ACID BASE BALANCE Paula Ruedebusch ARNP DNP ACIDBASE

ACID BASE BALANCE Paula Ruedebusch, ARNP, DNP
ACID-BASE BALANCE Acid-base balance is carefully regulated to maintain a normal PH via multiple mechanisms

ACID BASE IMBALANCES Rare – WHY? Secondary ABG needed Co-exist Life-threatening

IS PH IMPORTANT? Yes!! p. H <6. 9 or >7. 7: generally incompatible with life p. H determines shape and thus function of many biological molecules, particularly proteins Enzymes & ion channels are examples of protein molecules sensitive to p. H (modifies structural bonds, changing shape and function)

WHERE DOES H+ COME FROM? Intake (PO, IV, IM, NG, NJ) � Food: fatty acids, amino acids � Drugs : e. g. , aspirin, toxins, paraldehyde, ethylene glycol, methanol, formaldehyde, others ) CO 2 production: � CO 2 is not an acid but it combines with water to form carbonic acid which then dissociates into H+ and HCO 3 - Metabolic acids: � Citric acid cycle intermediates (aerobic metabolism) � Lactic acid (anaerobic metabolism) � Ketoacids--produced when some fats are incompletely metabolized. Other organic acid production.
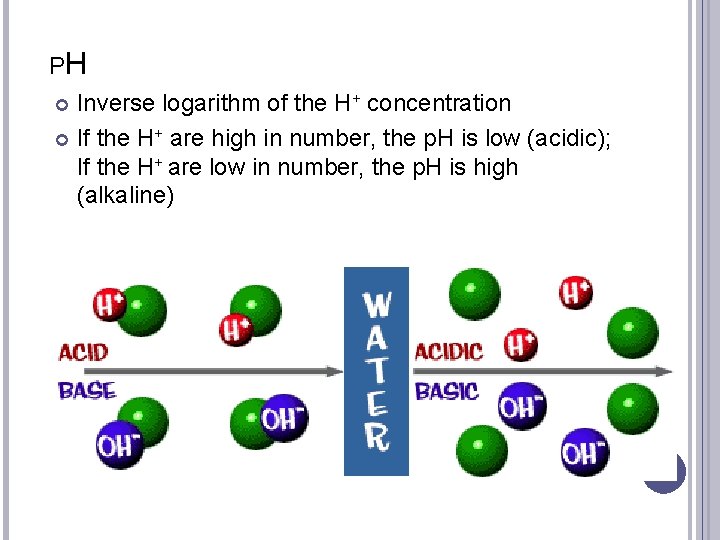
PH Inverse logarithm of the H+ concentration If the H+ are high in number, the p. H is low (acidic); If the H+ are low in number, the p. H is high (alkaline)
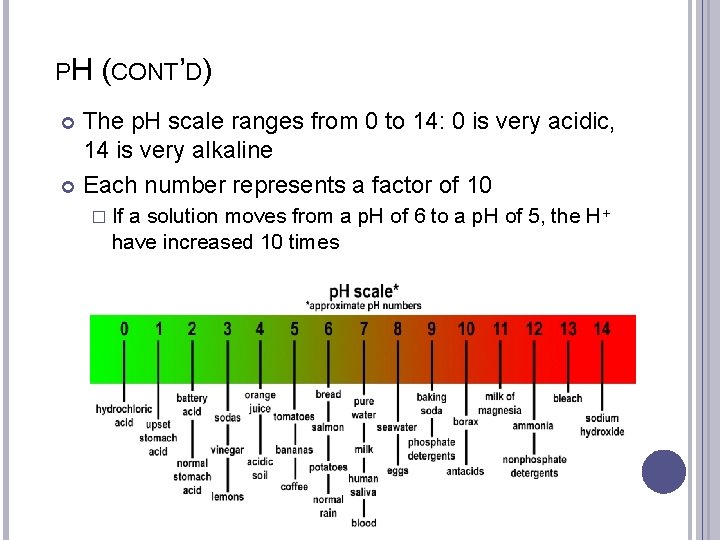
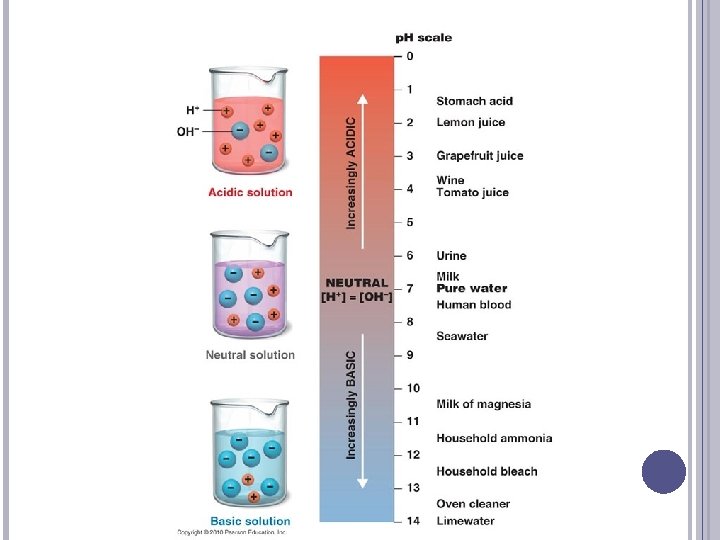
PH (CONT’D) The p. H scale ranges from 0 to 14: 0 is very acidic, 14 is very alkaline Each number represents a factor of 10 � If a solution moves from a p. H of 6 to a p. H of 5, the H+ have increased 10 times

PH (CONT’D) Acids are formed as end products of protein, carbohydrate, and fat metabolism To maintain the body’s normal p. H (7. 35 -7. 45), the H+ must be neutralized or excreted The bones, lungs, and kidneys are the major organs involved in the regulation of acid and base balance

ORGANIC ACID PRODUCTION Many metabolic intermediates are organic acids (fatty acids, amino acids, citric acid cycle components, lactic acid produced by anaerobic metabolism) � Pyruvic acid pyruvate + H+ � Lactic acid lactate + H+ Metabolic organic acid production can get out of control, producing an immense acid load � Anerobic metabolism produces lactic acid leading to lactic acidosis (seen in perfusion & oxygenation problems) � Ketoacids from incomplete fat metabolism (diabetes mellitus, starvation, alcoholism)

