Acid Base Balance NURS 108 ECC Majuvy L

Acid Base Balance NURS 108 ECC Majuvy L. Sulse MSN, RN, CCRN

TERMS Ø Acid • Substance that dissociates or lose ions Ø Acidosis • • • Ø Process that adds acids or eliminates base from body fluids Hydrogen ions increased p. H is decreased<7. 35 Base • Substance that accepts ions Ø Alkalosis • Process that adds base or eliminates acid from body fluids

TERMS Ø p. H • Negative logarithm of hydrogen ion concentration in the blood • Normal range-7. 35 -7. 45 (N=7. 40) • Inversely proportional to hydrogen concentration l l Ø Increase in H ions=low p. H (acidosis) Decrease in H ions=high p. H (alkalosis) Anion gap • Normal=5 -15 • Helpful in differential diagnosis of acidosis • Calculated by subtracting anions from cations (Anion gap=Na+K –Cl+HCO 3)

TERMS Ø Pa. O 2 • Partial pressure of O 2 dissolved in arterial blood • Normal value= 80 -100 mm. Hg Ø Sa. O 2 • Amount of O 2 bound to hemoglobin l l 3% of O 2 dissolved in plasma 97% bound to hemoglobin • Normal range-93 -100%

TERMS Ø Pa. CO 2 • Partial pressure of carbon dioxide dissolved in arterial blood • Normal range =35 -45 mm. Hg • Regulated in the lungs l l Ø Hypoventilation- respiratory acidosis-CO 2 retention Hyperventilation- respiratory alkalosis- CO 2 excretion HCO 3 • Concentration of sodium bicarbonate in the blood • Normal range= 22 -26 m. Eq • Regulated in the kidneys l l Metabolic alkalosis >26 m. Eq Metabolic acidosis < 22 m. Eq
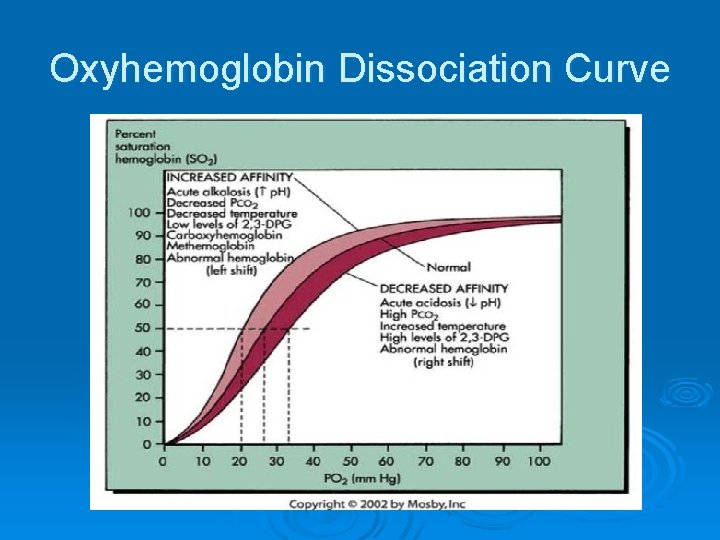
Oxyhemoglobin Dissociation Curve

Oxyhemoglobin Dissociation Curve Ø Low pa. O 2 at tissue level (hypoxia)=Shift to right-O 2 readily released from the hemoglobin • Acidemia, Hyperthermia, hypercarbia Ø High Pa. CO 2 at pulmonary capillary level=Shift to left-O 2 more bound to hemoglobin • Alkalosis, hypothermia, hypocarbia, high altitude, carcon monoxide poisoining

Acid-Base Regulation Ø Buffer system l l l Substance that reacts with an acid or base to prevent a large change in p. H Fast acting and a primary regulator of acid base balance Can react in two ways • As an acid-releasing H ions • As a base-binding a H ion l Carbonic acid/bicarbonate system H+HCO 3=H 20+ CO 2

Respiratory Mechanism 2 nd line of defense against changes in fluid p. H Ø Under control of the nervous system Ø Regulates the excretion and retention of carbonic acid Ø If p. H is down-rate & depth of ventilation increased Ø If p. H is up- rate and depth of ventilation is decreased Ø Rapid action following alteration in acid base balance Ø

Neural regulation of respiration and Hydrogen Ion concentration Decreased rate and depth of respiration Increased. Pa. CO 2 Inhibition of central chemoreceptors Stimulation of Chemoreceptors Decreased Pa. CO 2 Decreased H+ Increased rate and depth of respiration Increased H+

Renal Mechanism Ø 3 rd line of defense Ø Strongest of all mechanisms but takes long to completely respond (24 -48 hrs) Ø Operates on 3 mechanisms l l l Movement of bicarbonate Formation of acids Formation of ammonium

