Acid Base Disorders Made Simple Mayur Rali MD

Acid Base Disorders Made Simple Mayur Rali, MD, FAAFP Parth Rali, MD

Disclaimer None

Learning Objectives • Be able to define and understand key concepts of Acid Base Disorders. • Be able to use a systematic approach to diagnose and differentiate various Acid Base Disorders. • Be able to apply a step to step approach in analysis of the result of ABG. • Be able to identify clinical significance and application in real life scenarios with management strategies. • Be able to solve practice cases emphasizing some of the crucial concepts in understanding Acid Base Disorders.

General Approach to Acid Base Disorders • Normal values of electrolytes are based on the reference values of individual labs. • Interpretation of acid base disorder should be done in conjunction with history and physical examinations, which provide clue. • The cases are formed for teaching purposes where primary disorder and their compensation would be too accurate than real life. • When interpreting the acid base disorder in real clinical setting, don’t get stuck to exact numbers whole goal is not to miss an obvious disorder.

General Approach to Acid Base Disorders • For discussion purposes, normal range for all parameters are: • • p. H 7. 35 -7. 45 HCO 3 - 18 -24 m. Eq/L PCO 2 35 -45 mm. Hg Normal AG is 10 -12 AG = 2. 5 x albumin Normal albumin range 4 -4. 5 g/dl

General Approach to Acid Base Disorders • There are three different approaches of quantifying acid-base disorders • • • Physiological approach Base-access approach Physio-chemical approach • We will use physiological approach to interpret all cases. • The best way to master the acid-base disorder is repetition. Form a habit of calculating acid-base disorder on each and every patient you encounter. (Berned et al, 2014)

General Approach to Acid Base Disorders • We will use following step-wise method to analyze and interpret all acid-base disorders. 1. Acidosis (p. H<7. 35) or Alkalosis (p. H>7. 35) 2. Primary disorder metabolic or respiratory—look at HCO 3 - and PCO 2 (base decision on your labs reference values) 3. If it is metabolic acidosis, does any AG exist? 4. Is there appropriate compensation for primary disorder? 5. If it is metabolic acidosis, does any other concomitant metabolic disturbances exist?

General Approach to Acid Base Disorders • Let’s apply this general approach and try to solve each case together. • Reminder: Lab values are lab values. History and physical examinations have the best clue for interpreting lab values in real world scenarios.

Case 1 19 y/o male with no PMHx is brought to the Emergency Department (ED) by parents with 3 days of weakness, lethargy, and progressive confusion. There is a family history of IDDM (insulin dependent diabetes mellitus on paternal side). As per parents, patient has noticed increased thirst, polyuria in last few days. He also has been having nausea, vomiting, and inability to hold food down for last 48 hours. On examination in ED, patient appeared extremely tachypneic and dehydrated.
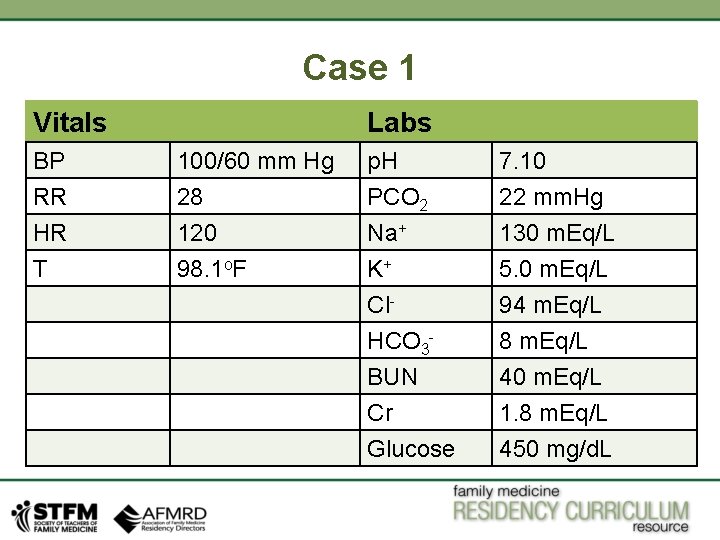
Case 1 Vitals Labs BP RR HR 100/60 mm Hg 28 120 p. H PCO 2 Na+ 7. 10 22 mm. Hg 130 m. Eq/L T 98. 1 o. F K+ Cl. HCO 3 BUN Cr Glucose 5. 0 m. Eq/L 94 m. Eq/L 8 m. Eq/L 40 m. Eq/L 1. 8 m. Eq/L 450 mg/d. L