ANAEROBIC METABOLISM Sugar can be burned without oxygen – anaerobically Far more energy is released from burning energy aerobically Glycolysis is anaerobic, and is carried out in the cytosol
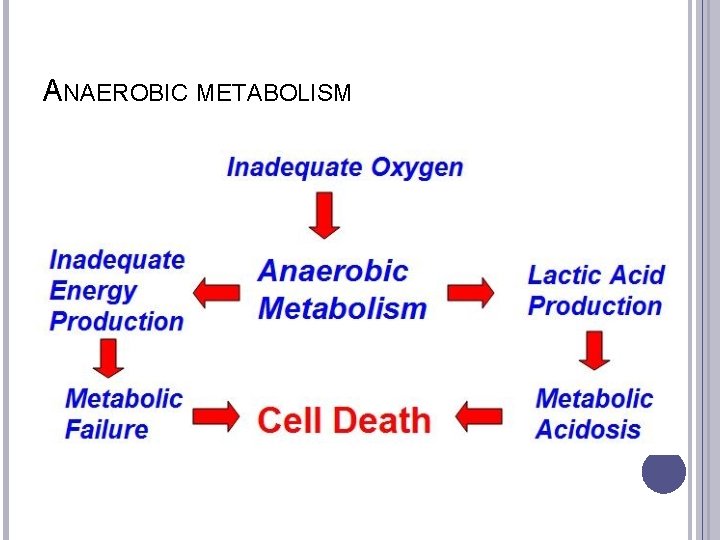
ANAEROBIC METABOLISM

ACID ELIMINATION Renal elimination in urine Respiratory elimination of CO 2 (ventilation) Emesis/gastric suction (Buffers keep p. H from changing much despite an acid load)

WHERE DOES BASE COME FROM? Some foods contain base—not many. Some fruits, vegetables (broccoli, artichoke, leek, eggplant, others); they contain an anion that binds H+ Some drugs contain base (bicarbonate; antacids)

HOW IS BASE ELIMINATED? There is not normally much base consumption so most of acid-base balance involves eliminating acid Loss of intestinal or colonic fluid results in loss of base Urine can be slightly basic (up to p. H 8. 0)
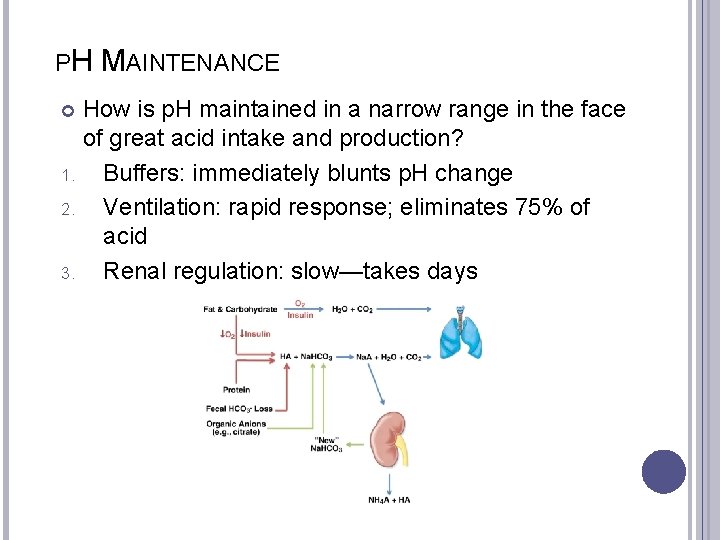
PH MAINTENANCE How is p. H maintained in a narrow range in the face of great acid intake and production? 1. Buffers: immediately blunts p. H change 2. Ventilation: rapid response; eliminates 75% of acid 3. Renal regulation: slow—takes days

BUFFERING SYSTEMS A buffer is a chemical that can bind excessive H+ or OH– without a significant change in p. H Buffers moderate p. H change: � If you add H+ to a buffer solution, p. H will change less than one would predict based on the amount of added H+. � This is because the buffer has bound up the H+ � Similarly, buffers blunt the effect of added base.

PHYSIOLOGIC BUFFERS In plasma: � HCO 3 - (goes to H 2 CO 3 then CO 2 and H 20) � Protein (goes to H+protein compound) In interstitial fluid: � HCO 3 - In intracellular fluid: � Hemoglobin � Protein � Phosphate
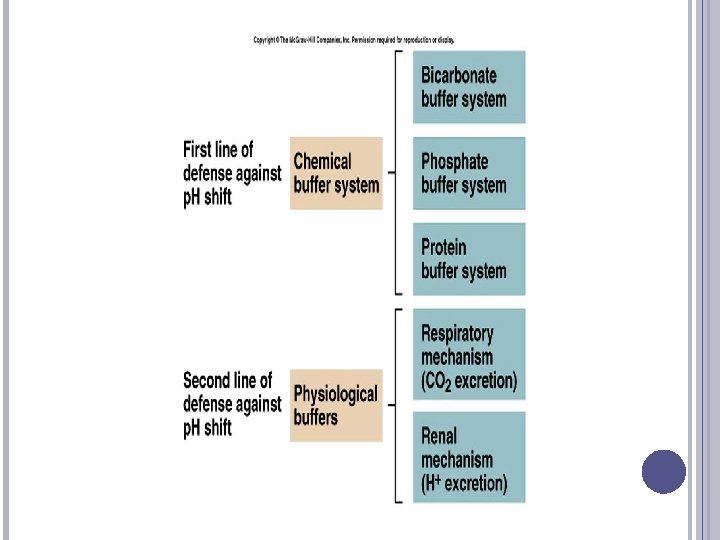

CARBONIC ACID-BICARBONATE PAIR Operates in the lung and the kidney The greater the partial pressure of carbon dioxide, the more carbonic acid is formed � At a p. H of 7. 4, the ratio of bicarbonate to carbonic acid is 20: 1 � Bicarbonate and carbonic acid can increase or decrease, but the ratio must be maintained

CARBONIC ACID–BICARBONATE PAIR (CONT’D) If the amount of bicarbonate decreases, the p. H decreases, causing a state of acidosis The p. H can be returned to normal if the amount of carbonic acid also decreases � This type of p. H adjustment is referred to as compensation

CARBONIC ACID–BICARBONATE PAIR (CONT’D) The respiratory system compensates by increasing ventilation to expire carbon dioxide or by decreasing ventilation to retain carbon dioxide The renal system compensates by producing acidic or alkaline urine
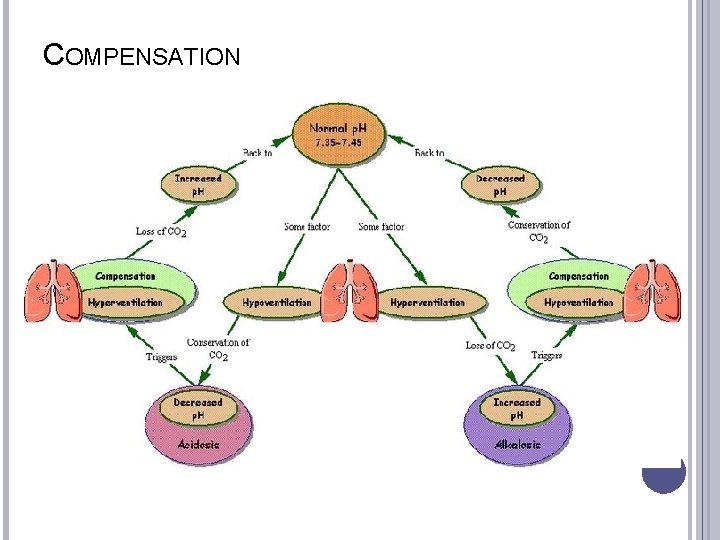
COMPENSATION

