ACID BASE BALANCE ACID BASE BALANCE n General

ACID BASE BALANCE

ACID BASE BALANCE n General Principles – Dynamic relationship which reflects the concentration of hydrogen (H+) ions – Hydrogen ions are acids which must be maintained within strict limits » Any deviation from norm affects biochemical events – Measured in a negative log (p. H) which is inversely proportional to the hydrogen ion concentration » High H+ ion concentration = Low p. H » Low H+ ion concentration = High p. H

THE p. H SCALE n n Most frequently used measure of acid base balance Ranges from 1 -14 – 1 means only hydrogen ions present – 14 means no hydrogen ions present – p. H of water is 7. 0 (neutral) p. H of human body is 7. 35 -7. 45 p. H compatible with life = 6. 9 -7. 8 – p. H of < 7. 35 indicates a state of acidosis – p. H of > 7. 45 indicates a state of alkalosis – Variation of 0. 4 in either direction can be fatal

Acid-Base Balance n n A p. H below 7. 35 is referred to as acidosis A p. H above 7. 45 is referred to as alkalosis

n n Body constantly produces acids through metabolism – Carbon dioxide – Metabolic acids (lactic and pyruvic acid) Acids must be constantly eliminated from the body – As acids increase, bodily functions decrease – Example » Diminishes myocardial contractility » Reduces vascular response to catecholamines » Interferes with the actions of pharmacological agents


BUFFER SYSTEM n n Fastest acting defense mechanism (seconds) Chief buffer is bicarbonate – Bicarbonate combines with excess hydrogen ions to form carbonic acid in a dynamic relationship: HCO 3 + H+ <----> H 2 CO 3 – For every molecule of carbonic acid, there are 20 molecules of bicarbonate – Any change in 20: 1 ratio is immediately corrected to maintain p. H » An increase in H+ causes an increase in H 2 CO 3 » A decrease in H+ causes a decrease in H 2 CO 3

Bicarbonate Buffer System Fastest mechanism n Hydrogen will bind with bicarbonate n Results in formation of carbonic acid n

Carbonate Buffer System An increase in hydrogen ions leads to an increase in carbonic acid. n A deficit in hydrogen ions (alkalosis); carbonic acid will dissociate into bicarbonate ion and hydrogen ion n

BUFFER SYSTEM (CONT. ) n Carbonic acid is a weak, volatile acid which must be eliminated n Enzyme carbonic anhydrase catalyzes the carbonic acid to convert to carbon dioxide and water: H 2 CO 3 ----->CO 2 + H 2 O n CO 2 and H 2 O are easily eliminated by the lungs and kidneys

n Functions within minutes (1 -3) n Lungs eliminate excess CO 2 by increasing respirations, causing a decrease in H+ ion and an increase in p. H n Lungs can retain more CO 2 by decreasing respirations, causing an increase in H+ ions and a decrease in p. H
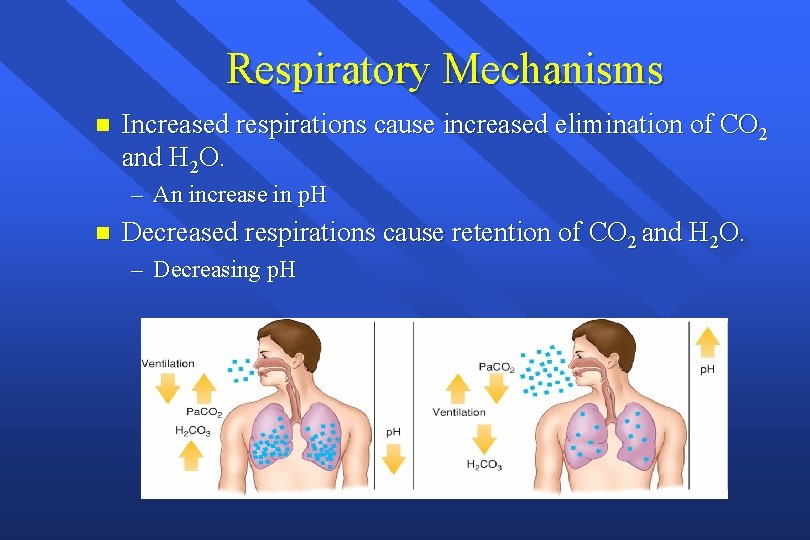
Respiratory Mechanisms n Increased respirations cause increased elimination of CO 2 and H 2 O. – An increase in p. H n Decreased respirations cause retention of CO 2 and H 2 O. – Decreasing p. H

Slowest onset and generally important in long term maintenance of acid-base balance (hours to days) n Function n – Kidneys retain bicarbonate, causing a decrease in H+ ions and an increase in p. H – Kidneys excrete bicarbonate, causing an increase in H+ ions and a decrease in p. H



Respiratory Acidosis n Caused by abnormal retention of CO 2 from impaired ventilation due to problems occurring in the lungs or respiratory center of the brain Respiration = CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 -


Respiratory Alkalosis Caused by increased respiration and excessive elimination of CO 2 The CO 2 level is decreased and the p. H is increased. Respiration = CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 -


ACID-BASE DERANGEMENTS (CONT. ) n Metabolic acidosis – Increased production of acids (lactic and pyruvic) – Can also result from diarrhea, vomiting, diabetes and medications (ASA) – p. H is decreased and CO 2 level is normal – Treatment is aimed at improving ventilations to eliminate CO 2 – Sodium bicarbonate with documented metabolic acidosis » Rarely administered during initial resuscitative strategies


ACID-BASE DERANGEMENTS (CONT. ) n Metabolic alkalosis – Caused by increased vomiting, overdosing on antiacids, diuretics – p. H is increased and CO 2 remains normal – Treatment is aimed at underlying cause n Combinations – Any combination of acid-base derangements – Only ABG can accurately diagnose problem


Thank you!
- Slides: 24