Case 1 Which acid-base status most accurately describes the patient’s clinical condition? A. Primary Metabolic Acidosis and Non compensated Respiratory Alkalosis B. Metabolic Acidosis and Metabolic Alkalosis C. Metabolic Acidosis, Simultaneous Non Gap Acidosis, Respiratory Alkalosis D. Primary Metabolic Acidosis with compensated respiratory alkalosis

Case 1 Clinical Clues: Young male, confused, extremely tachypneic, strong history of insulin dependent diabetes in family. Even before you see lab values, you should be already thinking about Diabetic Ketoacidosis. Step 1: Is it acidosis or alkalosis? • p. H is 7. 10. It is less then 7. 35, so it is acidosis. Step 2: Is it Primary respiratory or Metabolic disorder? • PCO 2 is 22 mm. Hg. If PCO 2 is higher than normal (45 mm Hg) will give you respiratory acidosis. Here it is lower. So it’s not primary respiratory disorder. • On the other side, HCO 3 - is 8 m. Eq/L (normal range 18 -24 m. Eq/L). So we are dealing with metabolic acidosis.

Case 1 Step 3: Is there any AG, normal AG is 12 • • Formula for AG is: Na+- (Cl- + HCO 3 -), so 130 – (94 + 8) = 28. So AG in patient is 28, higher than normal AG. So this is Primary AG Metabolic Acidosis Step 4: Is there appropriate compensation for primary disorder? • • So now we need to identify whether respiratory compensation for metabolic disorder is appropriate or not ? Now we need to apply Winter’s formula for respiratory compensation for metabolic acidosis. Expected PCO 2 = 1. 5 (HCO 3 -) + 8 +/- 2, 1. 5 (8) + 8 +/- 2 = range of 18 -22 mm. Hg. Our PCO 2 is 22 mm. Hg. So it is compensated respiratory alkalosis. (PCO 2 less then normal) So far, we have Primary AG metabolic acidosis with compensated respiratory alkalosis.

Case 1 Step 5: If it is metabolic acidosis, does any other concomitant metabolic disturbances exist? • Delta AG: Calculated AG–Normal AG • Add Delta AG to your patients Bicarbonate Value • If after doing that calculated value comes within normal Bicarb range which is 18 -24 m. Eq/L, then it is pure AG metabolic acidosis without any other metabolic dissociation. • If after doing that, calculated value comes higher than normal Bicarb range which in these examples >24 m. Eq/L, then you have additional metabolic alkalosis on top of AG metabolic acidosis. • If after doing that, calculated value comes lower then normal Bicarb range which in these examples is <18 m. Eq/L, then you have AG metabolic acidosis and Non Gap metabolic acidosis (NGMA)

Case 1 Step 5: If it is metabolic acidosis, does any other concomitant metabolic disturbances exist? • Delta AG: 28– 12 = 16 • Add Delta AG to your patient’s Bicarb Value : 16 + 8 = 24 • • So in this case value falls in normal range for bicarb so it is pure AG metabolic acidosis. • For example, if this value would have been >24 then we would have called AG metabolic acidosis + Metabolic alkalosis • For example, if this value would have been <18 then we would have called AG metabolic acidosis + Non Gap Metabolic acidosis. So final acid base disorder we determined is Pure AG Metabolic Acidosis with appropriate respiratory alkalosis (compensated) Answer D.