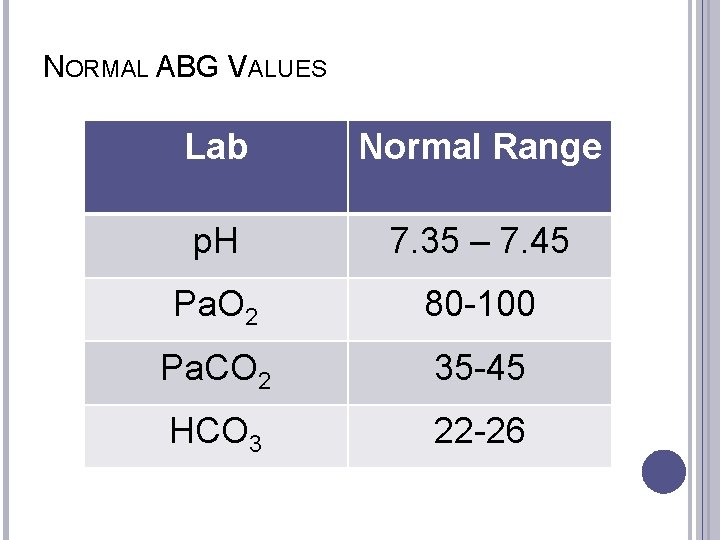
ACID-BASE IMBALANCES Normal arterial blood p. H � 7. 35 to 7. 45 � Obtained by arterial blood gas (ABG) sampling Acidosis � Systemic increase in H+ concentration or decrease in bicarbonate Alkalosis � Systemic decrease in H+ concentration or increase in bicarbonate
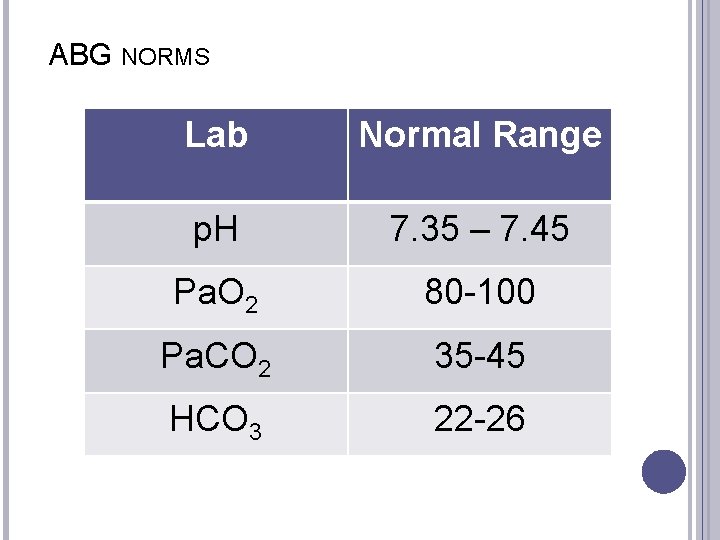
ABG NORMS Lab Normal Range p. H 7. 35 – 7. 45 Pa. O 2 80 -100 Pa. CO 2 35 -45 HCO 3 22 -26

ACIDOSIS AND ALKALOSIS Four categories of acid-base imbalances: � Respiratory acidosis—elevation of p. CO 2 as a result of ventilation depression � Respiratory alkalosis—depression of p. CO 2 as a result of alveolar hyperventilation � Metabolic acidosis—depression of HCO 3– or an increase in noncarbonic acids � Metabolic alkalosis—elevation of HCO 3– usually caused by an excessive loss of metabolic acids
RESPIRATORY ACIDOSIS
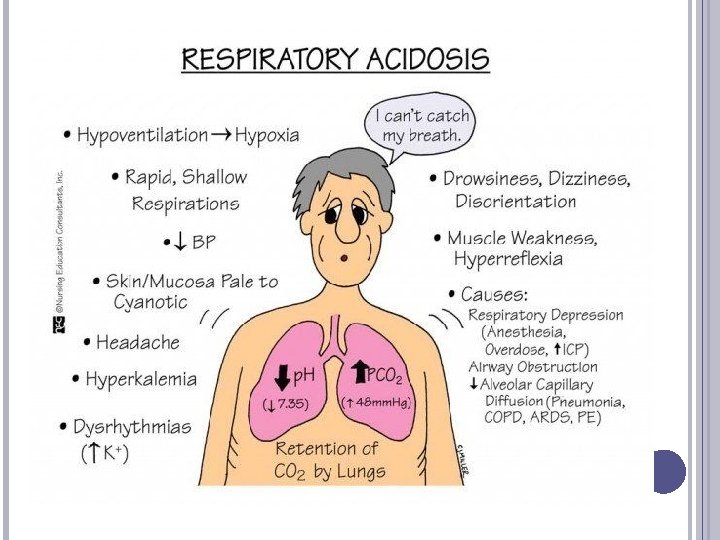

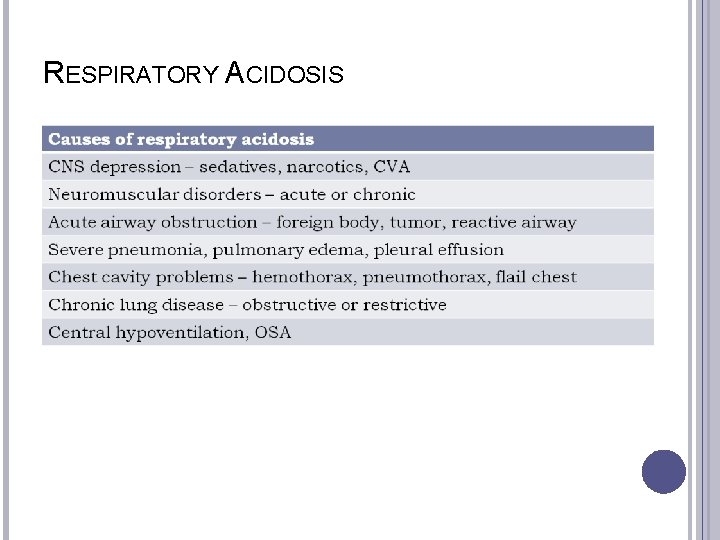
RESPIRATORY ACIDOSIS: THE PROBLEM Definition: Alveolar hypoventilation causes CO 2 retention; Pa. CO 2 increases Causes: � suppression of medullary respiratory centers by drugs (narcotics, sedation, alcohol) � trauma (increased intracranial pressure); � problems with the breathing muscles (paralysis, anesthesia, weakness) or fatigue; � Thoracic deformities, obesity � Lung problems (COPD, pneumonia, trauma)

