ACID BASE DISORDERS Asad Merchant MD FRCPC Nephrology

ACID BASE DISORDERS Asad Merchant MD FRCP(C) Nephrology

Objectives • Analyze acid base disturbances in an algorithmic manner • Develop a differential diagnosis for a given Acid-base disorder • Appreciate that acid base abnormalities can be complex and it is important to be systematic

Case 1 - ER • 36 y M found passed outside a convenience store • History of HTN – no meds • Obese, no history of DM • works at the local power plant


Brought to the ER

O/E • • GCS - 6 120/70 37. 5 C P = 79 RR 24 BS – 8 O 2 Sat 94% on RA
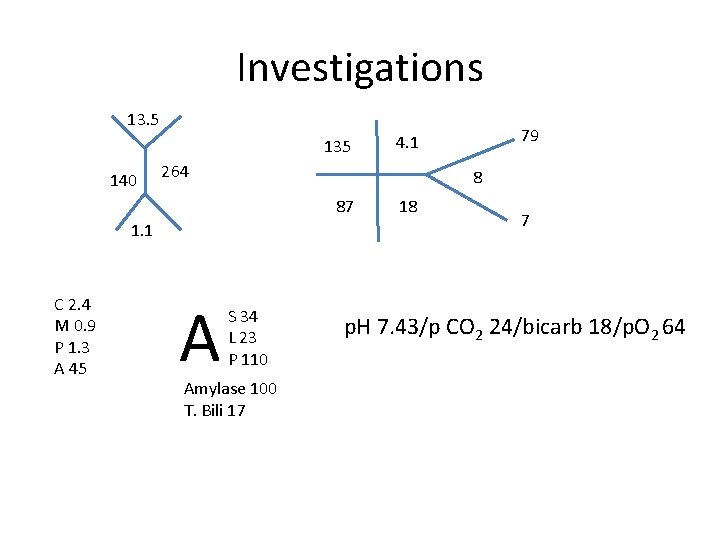
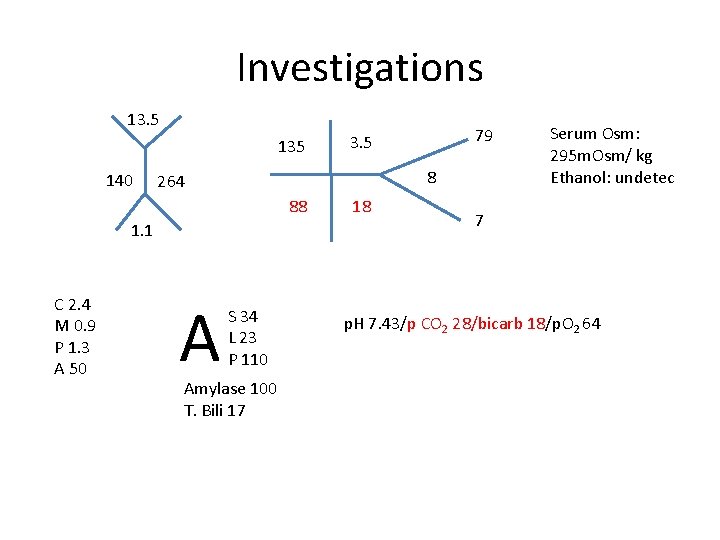
Investigations 13. 5 135 140 264 8 87 1. 1 C 2. 4 M 0. 9 P 1. 3 A 45 A 79 4. 1 S 34 L 23 P 110 Amylase 100 T. Bili 17 18 7 p. H 7. 43/p CO 2 24/bicarb 18/p. O 2 64
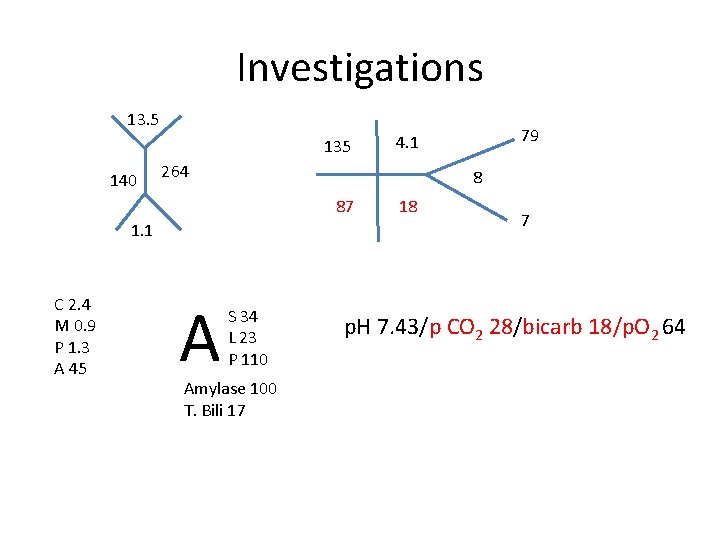
Investigations 13. 5 135 140 264 8 87 1. 1 C 2. 4 M 0. 9 P 1. 3 A 45 A 79 4. 1 S 34 L 23 P 110 Amylase 100 T. Bili 17 18 7 p. H 7. 43/p CO 2 28/bicarb 18/p. O 2 64