Case 1: Answer Which acid-base status most accurately describes the patient’s clinical condition? A. Primary Metabolic Acidosis and Non compensated Respiratory Alkalosis B. Metabolic Acidosis and Metabolic Alkalosis C. Metabolic Acidosis, Simultaneous Non Gap Acidosis, Respiratory Alkalosis D. Primary Metabolic Acidosis with compensated respiratory alkalosis

Case 2 56 y/o male with PMHx of DM, HTN, COPD on 2 L/min of oxygen for last 2 years, current smoker with 30 pack per day history of smoking presents to ED with worsening SOB, increased sputum production from his baseline, and productive cough with yellow-green phlegm. He has been usually noncompliant with his inhalers. But in last 2 days, he has been using his albuterol inhaler six to eight times a day without significant relief. Today he was to a point where he feels suffocated and can’t breathe. On presentation to ED, patient was in severe respiratory distress using accessory muscles and wheezing (which he usually doesn’t have. ) Labs were drawn, and patient was placed on Bi. PAP.
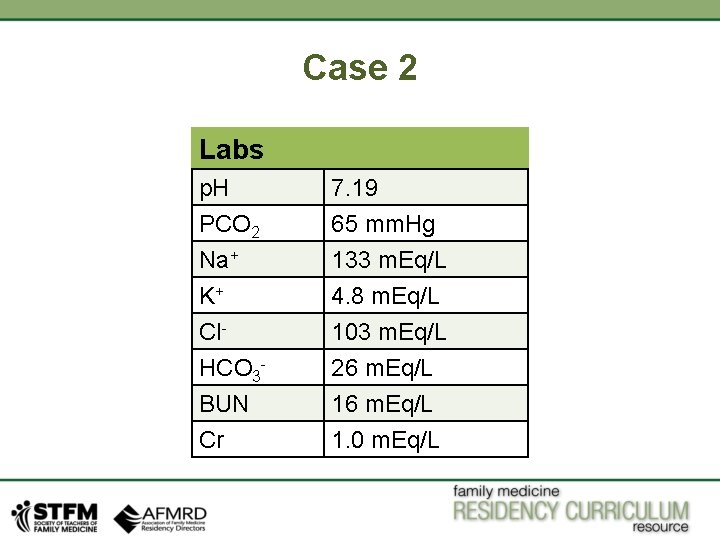
Case 2 Labs p. H PCO 2 Na+ K+ Cl. HCO 3 BUN Cr 7. 19 65 mm. Hg 133 m. Eq/L 4. 8 m. Eq/L 103 m. Eq/L 26 m. Eq/L 1. 0 m. Eq/L

Case 2 Which acid-base status most accurately describes the patient’s clinical condition? A. Primary Respiratory acidosis and Primary Metabolic alkalosis B. Acute Primary Respiratory Acidosis with Compensated Metabolic Alkalosis C. Primary Metabolic Acidosis and Primary Respiratory Alkalosis D. Acute on chronic respiratory acidosis.

Case 2 Clinical Clues: Middle aged male, who is on home oxygen and noncompliant with his medications and now he is in respiratory distress. Step 1: Is it acidosis or alkalosis? • p. H is 7. 19. It is less than 7. 35, so it is acidosis. Step 2: Is it Primary Respiratory or Metabolic disorder? • HCO 3 - is 26 m. Eq/L (normal range 18 -24 m. Eq/L), for acidosis of metabolic origin bicarb would be lower like previous case which is not the case here. • PCO 2 is 65 mm. Hg, which is higher than normal (35 -45 mm. Hg). So we are dealing with respiratory acidosis.

Case 2 Step 3: Is there any AG, normal AG is up to 12 • Formula for AG is, Na+- (Cl- + HCO 3 -), so 133 - (103 + 26) = 4 - So there is no anion gap. Step 4: Is there appropriate compensation for primary disorder? • • So now we need to identify weather metabolic compensation for metabolic disorder is appropriate or not ? For acute respiratory acidosis, bicarb would go up (body will try to counteract respiratory acidosis with metabolic alkalosis) by 1 m. Eq/L for every 10 mm. Hg rise in PCO 2 from baseline normal value. Here PCO 2 is 65 mm. Hg, 20 mm. Hg higher than normal upper limit ( 45 mm. Hg), so expected bicarb will go up by 2 m. Eq/L (for every 10 mm. Hg of PCO 2 for 1 m. Eq/L of HCO 3 -), making it 24+2 = 26 m. Eq/L, which equals to serum bicarb in this case. So this is compensated metabolic alkalosis.