RESPIRATORY ACIDOSIS: SIGNS, SYMPTOMS ABG: acid p. H, elevated Pa. CO 2 and HCO 3 Observe respiratory symptoms (slow or shallow or absent breathing) Chronic problem: metabolic compensation moves toward higher HCO 3 - & higher p. H
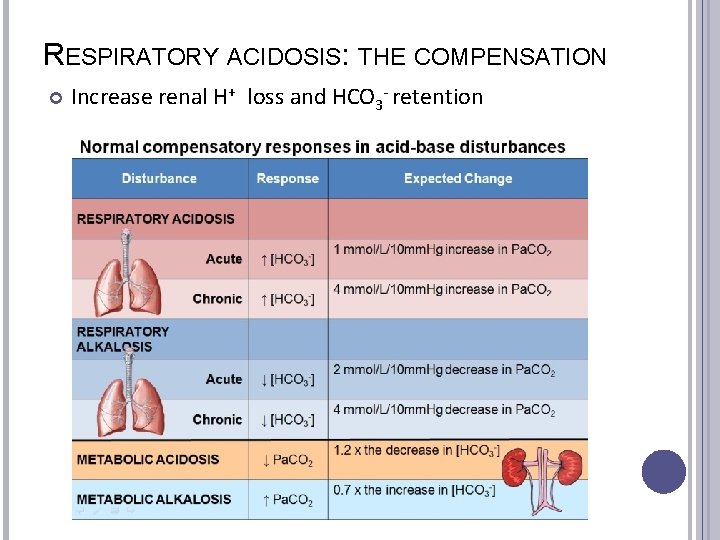
RESPIRATORY ACIDOSIS: THE COMPENSATION Increase renal H+ loss and HCO 3 - retention

RESPIRATORY ACIDOSIS: CLINICAL MANAGEMENT Acute problems: increase ventilation (reverse respiratory suppression, provide mechanical ventilation, relieve chest wall restriction, restore muscle function) Chronic problems: Support compensation; do not try to remedy the high HCO 3 - level; use caution in administering oxygen; support ventilation

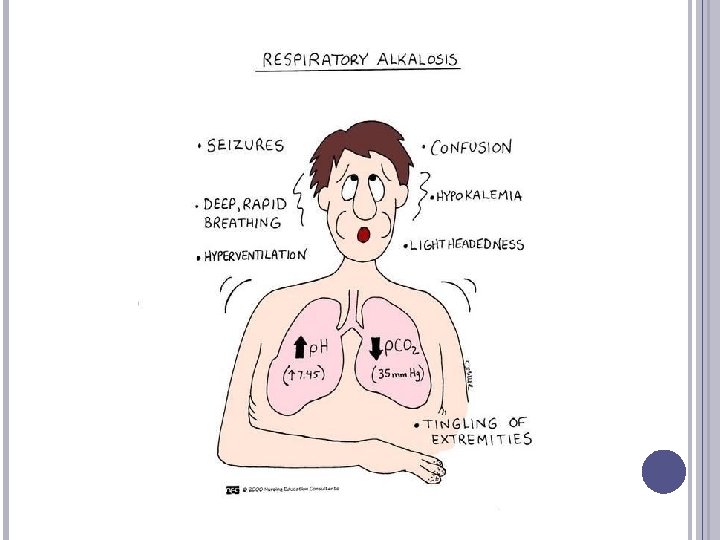
RESPIRATORY ALKALOSIS: DEFINITION & ETIOLOGY Definition: Alveolar hyperventilation causes excess CO 2 elimination; Pa. CO 2 falls Causes: � Iatrogenic (i. e. , caused by care provider: overventilation with mechanical ventilator—can be accidental or deliberate) � Hypoxemia (induces increased ventilation) � Trauma to medullary respiratory centers � Anxiety, pain � Spontaneous
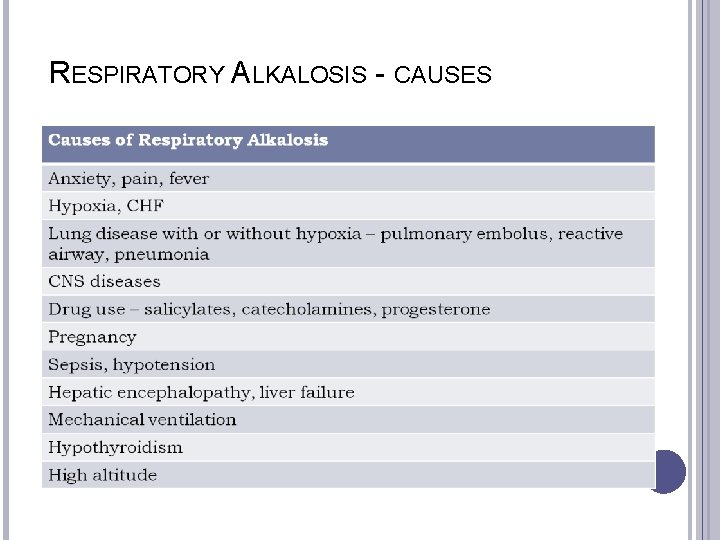
RESPIRATORY ALKALOSIS - CAUSES


RESPIRATORY ALKALOSIS: SIGNS, SYMPTOMS ABG: alkalotic p. H, low Pa. CO 2, low HCO 3 Chronic problem: renal compensation lowers HCO 3 -; partially corrects p. H Observe respiratory symptoms (hyperventilation) Irritability Paresthesia, cramps, tetany, carpopedal spasm, Chvostek’s sign, Trousseau’s sign, cardiac arrhythmias

CHOVSTEK’S SIGN

TROUSSEAU’S SIGN

CHOVSTEK AND TROUSSEAU VIDEO https: //www. youtube. com/watch? v=kvmws. TU 0 In. Q

RESPIRATORY ALKALOSIS: COMPENSATION Increase renal H+ retention This takes several days

