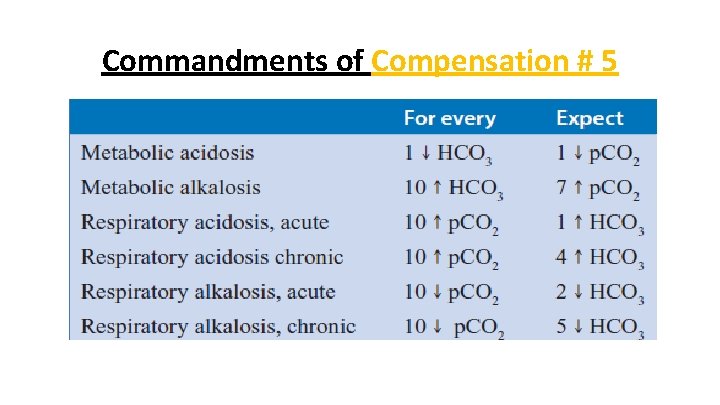
Commandments of Compensation # 5

Law of electrochemical neutrality • Any given solution, the net charge after accounting for all cations (positive ions) and anions (negative ions) • Should arrive at zero

Anion Gap • Difference between unmeasured anions and unmeasured cations • Equal to the difference between the plasma concentrations of the major cation (Na+) and the major measured anions (Cl-+HCO 3 -) • (Anionic proteins + inorganic phosphate + sulphate + organic anions)(potassium + calcium + magnesium + cationic proteins)
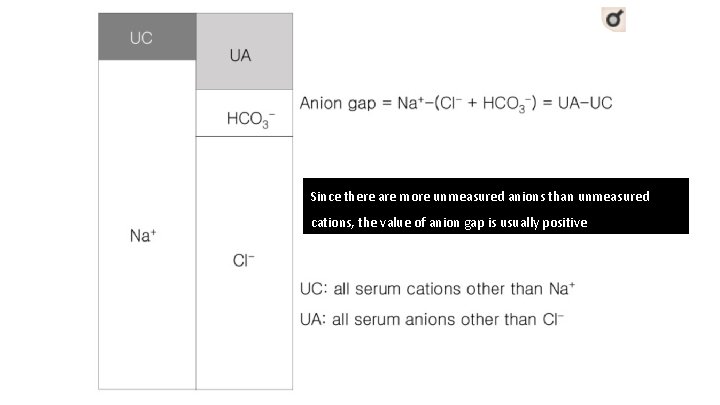
Since there are more unmeasured anions than unmeasured cations, the value of anion gap is usually positive

Utility of Anion gap • Evaluate for suspected acid-base disorders • Identify errors in the measurement of electrolytes • Detect paraproteins

Normal values of Anion Gap • Typically about 10 to 12 mmol • Clinicians should know about measuring methods for serum sodium and chloride in the laboratories to understand the range of normal anion gap

Bedside role of Anion Gap • For metabolic acidosis • Anion gap = Na – (Cl + HCO 3) • Corrected AG= AG + 2. 5 (4. 4 - serum albumin g/d. L) + 2. 5 (4. 4 - Po 4 mg/dl) • If the anion gap is ≥ 20 mmol/L, there is a primary metabolic acidosis regardless of p. H or serum bicarbonate concentration

Delta Ratio • Delta ratio = (change in anion gap) / (change in bicarbonate) • Delta ratio= Anion gap -12/ 24 - HCO 3 • Need of Ratio • Almost obliged to figure out whether those anions have been solely responsible for the acidosis, or whether another (non-anion-gap) cause is lurking in the background

Delta Ratio Delta ratio Interpretation <0. 4 • Hyperchloraemic acidosis 0. 4 -0. 8 • High Anion gap and Non anion gap Acidosis 0. 8 -2 • Anion Gap metabolic acidosis • Lactic Acidosis : Average value 1. 6 • DKA : Value is 1 due to urinary ketone loss • AG acidosis with pre-exiting metabolic Alkalosis >2