Case 2 Step 5: If it is metabolic acidosis, do any other concomitant metabolic disturbances exist? • There is no metabolic acidosis in this case, so we don’t have to worry about step 5. • So final acid base disorder we determined is primary respiratory acidosis with compensated metabolic alkalosis Answer B

Case 2: Answer Which acid-base status most accurately describes the patient’s clinical condition? A. Primary respiratory acidosis and primary metabolic alkalosis B. Acute Primary Respiratory Acidosis with Compensated Metabolic Alkalosis C. Primary Metabolic Acidosis and Primary Respiratory Alkalosis D. Acute on chronic respiratory acidosis.

Let’s Pause and Summarize • In case of Primary Metabolic acidosis to see respiratory compensation, we need to use winters formula which is: • Expected PCO 2= 1. 5 (Bicarb) + 8 +/- 2 • We should remember Winter’s formula, if you can’t, one crude trick is to look at last digit of p. H, it should correlate to your PCO 2. If p. H is 7. 24, your expected PCO 2 should be around 24. • AG: Na+ - (Cl- + HCO 3 -), normal anion gap is up 12, presuming albumin level is normal (4 -4. 5 g/dl) • If there is AG Metabolic acidosis we have to make sure, there is not concomitant metabolic disorder.

Let’s Pause and Summarize • • For acute respiratory acidosis, p. H would drop by 0. 08 for every 10 rise in PCO 2 above normal, ie, If PCO 2 goes to 55 mm Hg, p. H would drop from 7. 35 to 7. 27 For chronic respiratory acidosis, p. H would drop by 0. 03 for every 10 rise in PCO 2 above normal. p. H can remain normal if kidneys have long time to compensate for chronic state. For acute respiratory acidosis, for every 10 mm. Hg rise in PCO 2 bicarb would go up 1 m. Eq/L. For chronic respiratory acidosis, for every 10 mm. Hg rise in PCO 2 bicarb would go up by 3 m. Eq/L.

Case 3 24 y/o male is brought to emergency department by girlfriend after ingestion of 40 pills from his grandmother’s pill box in a suicidal attempt after a dispute with his girlfriend. He started feeling dizziness, abdominal pain after ingestion of pills and called his girlfriend who brought him to ED. As far as girlfriend can recall, grandmother takes this pill to keep her blood thin to prevent heart attack. Patient appears ill with tachypnea and altered mental status.
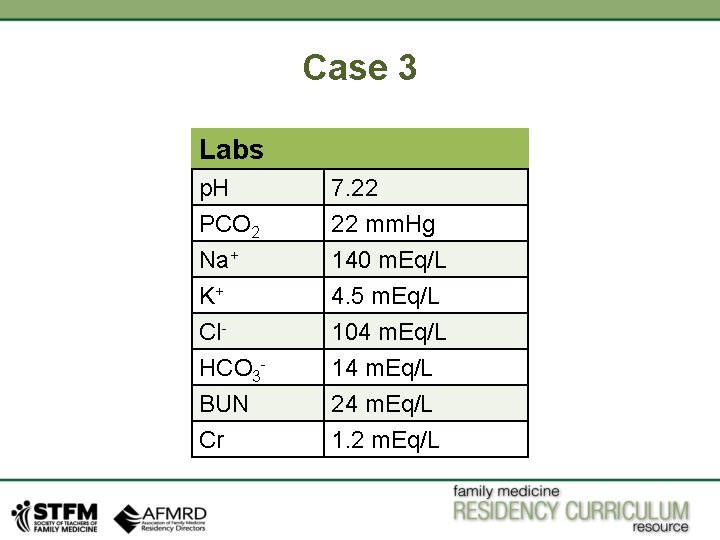
Case 3 Labs p. H PCO 2 Na+ K+ Cl. HCO 3 BUN Cr 7. 22 22 mm. Hg 140 m. Eq/L 4. 5 m. Eq/L 104 m. Eq/L 14 m. Eq/L 24 m. Eq/L 1. 2 m. Eq/L

Case 3 Which acid-base status most accurately describes the patient’s clinical condition? A. Anion Gap Metabolic acidosis, Non anion gap Metabolic Acidosis and Respiratory Alkalosis B. Non Anion gap Metabolic Acidosis and Respiratory Alkalosis C. Primary anion gap Metabolic Acidosis and Primary Respiratory Alkalosis D. Primary anion gap Metabolic Acidosis with compensated Respiratory alkalosis