RESPIRATORY ALKALOSIS: CLINICAL MANAGEMENT Fix acute problems: correct hypoxemia, reduce rate &/or volume of ventilation, relieve anxiety, relieve pain, etc If anxiety related, consider having the patient breathe with a paper bag over his face—this causes him to re-breathe some of his exhaled CO 2 and can raise the Pa. CO 2 Support compensation if needed

RESPIRATORY ALKALOSIS CASE Patient is a 40 y/o female with chronic asthma; she now has acute pneumonia (lung infection) ABG: p. H: 7. 6; Pa. CO 2: 24; HCO 3 - 22 m. Eq/l; hypoxemia S/Sx: hands & feet are twitching; reflexes are abnormally brisk; positive Chvostek sign; positive Trousseau’s sign Dx: acute respiratory alkalosis; no compensation Rx: administer additional inspired O 2 to improve Pa. O 2, eliminating hypoxic drive to overventilate

ETIOLOGY OF METABOLIC ACIDOSIS Excess GI HCO 3 - loss (diarrhea, loss of pancreatic fluid) Excess renal HCO 3 - loss (some types of renal failure) Drugs/toxins: amphotericin B, acetozolamide, others
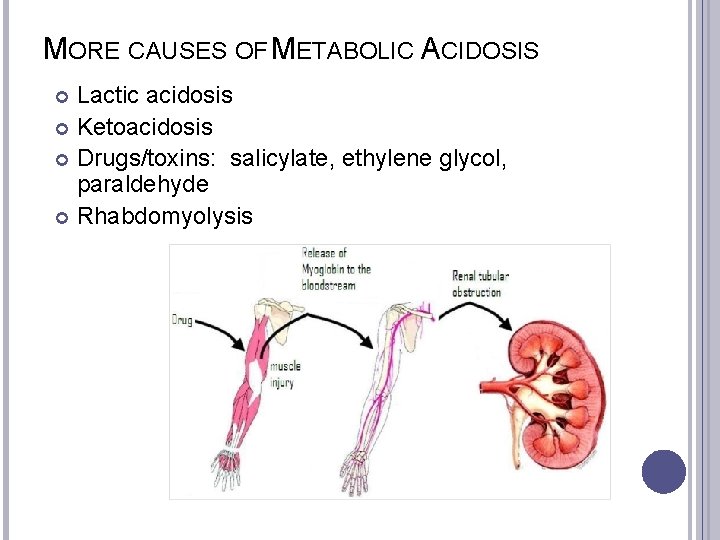
MORE CAUSES OF METABOLIC ACIDOSIS Lactic acidosis Ketoacidosis Drugs/toxins: salicylate, ethylene glycol, paraldehyde Rhabdomyolysis

METABOLIC ACIDOSIS: SIGNS, SYMPTOMS ABG: acid p. H, low HCO 3 -, low or normal Pa. CO 2 Observe respiratory symptoms (deep/rapid respirations) Headache, lethargy, progresses to coma Anorexia, nausea, vomiting, diarrhea Cardiac arrhythmias
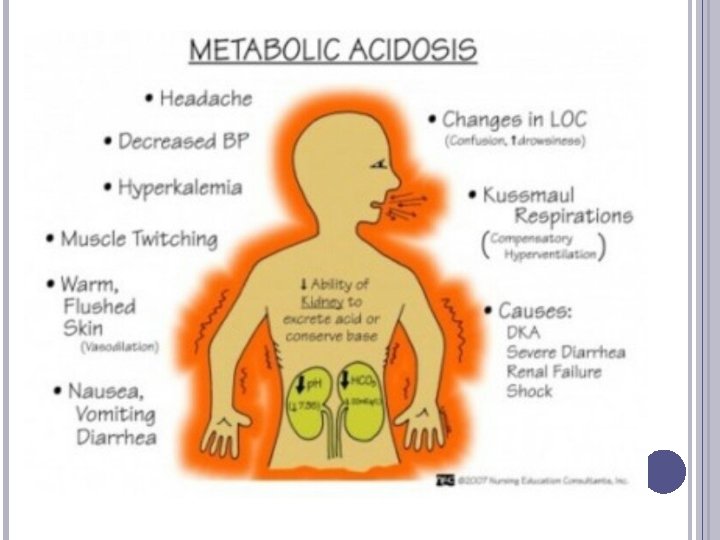

METABOLIC ACIDOSIS: THE COMPENSATION Respiratory: reduce Pa. CO 2 by breathing quickly and deeply

METABOLIC ACIDOSIS: CLINICAL MANAGEMENT Fix acute problems: improve perfusion, reverse hyperglycemia, etc. Support compensation: do not try to remedy the low Pa. CO 2; observe patient closely for signs of fatigue—this could slow ventilation and result in a combination of metabolic acidosis & respiratory acidosis If severe, (p. H<7. 1), administer sodium bicarbonate intravenously (IV)

METABOLIC ACIDOSIS CASE Patient is 30 y/o previously healthy man, now hemorrhaging Hypotensive; tachycardic Inadequate tissue perfusion, thus anaerobic metabolism ABGs: p. H: 7. 3; Pa. CO 2: 30; HCO 3 -: 14 m. Eq/l Dx: metabolic acidosis; respiratory compensation Rx: Correct problem (stop bleeding, correct hypotension by administering saline/blood products); support compensation (i. e. , allow him to continue rapid breathing)
![METABOLIC ALKALOSIS: DEFINITION & ETIOLOGY Definition: Primary increase in plasma [HCO 3 -] Causes: METABOLIC ALKALOSIS: DEFINITION & ETIOLOGY Definition: Primary increase in plasma [HCO 3 -] Causes:](http://slidetodoc.com/presentation_image_h2/bf1f969c20859d22e4f20ea8920309ff/image-51.jpg)
METABOLIC ALKALOSIS: DEFINITION & ETIOLOGY Definition: Primary increase in plasma [HCO 3 -] Causes: � Loss of H+ (gastric loss, renal loss) � Gain of HCO 3� Contraction alkalosis

METABOLIC ALKALOSIS: SIGNS, SYMPTOMS ABG: alkalotic p. H, high HCO 3 -; high Pa. CO 2 Observe respiratory symptoms (hyperventilation) Irritability Paresthesia, cramps, tetany, carpopedal spasm Hypokalemia & acidic urine if hypovolemic
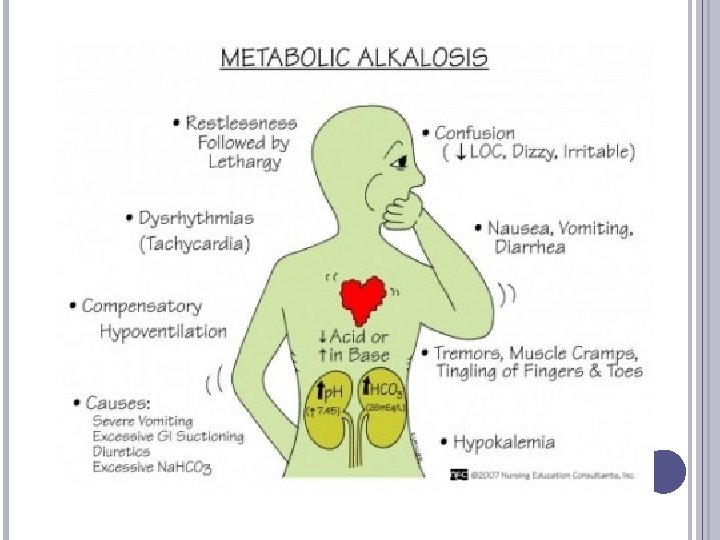

