Acid base balance By Dr Warda Ramadan Lecturer

Acid base balance By Dr/ Warda Ramadan Lecturer of critical care &emergency Nursing

Outlines n Introduction n Regulation of acid base balance q Buffers system q Role of kidney q Role of lungs n Metabolic acidosis n Metabolic alkalosis n Respiratory acidosis n Respiratory alkalosis

Introduction The p. H is a measure of the hydrogen ion concentration in the blood and provides information about the acidity or alkalinity of the blood. A normal p. H is 7. 35 to 7. 45. n n As hydrogen ions accumulate, the p. H drops, resulting in acidemia. n Decrease in hydrogen ions results in an elevation of the p. H and alkalemia.

Acids: n n n is a substance that can donate a hydrogen ion (H+) to a solution. There are two different types of acids: volatile acids and nonvolatile acids. Volatile acids ( )ﻣﺘﻄﺎﻳﺮ ﺍﻭ ﺳﺮﻳﻊ ﺍﻟﺰﻭﺍﻝ are those that can move between the liquid and gaseous states. Once in the gaseous state, these acids can be removed by the lungs. The major acid in the blood serum is carbonic acid (H 2 CO 3). This acid is broken down into carbon dioxide(Co 2) and water (H 2 O) by an enzyme produced in the kidneys.

Acids: n n Nonvolatile (“fixed”) acids are those that cannot change into a gaseous form and therefore cannot be excreted by the lungs. They can only be excreted by the kidneys (a metabolic process). Examples of nonvolatile acids are lactic acid and ketoacids.

Bases: n A base is a substance that can accept a hydrogen ion (H+), thereby removing it from the circulating serum. The main base found in the serum is bicarbonate (HCO 3). n The amount of bicarbonate that is available in the serum is regulated by the kidney (a metabolic process).

Mechanisms to maintain acid base balance Acid/base balance is controlled through three functions: n n n Respiratory and metabolic regulations Renal regulation, and chemical buffers).

Mechanisms to maintain acid base balance

Chemical buffers: n It substance that prevent major changes in the p. H of body fluids by removing or releasing H+; they can act quickly to prevent excessive changes in H+ concentration. n Buffers act chemically to change strong acids into weaker acids or to bind acids to neutralize their effect. This minimizes the effect of acids on blood p. H until their excretion from the body.

n Buffers cannot maintain p. H without the adequate functioning of the respiratory and renal systems. n Buffer systems occupy various locations in the body. Proteins and phosphates are buffers in the cells, hemoglobin is a buffer in red blood cells, and bicarbonate & proteins again, are buffers in the extracellular fluid. The combination of all the buffer system is called the total buffer base.

2 - Renal regulations: n n n The kidneys defend blood p. H by controlling bicarbonate concentration. This is accomplished by excretion of hydrogen ions in the urine when the blood is too acidic and excretion of bicarbonate in the urine when the blood is too alkaline. It may take hours to days for the kidney to affect p. H.

2 - Respiratory regulations: n Respiratory system controls carbon dioixide tension of the blood by regulating alveolar ventilation, that may very quickly correct an acid-base disturbance through hypoventilation or hyperventilation.

Acid- Bas Imbalance n An acid-base imbalance results when there is an alteration in the ratio of 20: 1 between base and acid content. n Acid-base imbalances respiratory or metabolic. are classified as

PH measurement: n Chemically, the p. H scale ranges from 0 -14, making p. H 7 chemically neutral. n Arterial blood is normally slightly alkaline (p. H 7. 35 - 7. 45). blood p. H < 7. 35 is acidotic, while p. H > 7. 45 is alkalotic. n Arterial p. H below 7. 0 usually leads to coma and death, while levels above 7. 8 over stimulate the nervous system, causing convulsions and respiratory arrest.