What’s the acid base abnormality? • Δp. Co 2 = 40 – 28 = 12 • Δ bicarb = 24 – 18 = 6 • Respiratory Alkalosis • Compensation?
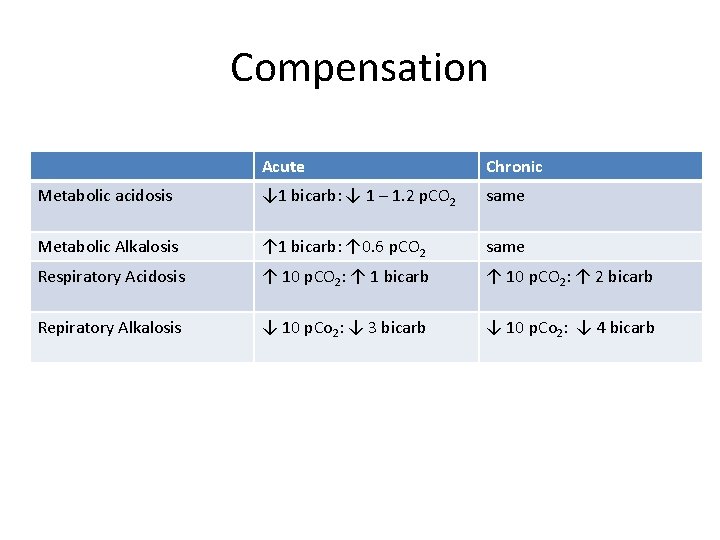
Compensation Acute Chronic Metabolic acidosis ↓ 1 bicarb: ↓ 1 – 1. 2 p. CO 2 same Metabolic Alkalosis ↑ 1 bicarb: ↑ 0. 6 p. CO 2 same Respiratory Acidosis ↑ 10 p. CO 2: ↑ 1 bicarb ↑ 10 p. CO 2: ↑ 2 bicarb Repiratory Alkalosis ↓ 10 p. Co 2: ↓ 3 bicarb ↓ 10 p. Co 2: ↓ 4 bicarb
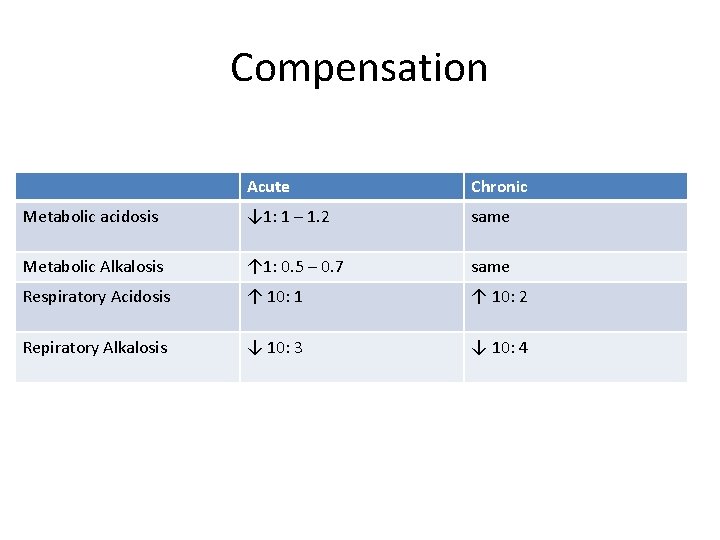
Compensation Acute Chronic Metabolic acidosis ↓ 1: 1 – 1. 2 same Metabolic Alkalosis ↑ 1: 0. 5 – 0. 7 same Respiratory Acidosis ↑ 10: 1 ↑ 10: 2 Repiratory Alkalosis ↓ 10: 3 ↓ 10: 4

What’s the acid base abnormality? • Δp. Co 2 = 40 – 28 = 12 • Δ bicarb = 24 – 18 = 6 • Δp. Co 2: Δ bicarb = 10: 5 • Respiratory Alkalosis • Appropriate Chronic Metabolic compensation • Should we stop here?

Anion Gap • How to calculate = Na – Cl – bicarb = 135 – 87 – 18 = 30 Interpretation: He has 18 unaccounted for anions What are the rest? – Albumin Note – For every 10 g/L decrease in albumin from 40, anion gap increases by 2. 5

• Anion Gap Metabolic Acidosis! • Should we stop here? • What else?