Case 3 Clinical Clues: Young male, suicidal attempt, and took grandmother’s blood thinner pills. Likely aspirin overdose. Step 1: Is it acidosis or alkalosis? • p. H is 7. 22. It is less then 7. 35, so it is acidosis. Step 2: Is it Primary respiratory or Metabolic disorder? • PCO 2 is 22 mm Hg If PCO 2 is higher than normal (45 mm Hg) will give you respiratory acidosis. Here it is lower. So acidosis is not caused by respiratory disturbance. • On the other side, HCO 3 - is 14 m. Eq/L (normal range 1824 m. Eq/L). So we are dealing with primary metabolic acidosis.

Case 3 Step 3: Is there any AG, normal AG is 12 • • Formula for AG is, Na+- (Cl- + HCO 3 -), so 140 – (104 + 14) = 22. So AG in patient is 22, higher than normal AG. So this is Primary AG Metabolic Acidosis Step 4: Is there appropriate compensation for primary disorder? • • So now we need to identify whether respiratory compensation for metabolic disorder is appropriate or not? Now we need to apply Winter’s formula for respiratory compensation for metabolic acidosis. Expected PCO 2 = 1. 5 (HCO 3 -) + 8 +/- 2, 1. 5 (14) + 8 +/- 2 = range of 27 -31 mm. Hg. Our PCO 2 is 22 mm. Hg, so it is not compensated, it is even lower than compensated value. So it is another primary disorder. So we have Primary respiratory alkalosis So far, we have primary AG metabolic acidosis and primary respiratory alkalosis.

Case 3 Step 5: If it is metabolic acidosis, does any other concomitant metabolic disturbances exist? • Delta AG: 22– 12 = 10 • Add Delta AG to your patient’s Bicarb Value: 14 + 10 = 24 • So in this case value falls in normal range for bicarb so it is pure AG metabolic acidosis. • So final acid-base disorder we determined is Pure AG Metabolic Acidosis and Primary Respiratory Alkalosis (compensated) Answer C.

Case 3: Answer Which acid-base status most accurately describes the patient’s clinical condition? A. Anion Gap Metabolic acidosis, Non anion gap Metabolic Acidosis and Respiratory Alkalosis B. Non Anion gap Metabolic Acidosis and Respiratory Alkalosis C. Primary anion gap Metabolic Acidosis and Primary Respiratory Alkalosis D. Primary anion gap Metabolic Acidosis with compensated Respiratory alkalosis

Case 4 24 y/o female presents to ED with light headedness and feeling weak. She is trying to become a model and trying to lose weight. On physical examination, she has poor dentition and scratch marks on the back of hand. Labs p. H PCO 2 Na+ K+ Cl. HCO 3 BUN Cr 7. 53 50 mm. Hg 123 m. Eq/L 3. 2 m. Eq/L 73 m. Eq/L 40 m. Eq/L 1. 0 m. Eq/L

Case 4 Which acid-base status most accurately describes the patient’s clinical condition? A. Respiratory Acidosis with compensated Metabolic Alkalosis B. Metabolic Alkalosis with compensated Respiratory Acidosis C. Primary Metabolic Alkalosis and Primary Respiratory Acidosis D. Metabolic acidosis, Metabolic Alkalosis and Respiratory Acidosis

Case 4 Clinical Clues: Young female trying to become model, signs of hypovolemia and self induced vomiting. Likely case of vomiting induced metabolic alkalosis. Step 1: Is it acidosis or alkalosis? • p. H is 7. 53. It is more then 7. 35, so it is alkalosis. Step 2: Is it Primary respiratory or Metabolic disorder? • PCO 2 is 50 mm. Hg. It is higher than normal (45 mm Hg). So it is not respiratory issue causing patient to be alkalotic. • On the other side, HCO 3 - is 40 m. Eq/L (normal range 1824 m. Eq/L). So we are dealing with metabolic alkalosis.