Renal Mechanism Ø Movement of bicarbonate • When H ions are high-bicarbonates are reabsorbed from the kidneys and back to circulation • When H ions are low-bicarbonates remain in the kidneys and excreted in the urine Ø Formation of acids • Phosphate (HPO 4) draws and combines with H ions into the urine forming an acid (H 2 PO 4) and excreted in the urine Ø Formation of ammonium • Ammonia (NH 3) is secreted in the urine-combines with H ions to form Ammonium (NH 4)-excreted in urine

Compensation Body attempts to correct for the changes in body/blood p. H Ø Respiratory system- more sensitive to aci-base changes thus can begin compensation within seconds to minutes Ø Renal compensation-more powerful but does not get stimulated until imbalance is sustained for several hours to days Ø

Compensation Ø Respiratory- Rapid • Metabolic acidosis- increase in depth and rate of respiration to blow off CO 2 -p. H returns to normal • Metabolic alkalosis-lungs decrease the rate & depth of respiration-pa. CO 2 returns to normal Ø Renal • Respiratory acidosis-kidneys increase excretion of H ions or increase reabsorption of bicarbonates-p. H returns to normal • Respiratory alkalosis-kidneys reabsorb more H ions or excrete more bicarbonates

Compensation Ø p. H –within normal 7. 35 -7. 45 Ø HCO 3 -must be abnormal Ø p. CO 2 - & HCO 3 going in the same direction Ø Example • p. H -7. 37 p. CO 2 -60 HCO 3 - 38

Acid Base Imbalances Ø Metabolic Acidosis-low p. H, low HCO 3, K elevated, • Overproduction of H ions l Excessive breakdown of fatty acids-ketoacidosis (DKA & Starvation) releases H ions l Hypermetabolism-lactic acidosis- excessive exercise, seizure, fever, hypoxia l Excessive intake of acid substances-ASA, alcoholic beverages • Under elimination of H ions –kidney failure • Underproduction of bicarbonates-renal/liver failure, dehydration • Overproduction of bicarbonates-but presence of diarrhea

Metabolic Acidosis-Clinical Manifestations Ø Neurologic- • Headache, drowsiness, confusion, coma Ø Neuromascular • Decrease in muscle tone and deep tendon reflexes Ø Respiratory • Deep rapid respirations-Kaussmaul breathing Ø Cardiovascular • Low BP arrhythmias • Warm flushed skin due to vasodilation Ø GI • Nausea, vomiting, diarrhea, abdominal pain

Metabolic Acidosis-Interventions Ø Hydration Ø Treat or control the causative cause • DKA- hydration & insulin • Diarrhea-rehydration & antidiarrheals Ø Bicarbonate is administered only if serum bicarbonate levels are low

Acid Base Imbalances Ø Respiratory Acidosis-low p. H, high p. CO 2, K l l Retention of CO 2 Respiratory depression • Anesthesia, drugs, trauma, neurologic disease, l Inadequate chest expansion • Skeletal deformities, muscle weakness, obesity, tumor l Airway obstruction • asthma, COPD, bronchiolitis l Reduced alveolar-capillary diffusion • Thrombus, pneumonia, TB, Cystic fibrosis, atelectasis, ARDS,

Respiratory Acidosis- Manifestations Ø Neurologic • Disorientation, drowsiness, dizziness, headache, coma Ø Respiratory • hypoventilation Ø Cardiovascular • Low BP, arrhythmias Ø Neuromascular • seizures

Respiratory Acidosis-Interventions Maintain patent airway, enhance gas exchange, adequate oxygenation Ø Pulmonary hygiene-positioning breathing techniques Ø Ventilatory support Ø Prevention of complications Ø Drug therapy aimed at Ø l Increasing diameter of airways • Induce relaxation • Increase bronchodilation l Thin secretions

Combined Respiratory & Metabolic Acidosis Ø Can occur simultaneously Ø Leads to anaerobic metabolism and lactic acidosis Ø Acidosis more profound than that caused by respiratory or metabolic acidosis Ø May lead to cardiac arrest

Acid Base Imbalances Ø Metabolic Alkalosis-high p. H hallmarked by an increased in bicarbonate and rising pa. CO 2, low K & Ca l Base excess • Ingestion of bicarbonates, acetates, citrates, and lactates l Acid deficit • Prolonged vomiting, Cushing’s syndrome, Thiazide diuretics, prolonged NGT suctioning