Important take home point Abnormal Anion Gap = Anion Gap Metabolic Acidosis
• Do the numbers add up?

DELTA/ DELTA (Δ/ Δ) • The ratio of unaccounted anions: decrease in bicarb • Δanion gap: Δ bicarb (Δ/ Δ) • Δ/ Δ = 18/ 6 = 3 • Interpretation: He has 3 x more anions than you would expect Or He lost 1/3 as many bicarbs as he should have given the number of extra anions AKA: He has too much bicarb

• Metabolic Alkalosis!!

Lets recap • Respiratory alkalosis • AG metabolic acidosis! • Metabolic Alkalosis!! • So what happenned?

Differential Diagnosis • Many different ways to approach it – Best way may be to approach the AGMA – finite list of possible differentials – MUDPILES/ KARMEL/ GOLDMARK

AGMA • • M U D P I L E S

AGMA • • • M - Methanol U - uremia D – Diabetic Ketoacidosis P – Phenformin/ metformin I - Isoniazid L - Lactate E – Ethylene Glycol S – Salicylates (Tylenol can cause AGMA via metabolism to oxoproline/ pyroglutamic acid)

Lactate acidosis – L- Lactate • • hypoperfusion (shock) Metformin Thiamine deficiency (malnutrition or alcoholism) Mitochondrial abnormality e. g. Cyanide poisoning or MELAS D- Lactate – not measured by labs routinely • From bacterial overgrowth

• Ketoacidosis – Diabetic ketoacidosis – insulin deficiency – Inability of acetyl coenzyme A to enter the Krebs cycle – Ketones such as beta hyroxybutyrate and acetoacetic acid produced – May be due to starvation (often milder)

• Toxic alcohols – Methanol – primarily neurotoxicity and blindness – Ethylene glycol – neurotoxicity and renal failure • Metabolized to oxalate – look for oxalate crystals
Investigations 13. 5 135 140 8 264 88 1. 1 C 2. 4 M 0. 9 P 1. 3 A 50 A 79 3. 5 S 34 L 23 P 110 Amylase 100 T. Bili 17 18 Serum Osm: 295 m. Osm/ kg Ethanol: undetec 7 p. H 7. 43/p CO 2 28/bicarb 18/p. O 2 64

Osmolal Gap • Calculated Osmolality: 2 x Na + BS + BUN (Urea) – Two salts and a sugar bun • 135 x 2 + 8 + 7 = 285 • Osmolal gap = Serum Osm (measured) – calc Osm – 295 – 285 = 10 (N 10 – 15)
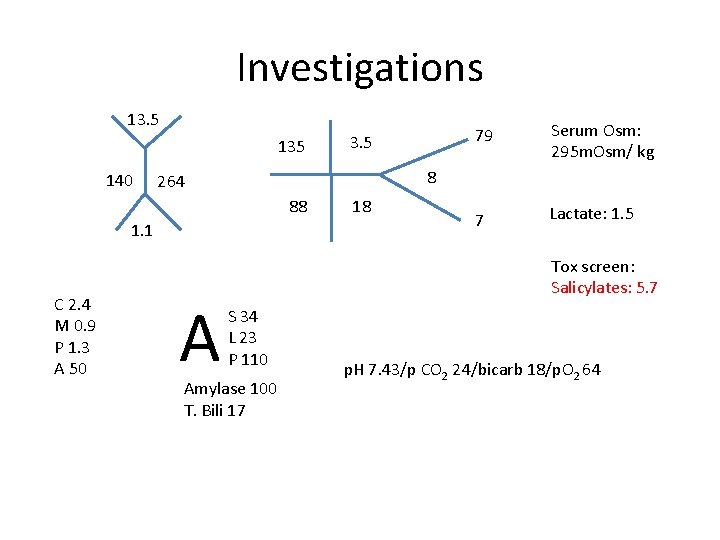
Investigations 13. 5 135 140 1. 1 A Serum Osm: 295 m. Osm/ kg 7 Lactate: 1. 5 8 264 88 C 2. 4 M 0. 9 P 1. 3 A 50 3. 5 79 18 Tox screen: Salicylates: 5. 7 S 34 L 23 P 110 Amylase 100 T. Bili 17 p. H 7. 43/p CO 2 24/bicarb 18/p. O 2 64

Diagnosis • ASA overdose – Respiratory Alkalosis – Hyperventilation – AG metabolic acidosis – salicylic acid – Metabolic alkalosis – GI symptoms – nausea vomiting

The plot thickens • RR – 24 – 22 – 18 – 15 • Next ABG = 7. 15/60/18/54 • Respiratory Acidosis • BP 80/60, T = 38. 5 • Need to call ICU and Nephrology for dialysis