Case 4 Step 3: Is there any AG? Normal AG is 12 • • Formula for AG is, Na+- (Cl- + HCO 3 -), so 123 – (73 + 40) = 10. So there is no anion gap. Step 4: Is there appropriate compensation for primary disorder? • • So now we need to identify weather respiratory compensation for metabolic disorder is appropriate or not ? Now we need to apply formula as below, Expected PCO 2 = 0. 7 (HCO 3 -) + 21 +/- 1. 5, 0. 7 (40) + 21 +/- 1. 5 = range of 47. 5 - 50. 5 mm. Hg. Our PCO 2 is 50 mm. Hg. So it is compensated respiratory acidosis. ( PCO 2 more than normal) So far, we have Primary Metabolic Alkalosis with compensated respiratory acidosis.

Case 4 Step 5: There is no metabolic acidosis, so step 5 doesn’t apply. • So final acid base disorder we determined is Primary Metabolic Alkalosis and compensated Respiratory Acidosis Answer B.

Case 4 Which acid-base status most accurately describes the patient’s clinical condition? A. Respiratory Acidosis with compensated Metabolic Alkalosis B. Metabolic Alkalosis with compensated Respiratory Acidosis C. Primary Metabolic Alkalosis and Primary Respiratory Acidosis D. Metabolic acidosis, Metabolic Alkalosis and Respiratory Acidosis

Acid-Base Disorders

Metabolic Acidosis Definition: • Metabolic acidosis is defined as p. H < 7. 38 and Bicarbonate concentration less than 22 mmol per liter. Clinical Patho physiology: • Metabolic acidosis is in general caused by either excess production of H+ or loss of HCO 3 - • Metabolic acidosis is divided into • Anion Gap (AG) is defined by difference between measured cations minus measured anions. (AG = Na+ - (CL-+ HCO 3 -)). • AG is usually 2. 5 x albumin, which is around 3. 5 -4 d/d. L, producing normal AG around 10 -12 Anion Gap Metabolic Acidosis or Non Anion Gap Metabolic Acidosis. Pocket Medicine, Massachusetts General Hospital, Handbook of Internal Medicine

Metabolic Acidosis (AG) Elevated AG Metabolic Acidosis: Causes Mnemonic (MUDPIES) Methanol poisoning Uremia Diabetic ketoacidosis, Drugs Paraldehyde Ischemia-Lactic Acidosis* Ethylene glycol (antifreeze) Salicylate, Acetaminophen.

Metabolic Acidosis (AG) * Lactic acidosis has been further classified into Type A and Type B lactic acidosis. • Type A lactic acidosis is secondary to tissue hypo perfusion or impairment in tissue oxygenation. Examples include septic shock, respiratory failure, circulatory shock —hemorrhagic or cardiogenic, ischemic bowel. • Type B lactic acidosis is not secondary to tissue hypo perfusion but likely originating from tissue metabolism and toxins. Examples include malignancy, alcoholism, meds like metformin, propelyne glycol, HIV meds like NRTIs.

Metabolic Acidosis (AG) • Osmolar Gap: In work up of AG metabolic acidosis, Osmolar gap should always be calculated particularly when there is high likelihood of toxic ingestion. • • • OG >10 is considered elevated. Ethanol, Methanol, Ethylene Gylcol, propyelene ingestion will have elevated OG and AG. Isopropyl Alcohol will have elevated OG but normal AG.

Metabolic Acidosis Non AG metabolic acidosis: Causes • Renal tubular acidosis (RTAs) • Hyperchloremic metabolic acidosis (from iatrogenic infusion of large volume of isotonic fluids) • Gastro-intestinal loss of bicarbonate containing fluids, eg, diarrhea • Toluene, acetazolamide ingestion • Early renal failure

Metabolic Acidosis General Management: • • • Treatment of underlying cause is the cornerstone in terms of management. Role of IV bicarbonate remains extremely controversial in management of metabolic acidosis particularly secondary to lactic acidosis Urgent Renal Replacement therapy in terms of Continuous Renal Replacement therapy (CRRT) or Hemodialysis in refractory cases of Metabolic acidosis.