METABOLIC ALKALOSIS: CLINICAL MANAGEMENT Correct problem Support compensation if needed Compensation = Slowed ventilation: increased Pa. CO 2

METABOLIC ALKALOSIS CASE 25 y/o male; 4 day hx of vomiting; C/O exhaustion; tachycardic, orthostatic hypotension; hypopneic; brisk reflexes; Labs: hypokalemia, hypochloremia; acidic urine ABG: p. H: 7. 5; Pa. CO 2: 48; HCO 3 -: 36 m. Eq/l Dx: metabolic alkalosis, respiratory compensation; this is “contraction alkalosis” or hypokalemic, hypochloremic metabolic alkalosis, caused by loss of GI fluids & sustained by hypovolemia Rx: control vomiting; correct saline (extracellular fluid) deficit; then administer K+; do not try to increase ventilation

VENTILATION CAN IMPROVE ACID BASE BALANCE Ventilation can eliminate metabolically produced acid (eliminate CO 2 thus eliminate acid). This is the normal way to eliminate 75% of the acid load. Ventilation can eliminate excess acid when it is produced by metabolism or gained. e. g. , from aspirin poisoning. This is called respiratory compensation for a metabolic acidosis. Ventilation can retain acid when the body is too alkalotic. This is called respiratory compensation for a metabolic alkalosis.

VENTILATION CAN WORSEN ACID BASE BALANCE Decreased ventilation: raises Pa. CO 2; results in higher H+ levels; called respiratory acidosis Increased ventilation: lowers Pa. CO 2; results in lower H+ levels; called respiratory alkalosis

VENTILATION High Pa. CO 2 and low Pa. O 2 both stimulate ventilation When Pa. CO 2 rises, this stimulates ventilation; the patient breathes faster/deeper; this lowers Pa. CO 2 (“blows it off”) When Pa. CO 2 is lower than normal, this can inhibit drive to breathe The greater the respiratory rate/depth, the lower will be the patient’s Pa. CO 2 The lower the respiratory rate/depth, the higher will be the patient’s Pa. CO 2
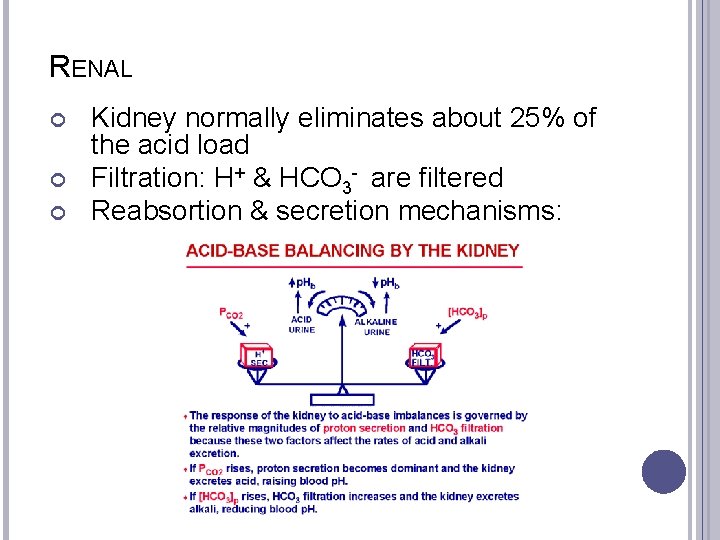
RENAL Kidney normally eliminates about 25% of the acid load Filtration: H+ & HCO 3 - are filtered Reabsortion & secretion mechanisms:

REMEMBER…

CLEAR YOUR HEAD AND GET READY!

ABGS!

STEPS FOR ABG ANALYSIS 1. 2. 3. 4. 5. What is the p. H? Acidoticor Alkalotic? What is the primary disorder present? Is there appropriate compensation? Is the compensation acute or chronic? What is the differential for the clinical processes?
NORMAL ABG VALUES Lab Normal Range p. H 7. 35 – 7. 45 Pa. O 2 80 -100 Pa. CO 2 35 -45 HCO 3 22 -26
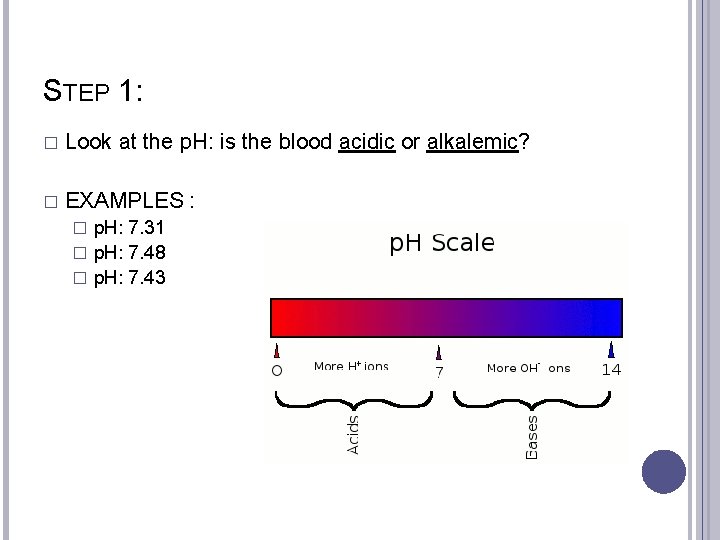
STEP 1: � Look at the p. H: is the blood acidic or alkalemic? � EXAMPLES : p. H: 7. 31 � p. H: 7. 48 � p. H: 7. 43 �
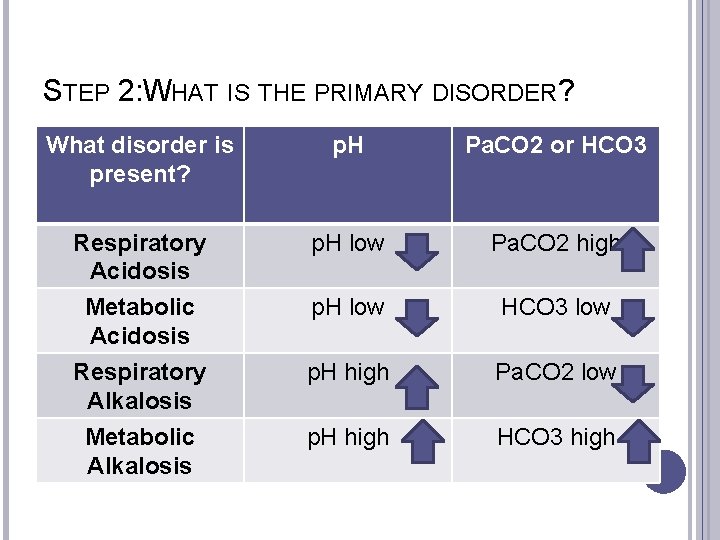
STEP 2: WHAT IS THE PRIMARY DISORDER? What disorder is present? p. H Pa. CO 2 or HCO 3 Respiratory Acidosis p. H low Pa. CO 2 high Metabolic Acidosis Respiratory Alkalosis Metabolic Alkalosis p. H low HCO 3 low p. H high Pa. CO 2 low p. H high HCO 3 high