Algorithm for Acid Base Analysis • Step 1 – Dominant acid base abnormality – Δp. CO 2 and Δ bicarb • • Step 2 – Compensation? – acute vs chronic Step 3 – Anion gap calculation Step 4 – If AGMA? Delta/ delta (Δ/ Δ) Step 5 – Cause? Generate differential based on specific acid base abnormality

So what happenned to H S • Viral infection – nausea + vomiting • +++ Aspirin (favourite hangover cure) • Severely ill at work – vomited x 3 – Left early to go to the kwiki mart for more aspirin – Vomited in the washroom – Passed out as he left

Case 2 – General Surgery Ward • 57 y male previously healthy • Diagnosed with colon cancer – has hemicolectomy with colostomy – OR uneventful • 1 L/ day stool output – given immodium

• Day 4 – Fever • Abdo CT – intrabdominal abscess, drain placed • Blood Cultures - ESBL bacteremia – also cipro resistant • Treated with tobramycin
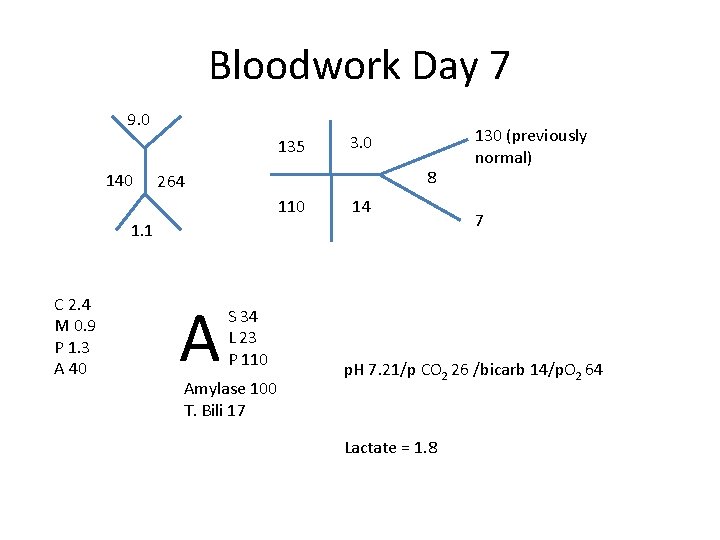
Bloodwork Day 7 9. 0 135 140 3. 0 8 264 110 14 1. 1 C 2. 4 M 0. 9 P 1. 3 A 40 A S 34 L 23 P 110 Amylase 100 T. Bili 17 130 (previously normal) 7 p. H 7. 21/p CO 2 26 /bicarb 14/p. O 2 64 Lactate = 1. 8

What’s the acid base abnormality? • Acidosis p. H = 7. 21 • Δ bicarb = 24 – 14 = 10 • Δp. Co 2 = 40 – 28 = 12 • Metabolic Acidosis • Compensation?
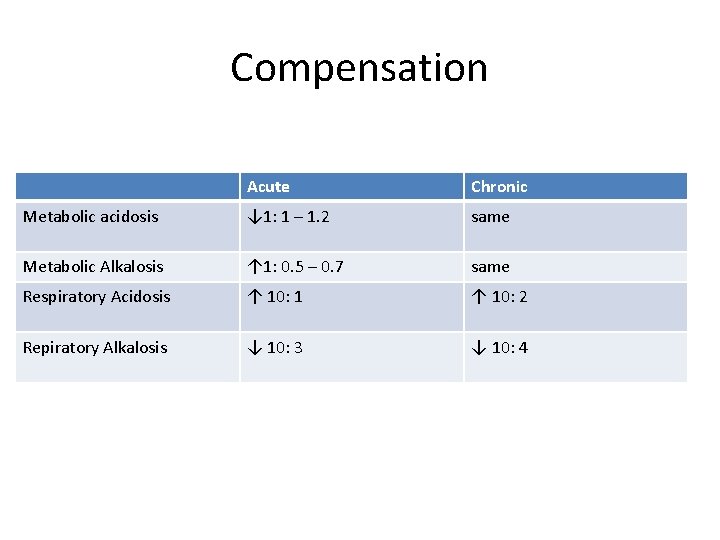
Compensation Acute Chronic Metabolic acidosis ↓ 1: 1 – 1. 2 same Metabolic Alkalosis ↑ 1: 0. 5 – 0. 7 same Respiratory Acidosis ↑ 10: 1 ↑ 10: 2 Repiratory Alkalosis ↓ 10: 3 ↓ 10: 4

Diagnosis • Metabolic acidosis • Anion Gap? 135 – 110 – 14 = 11 • Non Anion Gap Metabolic Acidosis