Expected ABG Outcomes n PH: hydrogen ion concentration of blood and is an indicator of Acid- Base status. n PH 7. 35 -7. 45 n ↓ PH acidosis n ↑ PH alkalosis

n Pa. CO 2: - the partial pressure of carbon dioxide in the arterial blood. Pa. CO 2 35 -45 mm. Hg ↓ Pa. CO 2 respiratory alkalosis ↑ Pa. CO 2 respiratory acidosis n n - HCO 3: - the serum bicarbonate, which is the major component of the renal compensatory mechanism. HCO 3 22 - 26 m. Eq/L ↓ HCO 3 metabolic acidosis ↑ HCO 3 metabolic alkalosis n n n

n n Sa. O 2: -the saturation of hemoglobin by oxygen n Sa. O 2: 95 - 99% - BE: - base excess reflects an increase or decrease in total buffer base. n BE: - -2 to +2 m. Eq/L n ↓ BE metabolic acidosis n ↑ BE metabolic alkalosis - Pa. O 2: - the partial pressure of oxygen in the arterial blood n Pa. O 2: -80 -100 mm. Hg Values less than 60 may result in lactic acid production and metabolic acidosis because of anaerobic metabolism brought on by hypoxemia.

Metabolic acidosis (base deficit or acid excess) n n n It is characterized by a low p. H (increased H+ concentration) and a low plasma bicarbonate concentration. It can be divided clinically into two forms, according to the values of the serum anion gap: high anion gap acidosis and normal anion gap acidosis. The anion gap reflects normally unmeasured anions (phosphates, sulfates, and proteins) in plasma. Measuring the anion gap is essential in analyzing acid–base disorders correctly.

Metabolic acidosis n PH ↓ 7. 35 n HCO 3 ↓ 22 m. Eq/L n Pa. CO 2 ↑ 45 mm. Hg acidosis metabolic acidosis

Etiological factors n Normal anion gap acidosis results from the {direct loss of bicarbonate}, as in q diarrhea, q lower intestinal fistulas, q ureterostomidostomy, q use of diuretics; q early renal insufficiency (ATN); q administration of acidifying drugs( as ammonium chloride)

n High anion gap acidosis results from {excessive accumulation of fixed (nonvolitile) acid}. q ketoacidosis, q lactic acidosis, q the late phase of salicylate poisoning, uremia, methanol or ethylene glycol toxicity, and q ketoacidosis with starvation. q Renal failure.


Treatment n Treatment is aimed metabolic defect: at correcting q Maintain tissue perfusion and oxygenation q IV bicarbonate q q the Potassium management (Hyperkalemia may also occur due to shift of potassium out of the cells. Hypokalemia may occur once the acidosis is corrected) Dialysis

Metabolic alkalosis (excess base or loss acid) n n Metabolic alkalosis is a clinical disturbance characterized by a high p. H (decreased H+ concentration) and a high plasma bicarbonate concentration. It can be produced by a gain of bicarbonate or a loss of H+. Body may attempt to compensate decreasing respirations to increase CO 2 by

Metabolic alkalosis n PH ↑ 7. 45 alkalosis n HCO 3 ↑ 26 m. Eq/L metabolic alkalosis n Pa. CO 2 ↓ 45 mm. Hg alkalosis

Etiological factors n Vomiting or gastric suction. n Hypokalemia q q In hypokalemia, kidney conserve potassium and thus increase hydrogen ion excretion. Cellular potassium shifts out into ECF, hydrogen must enter to maintain electroneutrality. n Excessive alkali ingestion. n Potassium-losing diuretics. n Cushing syndrome. n Hyperaldosteronism n Excess Na. HCO 3 administration for CPR.

Treatment n Treatment is aimed at treating the underlying disorder q Chloride supplementation for the kidney to absorb sodium with chloride (allowing the excretion of excess bicarbonate). q Restore normal fluid volume q Maintain potassium q Carbonic anhydrase inhibitor if unable to tolerate volume expansion (as heart failure)

Respiratory acidosis n PH ↓ 7. 35 n Pa. CO 2 ↑ 45 mm. Hg n HCO 3 ↓ 22 m. Eq/L acidosis Respiratory acidosis