Metabolic Alkalosis. Manifestations Ø Neurologic • Dizziness, irritability, nervousness, confusion Ø Respiratory • Hypoventilation-a compensatory action Ø Cardiovascular • Tachycardia, arrhythmia related to low K Ø Neuromascular • Tetany, tremors, tingling of fingers & toes, hypertonic muscles, cramps & seizures Ø GI • Anorexia, nausea & vomiting

Metabolic Alkalosis. Interventions Ø Restore normal fluid & electrolyte balance Ø Drug therapy to restore electrolyte balance • K sparing diuretics • Antiemetics Ø Avoid administration of alkaline substances-Na Bicarbonate or antacids

Acid Base Imbalances Ø Respiratory Alkalosis-high p. H, low bicarbonate, low Pa. Co 2, low K & Ca Ø Co 2 level is so low because of hyperventilation l l l Direct stimulation of respiratory center due to fever, compensation for metabolic acidosis, CNS lesions, drugs, pain ventilation settings too high or fast Anxiety, fear

Respiratory alkalosis. Manifestations Ø Neurologic • Light headedness, lethargy, confusion Ø Respiratory • Hyperventilation- lungs cannot compensate for the respiratory problem Ø Cardiovascular • Tachycardia, arrhythmias Ø Neuromascular • Numbness, tetany, tingling of extremity, • Hyperflexia, seizures Ø GI • Nausea, vomiting, epigastric pain

Respiratory alkalosis-Interventions Ø Monitor for indications of respiratory failure Ø Use rebreather mask Ø Provide mechanical ventilatory support Ø Reduce O 2 consumption to minimize hyperventilation-reduce fever, pain, anxiety and promote comfort Ø Monitor labs-ABGs, lytes

Arterial Blood Gases Provides acid-base status Provides information on the origin of the imbalance Ø Provides an idea of body’s ability to regulate p. H Ø Provides reflection of overall oxygenation status Ø Finding interpreted in conjunction with patient clinical history, physical assessment, and previous ABG Ø Ø

Steps in determining ABGs Ø Ø 1)Determine if p. H is acidotic or alkalotic 2)Analyze the p. CO 2 to determine respiratory acidosis or metabolic. CO 2 is controlled by the lungs, • high CO 2=acidosis, low CO 2 =alkalosis Ø 3)Analyze HCO 3 to determine metabolic acidosis or alkalosis. HCO 3 is the metabolic component controlled by the kidney • high HCO 3=alkalosis, low HCO 3=acidosis Ø 4)Determine if CO 2 or HCO 3 matches the acid or base alteration of p. H. • If p. H is acidotic and CO 2 is high=respiratory acidosis • . If p. H and HCO 3 high= metabolic acidosis Ø 5)Decide if the body is attempting to compensate for the p. H change
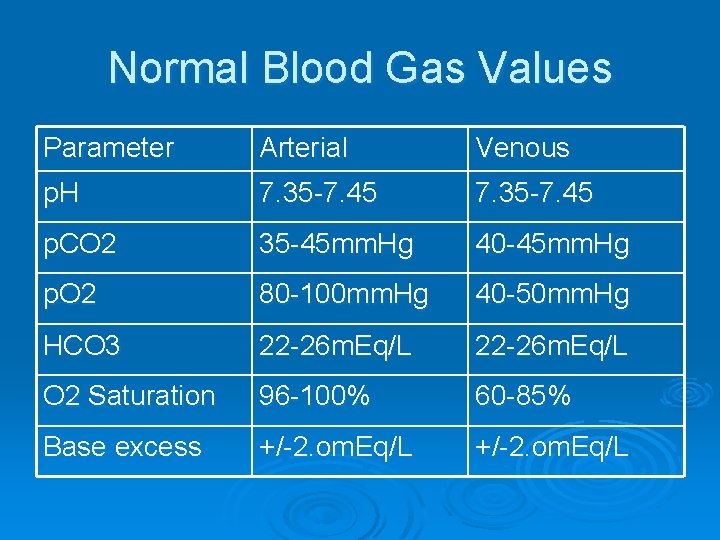
Normal Blood Gas Values Parameter Arterial Venous p. H 7. 35 -7. 45 p. CO 2 35 -45 mm. Hg 40 -45 mm. Hg p. O 2 80 -100 mm. Hg 40 -50 mm. Hg HCO 3 22 -26 m. Eq/L O 2 Saturation 96 -100% 60 -85% Base excess +/-2. om. Eq/L

What acid-base disorders are represented by the following arterial blood tests Ø Ø Ø Ø p. H 7. 18 7. 56 7. 21 7. 32 7. 50 7. 49 7. 37 p. Co 2 68 50 51 49 22 32 57 HCO 3 28 32 19 22 29 31 17
- Slides: 32