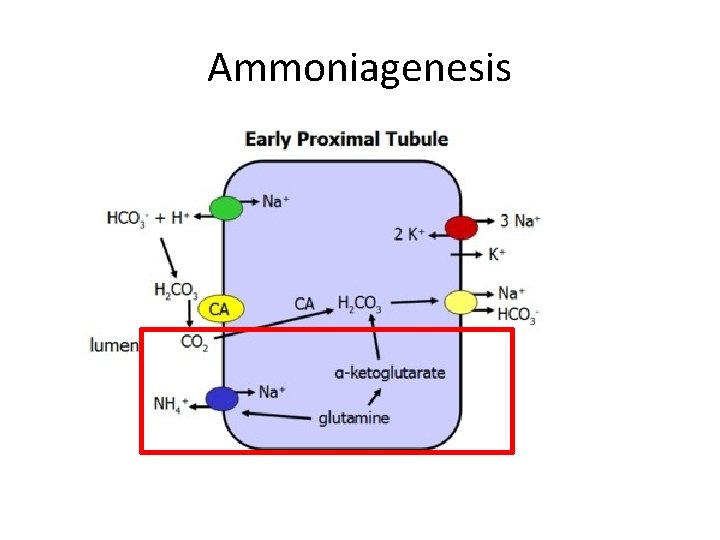
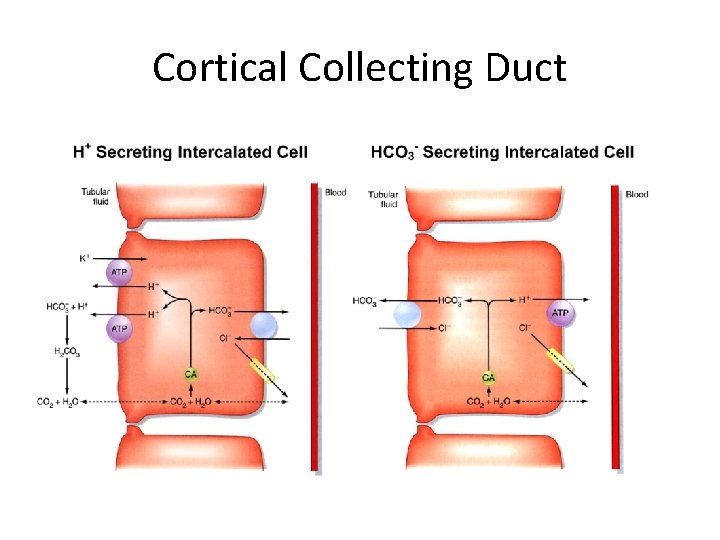
Renal bicarbonate Physiology • Most of bicarbonate handling by the kidneys is at the proximal convoluted tubule and the cortical collecting duct • The kidney is able to titrate acid in the lumen of the tubules with phosphate ions • The kidney is able to generate new bicarbonate via a process known as ammoniagenesis


• Factors that increase bicarbonate absorption – Urine flow – Na+ – Hypokalemia – Angiotensin II – Endothelin – p. CO 2 and bicarbonate concentrations

Renal vs GI • If there is no renal pathology – Kidney will increase ammonium (NH 4+) production • Urine anion gap (UAG) – Na + K – Cl = extra urinary anions • Positive Anion gap means – extra HCO 3 - in the urine • Negative Anion gap means – extra NH 4+ in the urine • Therefore positive UAG = RTA • Ne. GUTive UAG = Gut losses

Urine Osmolal Gap • We can calculate how much Ammonium there is in urine by the Urine Osmolal gap • NH 4+ = [Urine osm – (2[Na+K] + Urea)]/2 • Can distinguish between true RTA and toluene poisoning – both will have +ve urine anion gap

Case 2 - continued • Urine chemistry – Urine p. H = 5. 0 – Na+ = 50 – K + = 40 – Cl - = 40 • Urine Anion Gap = UNa + UK – Ucl = 50 (positive) • Renal cause!