Metabolic Alkalosis Definition: • Metabolic alkalosis has been defined as elevation of p. H >7. 38 with rise of bicarbonate value in excess of normal limits. Clinical Pathophysiology: • To generate a state of metabolic alkalosis, loss of acid or addition of alkali and enhanced renal reabsorption of bicarbonate must be present. • Metabolic alkalosis can be categorized in to saline responsive (also classified as metabolic alkalosis associated with volume contraction) or saline resistant (also classified as metabolic alkalosis associated with volume expansion or euvolemia)

Metabolic Acidosis Saline responsive metabolic alkalosis (Urine chloride <20): Causes • • Vomiting, NG suction Diuretic Therapy Post hypercapnia Cystic Fibrosis Saline resistant metabolic alkalosis (Urine chloride >10): Causes • • Mineralocorticoid access: i. Primary aldosteronism ii. Secondary aldosteronism- CHF, Cirrhosis, malignant HTN Exogenous Alkali–licorice, high amount infusion of sodium bicarbonate containing fluids Barter’s and Gitelman’s syndrome. Severe Hypokalemia

Metabolic Alkalosis General Management: • • • In saline responsive group, if hypovolemia replenishment of isotonic fluids is sufficient. If diuretics are the offending cause, then discontinue the diuretics. If isotonic fluid administration can’t be given because of cardio-pulmonary issues then treatment with KCL, acetazolamide is appropriate. If nasogastric suction can’t be discontinued then patient should be placed on PPI to decrease H+ secretion and loss via nasogastric tube suction.

Respiratory Acidosis Definition: • Respiratory acidosis occurs either secondary to excess CO 2 production or difficulty in excretion Acute respiratory acidosis: Causes • Depression of central nervous system, drugs (narcotics, benzodiazepines)* • Acute exacerbation of COPD (chronic obstructive pulmonary disease), asthma or pneumonia, pulmonary embolism$ Chronic respiratory acidosis: Causes • Myasthenia gravis* • Muscular dystrophy* • Gillian Barre Syndrome* • COPD$ • Kyphoscoliosis*

Respiratory Acidosis General Management: • On top of treating underlying cause, Bi. PAP support to hyperventilate might be useful. • If patient is not tolerating the Bi. PAP or failing, should proceed with intubation and mechanical ventilation in severe cases.

Respiratory Alkalosis Definition: • Respiratory alkalosis occurs as a result of reduction in PCO 2 due to increased ventilation relative to production. Acute respiratory alkalosis: Causes • Pain, fever, panic attacks. Stroke, meningitis, salicylate toxicity* • Early stages of pneumonia, pulmonary embolism, sepsis, congestive heart failure, pulmonary edema$ Chronic respiratory alkalosis: Causes • Hyperthyroidism, pregnancy, liver failure

Let’s Do Posttest Questions and Try to Diagnose Acid-Base Disorder

Post Test Quiz 1 Q: 28 y/o female with suicidal attempt, found comatose after ingesting radiator fluid. Which acid-base status most accurately describes patient clinical condition? Labs p. H 6. 90 PCO 2 18 mm. Hg Na+ 135 m. Eq/L K+ 7. 0 m. Eq/L Cl- 98 m. Eq/L HCO 3 - 6 m. Eq/L BUN 20 m. Eq/L Cr 5 m. Eq/L Glucose 135 mg/d. L

Answer: Metabolic Acidosis secondary to ethylene glycol toxicity. Patient has acidosis as p. H is 6. 90. HCO 3 - is low so is PCO 2, so it is primary metabolic acidosis. AG is 31. Compensated PCO 2 using Winter’s formula would be 1. 5(6) + 8 +/- 2, would be between 15 -19 mm Hg, here it is 18 mm Hg. So the respiratory compensations is appropriate. Delta gap is 19, adding that to serum bicarb of 6 would come up to 25. So this is pure AG metabolic acidosis with appropriate respiratory compensation. Patient ingested a radiator fluid, which has ethylene glycol in it, which explains patient current metabolic state. This patient has osmolar gap, renal failure which further confirms ethylene glycol. If urine is shinned, oxalate crystals might be seen, and ethylene glycol levels would be high in serum if checked. Patient would need an urgent hemodialysis in terms of management.