ROME
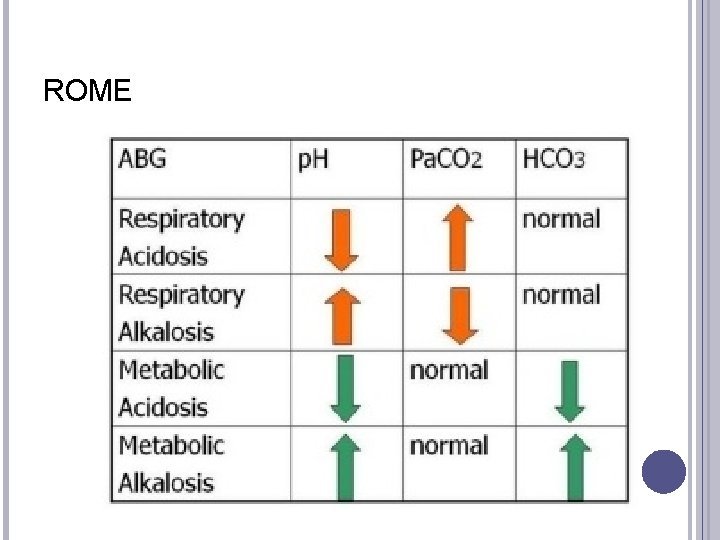
ROME
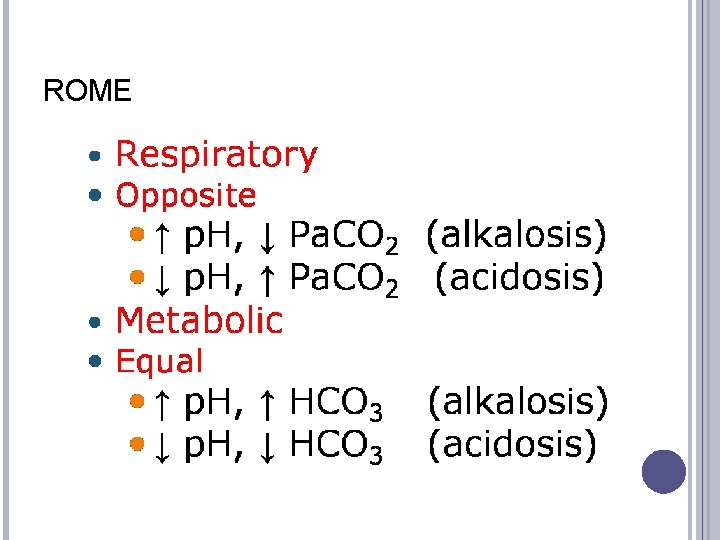
ROME

STEP 3 -4: COMPENSATED VS. UNCOMPENSATED Compensated: p. H is anywhere inside the normal ranges (Anything between 7. 35 to 7. 45) Uncompensated: p. H is anywhere outside the normal ranges - greater than 7. 45 or less than 7. 35, and the value (CO 2 or HCO 3) that does not match the p. H will still be in the normal range. Partially compensated: p. H is anywhere outside the normal ranges, and the value that does not match the p. H will be outside its normal range, indicating the body is attempting to get the p. H back to normal. For example, if the p. H (7. 20) and CO 2 (50) are acidotic, the HCO 3 should be on the alkalotic side (27).