Non Anion Gap Metabolic Acidosis • Loss of bicarbonate due, or addition of HCl – NS infusion – Diarrhea – Renal tubular acidosis – Post hypercapnea
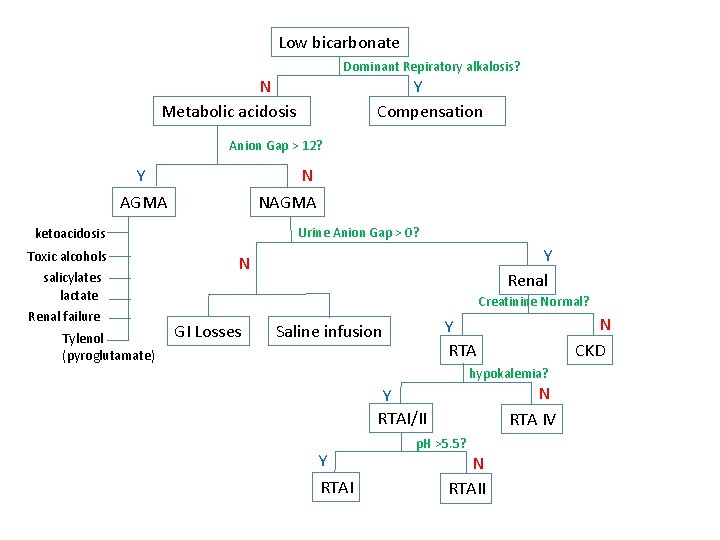
Low bicarbonate Dominant Repiratory alkalosis? N Metabolic acidosis Y Compensation Anion Gap > 12? N NAGMA Y AGMA Urine Anion Gap > 0? ketoacidosis Toxic alcohols salicylates lactate Renal failure Tylenol (pyroglutamate) Y Renal N Creatinine Normal? GI Losses N CKD Y RTA Saline infusion hypokalemia? N RTA IV Y RTAI/II Y RTAI p. H >5. 5? N RTAII

Renal Tubular acidosis • RTA I – distal – H+ cannot be secreted • RTA II – Proximal – Bicarbonate cannot be reabsorbed above a threshold concentration • RTA IV – hyporeninemia – Decreased H+ and K+ excretion – aldosterone mediated

Distinguishing features of RTA’s

Distal RTA • Often associated with nephrocalcinosis and stones • Causes – Genetic – may be syndromic – associated with deafness, or part of a generalized tubulopathy – Autoimmiune diseases – Sjogren’s disease – Medullary sponge kidney – Thyroiditis – Hyperparathyroidism – Drugs – amphotericin B – Lithium – Ifosfamide – Tubulointerstitial diseases

Proximal RTA • Associated with Fanconi Syndrome – Hypophosphatemia, hypomagnesemia, glucosuria, amino aciduria • Causes – Multiple Myeloma – Genetic conditions – Wilson’s disease, Lowe’s disease, Dent Disease, tyrosinemia, hereditary fructose intolerance – Heavy metal poisoning – cadmium, mercury, copper – Drugs – tenofivir, ifosfamide, aminoglycosides, foscarnet, amphotericin B, expired tetracyclines

Type IV RTA • Modest non anion gap metabolic acidosis (NAGMA) • Associated with hypokalemia • Decreased urinary NH 4+ excretion • Hyporeninemia and hypoaldosteronism • Often associated with DMII • Treat with fludrocortisone
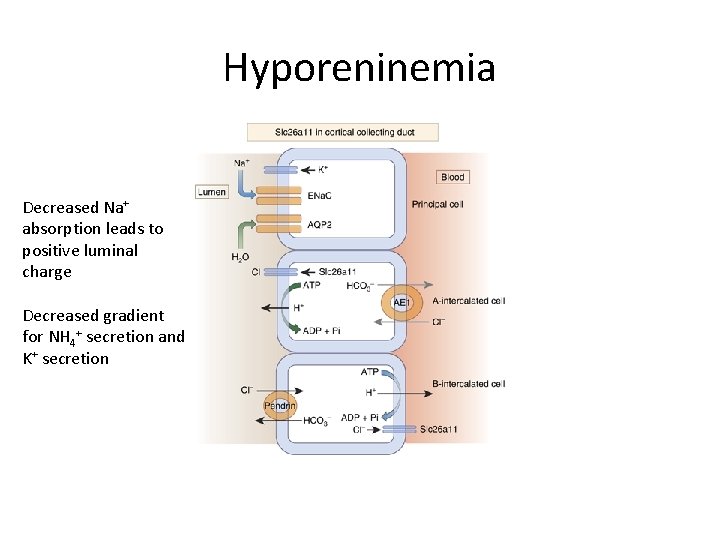
Hyporeninemia Decreased Na+ absorption leads to positive luminal charge Decreased gradient for NH 4+ secretion and K+ secretion