Post Test Quiz 2 Q: 54 y/o female with gallstone pancreatitis going on for 3 -4 days with severe vomiting. Which acid-base status most accurately describes patient clinical condition? Labs p. H 7. 54 PCO 2 46 mm. Hg Na+ 132 m. Eq/L K+ 2. 1 m. Eq/L Cl- 88 m. Eq/L HCO 3 - 36 m. Eq/L BUN 26 m. Eq/L Cr 0. 4 m. Eq/L Glucose 120 mg/d. L

Answer: Metabolic Alkalosis. Patient has alkalosis as p. H is 7. 54. HCO 3 - is elevated, so is PCO 2, so it is primary metabolic alkalosis. Compensated PCO 2 should be 0. 7 (HCO 3 -) + 21 +/- 1. 5 which would be 0. 7(36) + 21 +/- 1. 5 45 -47 mm. Hg, so it appropriately compensated. Patient has gallstone pancreatitis and she has been vomiting, and losing chloride containing fluid, and getting dehydrated. In order to compensate for loss of volume, body will try to retain Na+, which will be reabsorbed with HCO 3 - by kidneys giving increase in bicarbonate to give a state of metabolic alkalosis.

Post Test Quiz 3 Q: 38 y/o male with asthma exacerbation, feeling dyspneic for 1– 2 days. Which acid-base status most accurately describes patient clinical condition? Labs p. H 7. 57 PCO 2 21 mm. Hg Na+ 134 m. Eq/L K+ 3. 4 m. Eq/L Cl- 107 m. Eq/L HCO 3 - 20 m. Eq/L BUN 20 m. Eq/L Cr 0. 4 m. Eq/L Glucose 100 mg/d. L

Answer: Respiratory Alkalosis Patient has alkalosis as p. H is 7. 56. HCO 3 - is lower normal and PCO 2 is low, so it is primary respiratory alkalosis. Patient has asthma exacerbation, and he is feeling short of breath for last 3 -4 days, needless to say he has increased A-a gradient secondary to his respiratory issues. For acute respiratory changes, p. H will change by 0. 08 for every 10 change in PCO 2. His p. H is approximately 0. 12 points above normal (7. 45), so his PCO 2 would drop by approximately 15 mm Hg which would be around 20 mm Hg (Patient’s PCO 2 is 21 mm Hg). For acute respiratory alkalosis, for every 10 mm Hg drop in PCO 2, HCO 3 would drop by 2 meq/L. Patient’s Bicarb is 20 meq/l, which is near lower limit of normal, so it is appropriately compensated. So, in summary this patient has increase A-a gradient from his asthma exacerbation and he is trying to compensate by hyperventilating and creating a state of respiratory alkalosis.

Post Test Quiz 4 Q: 58 y/o male s/p hip replacement today, now in recovery room found lethargic. Which acid-base status most accurately describes patient clinical condition? Labs p. H 7. 23 PCO 2 60 mm. Hg Na+ 135 m. Eq/L K+ 3. 5 m. Eq/L Cl- 107 m. Eq/L HCO 3 - 20 m. Eq/L BUN 21 m. Eq/L Cr 0. 9 m. Eq/L Glucose 100 mg/d. L

Answer: Respiratory Acidosis Patient has acidosis as p. H is 7. 23. HCO 3 - is lower normal and PCO 2 is elevated so it is primary respiratory acidosis. Patient is in recovery room, possibly recently extubated post op from his hip surgery. It is very common situation where patients will still have effect of sedation. Patient PCO 2 is 60 which suggests patient has decreased minute ventilation creating a state of retaining PCO 2 and creating a state of respiratory acidosis. As mentioned earlier for acute respiratory changes, p. H will change by 0. 08 for every 10 change in PCO 2. So here p. H is 0. 12 points lower, so PCO 2 would be 15 mm Hg higher then normal limits, which would be around 60 mm Hg( upper limit of normal PCO 2 would be 45 mm Hg). Patient’s PCO 2 is exactly 60 mm Hg. For acute respiratory acidosis, for every 10 increase in PCO 2, HCO 3 - will drop by 1 meq/L. Here bicarb is appropriately lowered. Metabolic compensation usually takes time in primary respiratory disorders. Giving patient small dose of naloxone if opioids were involved in anesthesia, or placing patient on Bi. PAP would be appropriate approach in this patient.

Take Home Quiz Please see your faculty for answers to these questions with explanation.
- Slides: 61