Etiological factors Neuromuscular problems - Guillain-Barre syndrome -Myasthenia gravis - Poliomyelitis -Spinal cord injury n Respiratory center depression - CNS trauma -Brain lesions - Obesity -Primary hypoventilation - Use of certain drugs n Lung diseases - Respiratory infection -COPD - Acute asthma attacks -Chronic bronchitis - ARDS -Pulmonary edema - Chest trauma n

Airway obstruction - Retained secretions -Tumors - Anaphylaxis -Laryngeal spasm - Lung diseases that alter alveolar ventilation. n CAUSES OF Chronic respiratory acidosis n Emphysema n Cystic fibrosis n Bronchiectasis n Bronchial asthma

Diagnostic test: n ABG findings indicative of respiratory acidosis Chest X-ray - Evidence of COPD -Evidence of pneumonia, pneumothorax or other causes n Electrolyte levels - Potassium level greater than 5 m. Eq/L n Other blood test - Drug screening that may detect overdose n

Treatment n n n Maintain a patent airway Give bronchodilator to open constricted airways Administer supplemental oxygen as needed. Watch for worsening hypercapnia in patient with COPD. Give an antibiotic to treat infection Perform chest physiotherapy to remove secretions from the lung

Treatment (Count) n n Perform tracheal suctioning, incentive spirometry, and postural drainage, and assist with coughing and deep breathing as needed. Monitor for changes in the patient's cardiac rhythm and respiratory pattern. Closely observe the patient's neurologic status and report significant changes. Promote fluid intake and carefully track fluid intake and output.
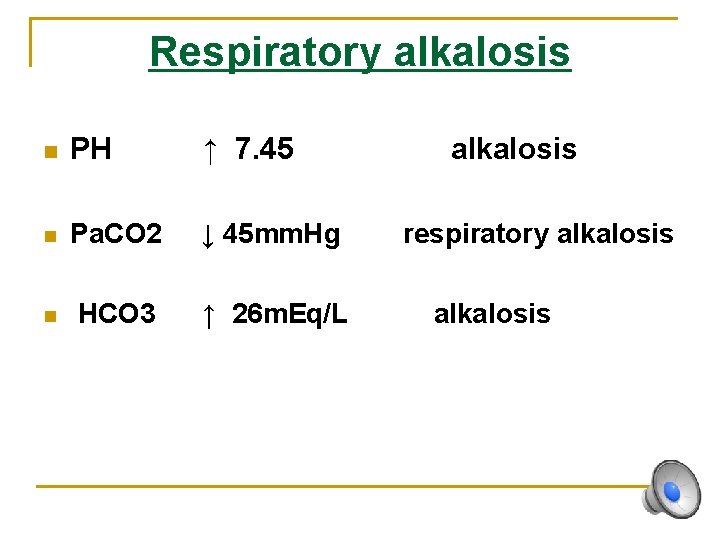
Respiratory alkalosis n PH ↑ 7. 45 n Pa. CO 2 ↓ 45 mm. Hg n HCO 3 ↑ 26 m. Eq/L alkalosis respiratory alkalosis

Etiological factors Hyperventilation = Anxiety, Pain, salicylates intoxication (early onset) = Use of certain drugs n Hyper-metabolic states = Fever, Liver failure, Early sepsis n Conditions that affect respiratory control center n Other causes = Acute hypoxia secondary to high altitude = Pulmonary disease, Severe anemia, Pulmonary embolus hypotension q

Diagnostic test: n n n ABG findings indicative of respiratory Alkalosis ECG changes, arrhythmia Characteristics indications of hypokalemia, hypocalcemia, or hypomagnesemia Electrolyte levels Serum calcium, Potassium , magnesium and chloride level below normal Other blood test: Toxicology screening with evidence of salicylate poisoning

Treatment n n n n Correct the underlying cause, for example by treating Salicylates intoxication or Early sepsis Administer supplemental oxygen as needed. Give sedative or anxiolytic if anxiety is the cause. If the cause is iatrogenic, adjust the ventilator settings Monitor vital signs. Report changes in neurologic, neuromuscular, or cardiovascular functioning. Monitor ABG and serum electrolyte levels and immediately report any changes Institute seizure precautions as needed.

Thank you Any Questions ? ?
- Slides: 38