Case 2 • Switched to meropenem • Given Potassium supplementation, sodium bicarbonate intravenously • (Note important to replete potassium first) • Eventually acidosis resolved, and bicarbonate not required
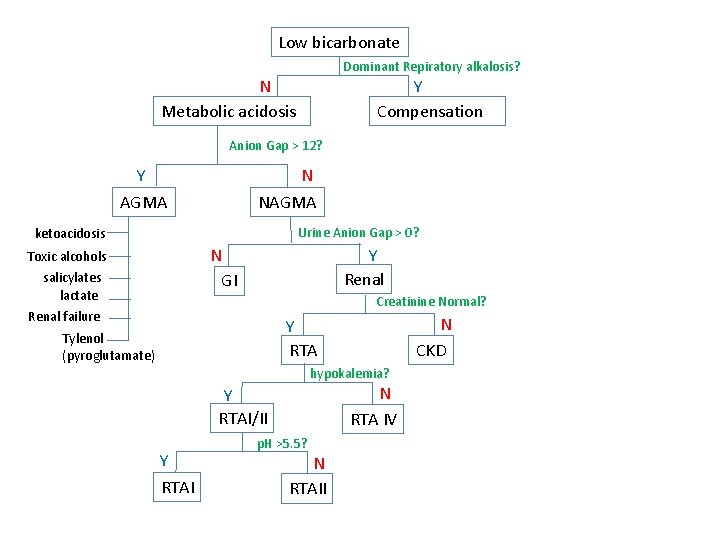
Low bicarbonate Dominant Repiratory alkalosis? N Metabolic acidosis Y Compensation Anion Gap > 12? N NAGMA Y AGMA Urine Anion Gap > 0? ketoacidosis Y Renal N GI Toxic alcohols salicylates lactate Renal failure Creatinine Normal? N CKD Y RTA Tylenol (pyroglutamate) hypokalemia? N RTA IV Y RTAI/II Y RTAI p. H >5. 5? N RTAII

Case 3 – Cardiology Ward • 62 y F with Ischemic Cardiomyopathy – LVEF = 24% • Developed severe orthopnea, NYHA IV symptoms, weight gain 10 kg over 4 days • Admitted to cardiology for diuresis – Furosemide 80 mg iv tid • Weight decreased by 12 kg in 3 days, improved symptoms
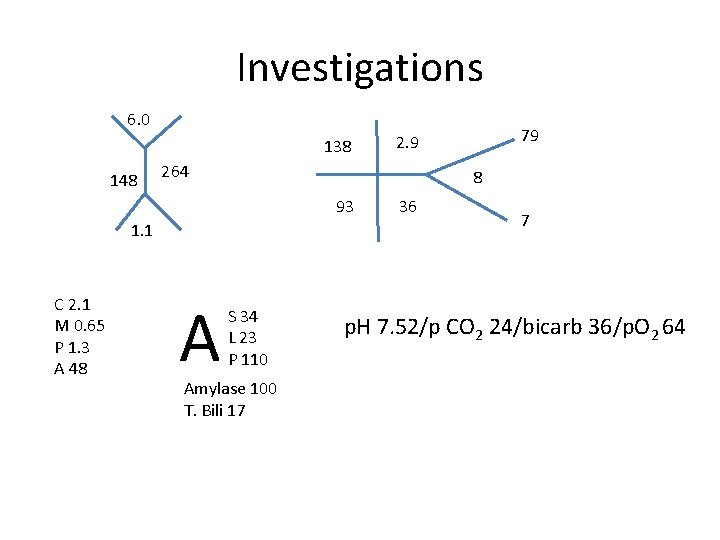
Investigations 6. 0 138 148 264 8 93 1. 1 C 2. 1 M 0. 65 P 1. 3 A 48 A 79 2. 9 S 34 L 23 P 110 Amylase 100 T. Bili 17 36 7 p. H 7. 52/p CO 2 24/bicarb 36/p. O 2 64

What’s the acid base abnormality? • Alkalosis p. H = 7. 51 • Δ bicarb = 24 – 36 = 12 • Δp. Co 2 = 40 – 46 = 6 • Metabolic Alkalosis • Compensation?
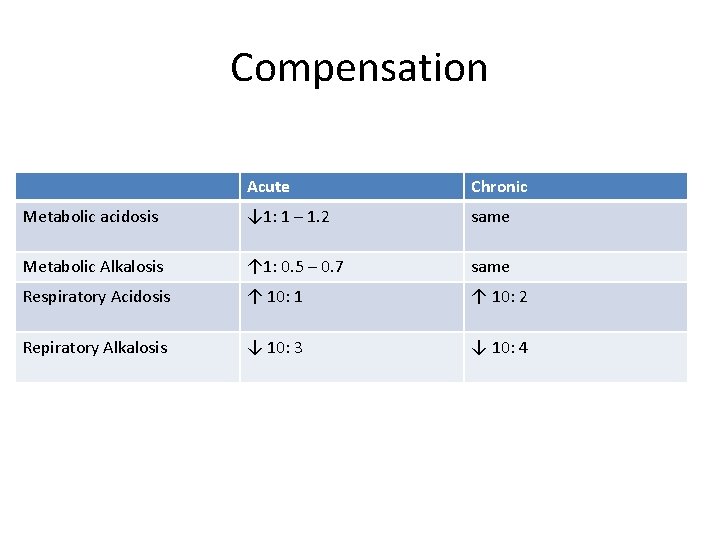
Compensation Acute Chronic Metabolic acidosis ↓ 1: 1 – 1. 2 same Metabolic Alkalosis ↑ 1: 0. 5 – 0. 7 same Respiratory Acidosis ↑ 10: 1 ↑ 10: 2 Repiratory Alkalosis ↓ 10: 3 ↓ 10: 4