TEST YOURSELF! 2. An arterial blood gas value of Pa. CO 2 = 47, p. H = 7. 30, Bicarbonate = 24 indicates: A. B. C. D. Metabolic acidosis Metabolic alkalosis Respiratory acidosis Respiratory alkalosis
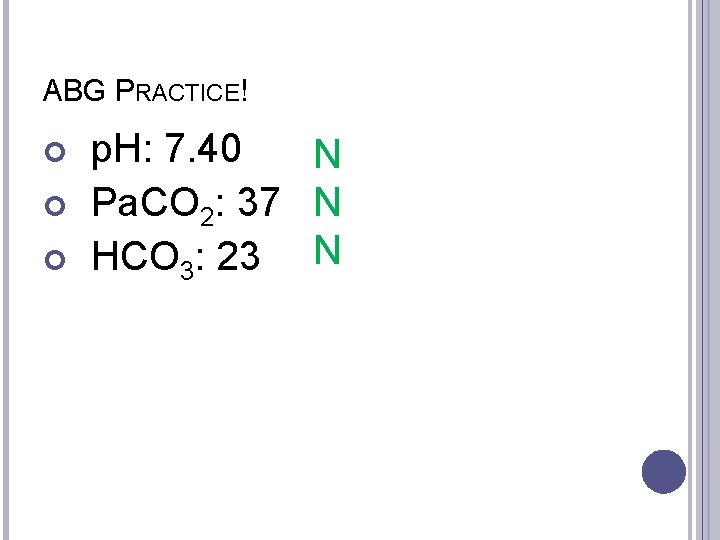
ABG PRACTICE! p. H: 7. 40 N Pa. CO 2: 37 N HCO 3: 23 N
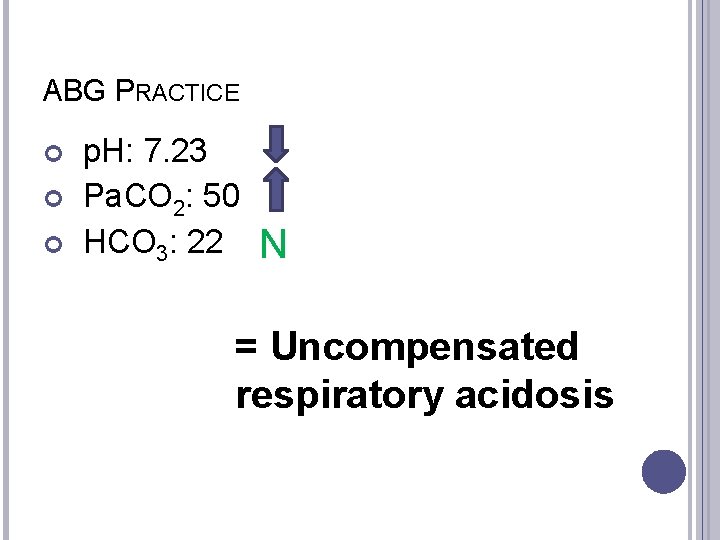
ABG PRACTICE p. H: 7. 23 Pa. CO 2: 50 HCO 3: 22 N = Uncompensated respiratory acidosis
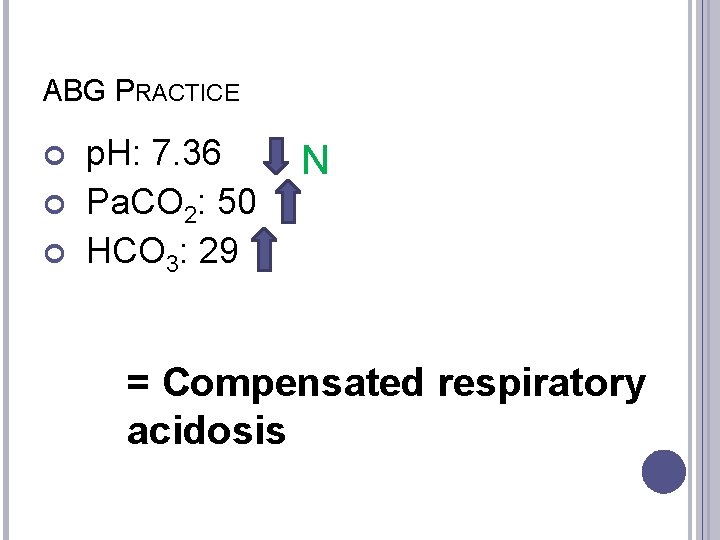
ABG PRACTICE p. H: 7. 36 Pa. CO 2: 50 HCO 3: 29 N = Compensated respiratory acidosis
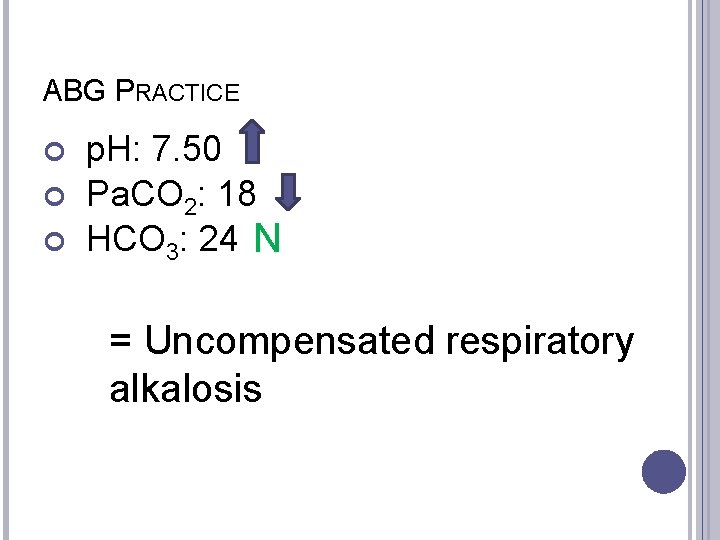
ABG PRACTICE p. H: 7. 50 Pa. CO 2: 18 HCO 3: 24 N = Uncompensated respiratory alkalosis
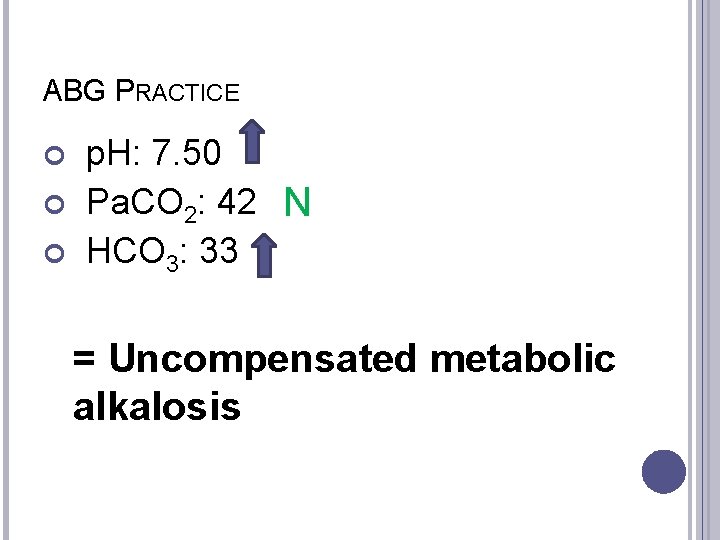
ABG PRACTICE p. H: 7. 50 Pa. CO 2: 42 N HCO 3: 33 = Uncompensated metabolic alkalosis
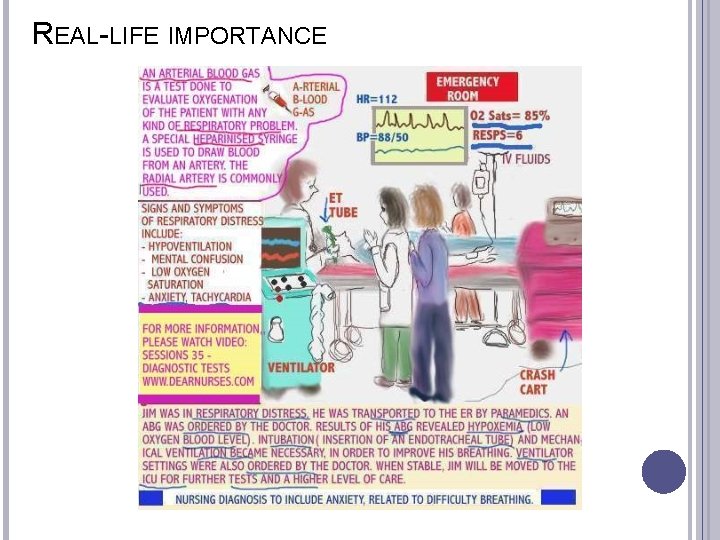
REAL-LIFE IMPORTANCE

QUESTIONS?
- Slides: 78