Case 1 • 3 year old boy previously normal thriving well has now presented with 5 days history of vomiting and drowsiness v 8 to 10 episodes daily v Non bilious, Non projectile v Positive triage findings of RR 12/min with drowsiness and features of mild dehydration
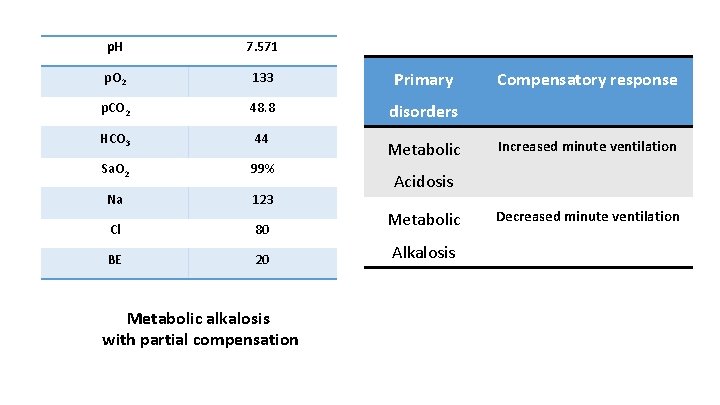
p. H 7. 571 p. O 2 133 Primary p. CO 2 48. 8 disorders HCO 3 44 Sa. O 2 99% Na 123 Cl 80 BE 20 Metabolic alkalosis with partial compensation Metabolic Compensatory response Increased minute ventilation Acidosis Metabolic Alkalosis Decreased minute ventilation
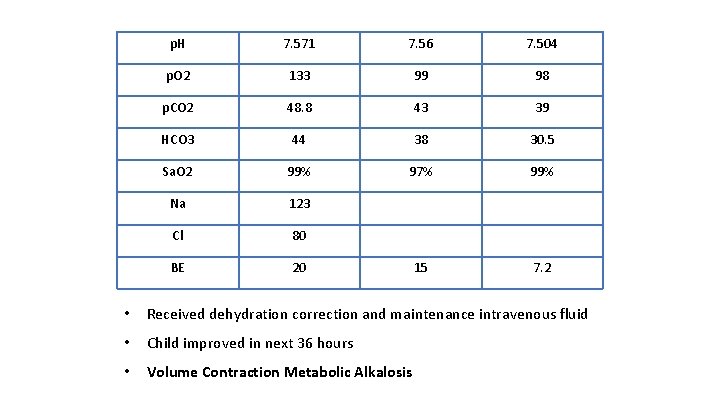
p. H 7. 571 7. 56 7. 504 p. O 2 133 99 98 p. CO 2 48. 8 43 39 HCO 3 44 38 30. 5 Sa. O 2 99% 97% 99% Na 123 Cl 80 BE 20 15 7. 2 • Received dehydration correction and maintenance intravenous fluid • Child improved in next 36 hours • Volume Contraction Metabolic Alkalosis
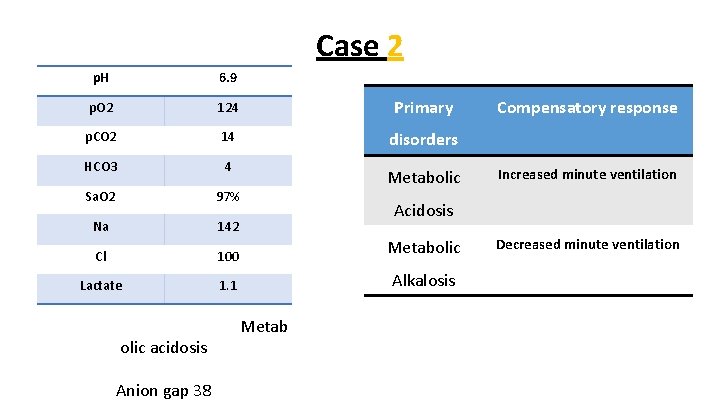
Case 2 p. H 6. 9 p. O 2 124 Primary p. CO 2 14 disorders HCO 3 4 Sa. O 2 97% Na 142 Cl 100 Metabolic Lactate 1. 1 Alkalosis olic acidosis Anion gap 38 Metabolic Compensatory response Increased minute ventilation Acidosis Metab Decreased minute ventilation
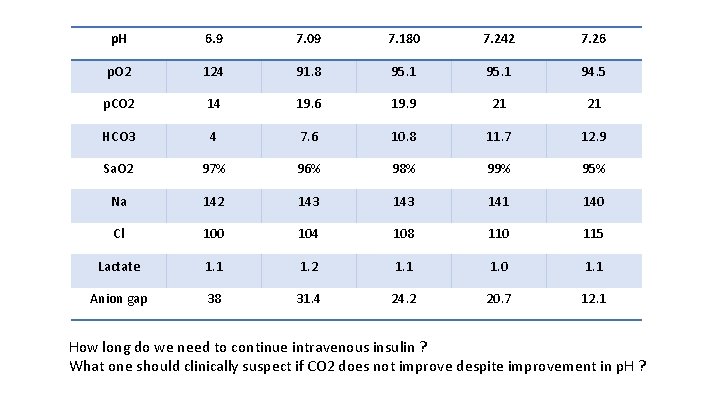
p. H 6. 9 7. 09 7. 180 7. 242 7. 26 p. O 2 124 91. 8 95. 1 94. 5 p. CO 2 14 19. 6 19. 9 21 21 HCO 3 4 7. 6 10. 8 11. 7 12. 9 Sa. O 2 97% 96% 98% 99% 95% Na 142 143 141 140 Cl 100 104 108 110 115 Lactate 1. 1 1. 2 1. 1 1. 0 1. 1 Anion gap 38 31. 4 24. 2 20. 7 12. 1 How long do we need to continue intravenous insulin ? What one should clinically suspect if CO 2 does not improve despite improvement in p. H ?
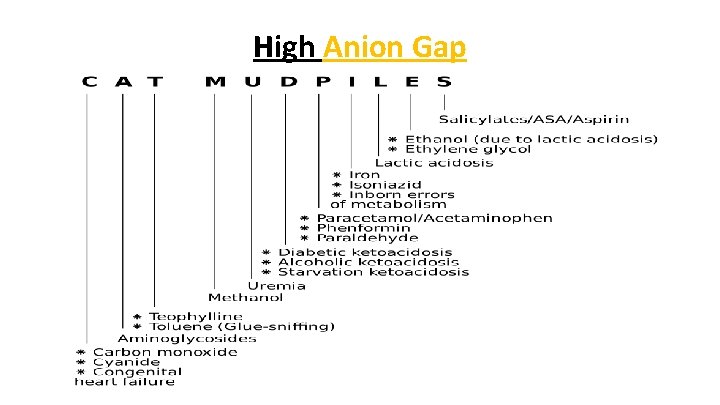
High Anion Gap

Decreased Anion Gap • Reduction in a major plasma protein such as albumin • Increase in unmeasured cations • Hyper-lipidemias
- Slides: 32