Diagnosis • Anion Gap? 135 – 110 – 14 = 11 • Chronic Metabolic alkalosis with appropriate compensation
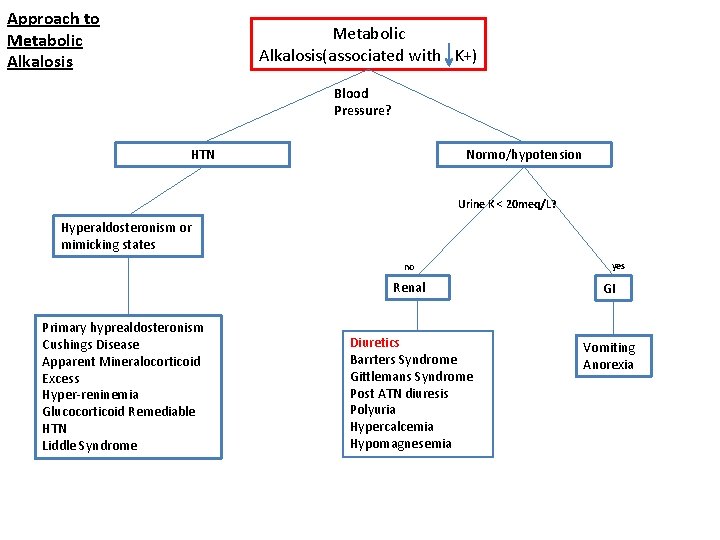
Approach to Metabolic Alkalosis(associated with K+) Blood Pressure? Normo/hypotension HTN Urine K < 20 meq/L? Hyperaldosteronism or mimicking states no Primary hyprealdosteronism Cushings Disease Apparent Mineralocorticoid Excess Hyper-reninemia Glucocorticoid Remediable HTN Liddle Syndrome yes Renal GI Diuretics Barrters Syndrome Gittlemans Syndrome Post ATN diuresis Polyuria Hypercalcemia Hypomagnesemia Vomiting Anorexia

Case 3 • Switched to oral diuretics • Potassium replaced • Alkalosis improved – bicarb came down to 28

Summary and take home points • Important to follow a step wise approach to analyzing acid base abnormalities • Always look for hidden anion gap met acidosis – Beware of missing dangerous acute intoxications that need to be managed in ICU or with dialysis • Context matters – history gives clues to the type of disorder – patient with NG tube on surgery vs patient on diuretics on medicine – both have Met Alk • Not all NAGMA is RTA – check the Urine Anion Gap

Bogged down by questions? contact: asad. merchant@uhn. ca

Extra slides

Theoretical risk of acidosis/ alkalosis • Acidosis – Nausea/ vomiting – Cramps – Cardiac irritability – Confusion – Protein dysfunction • alkalosis – coagulopathy – Protein dysfunction
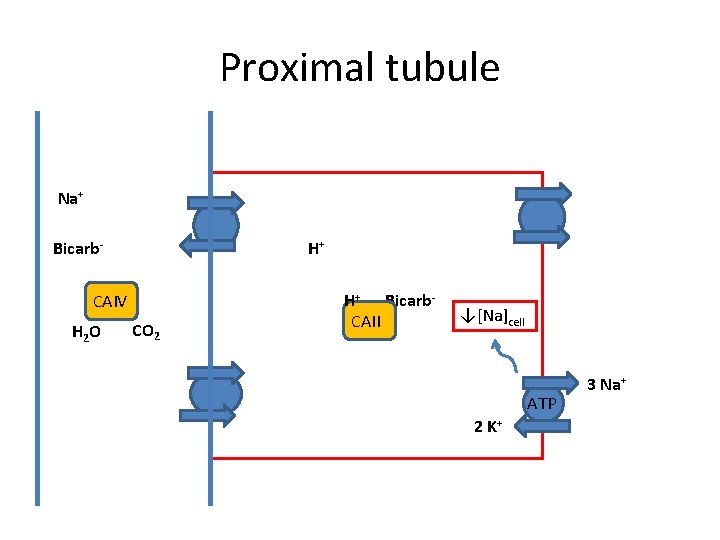
Proximal tubule Na+ Bicarb- H+ CAIV H 2 O CO 2 H+ Bicarb. CAII ↓[Na]cell ATP 2 K+ 3 Na+

Ammoniagenesis
Cortical Collecting Duct

- Slides: 70