Acid base balance 341 Mohammed AlGhonaim MBBS FRCPC

Acid base balance 341 Mohammed Al-Ghonaim, MBBS, FRCPC, FACP

Objective • At the end of this tutorial you will be able to: – State the normal value for PH, PCO 2, HCO 3 – Interpret basic acid base disturbance – List common differential diagnosis for different acid base disorder

Steps in Acid-Base Analysis • Step 1: Acidemic or Alkalemic? • Step 2: Is the primary disturbance respiratory or metabolic? • Step 3: Is there appropriate compensation for the primary disorder? – Metabolic acidosis: PCO 2 = [1. 5 x (serum HCO 3)] + 8 (± 2) – Metabolic alkalosis: ↑PCO 2 = 0. 6 x ↑HCO 3 (± 2) – Respiratory acidosis: ↑PCO 2 10, ↑ HCO 3 by 1 (acute) or 3 -3. 5 (chronic) – Respiratory alkalosis: ↓PCO 2 10, ↓ HCO 3 by 2 (acute) or 4 -5 (chronic)

Steps in Acid-Base Analysis • Step 4: Is the respiratory disturbance acute or chronic? • Step 5: For a metabolic acidosis, is there an increased anion gap? – Anion gap = [Sodium] - ([Chloride] + [Bicarbonate]) Or AG = [Na+] - ([Cl-] + [HCO 3 -]). – Normal AG 8 -16 – Serum Osmolality = (2 x (Na + K)) + (BUN ) + (glucose )

Step 1: Acidemic or Alkalemic? • The p. H of the arterial blood gas measurement identifies the disorder as alkalemic or acidemic. • Normal arterial blood p. H = 7. 35 – 7. 45 – Acidemic: p. H < 7. 35 – Alkalemic: p. H > 7. 45 • Bicarbonate - (HCO 3): 22 - 26 m. Eq/liter • Partial pressure of carbon dioxide (Pa. CO 2): 35 - 45 mm. Hg

Step 2: Is the primary disturbance respiratory or metabolic? • To determine whether the disturbance affects primarily – The arterial Pa. CO 2 or – The serum HCO 3 -. • Respiratory disturbances alter the arterial Pa. CO 2 (normal value 35 -45) • Metabolic disturbances alter the serum HCO 3 - (normal value 22 -26)
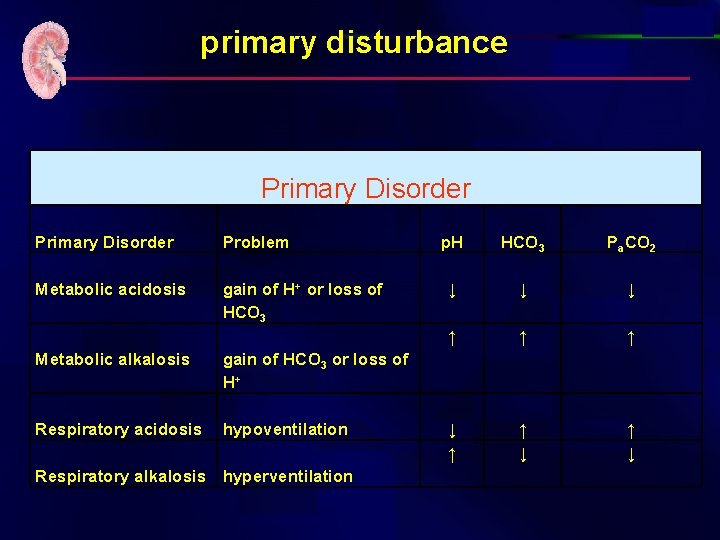
primary disturbance Primary Disorder Problem Metabolic acidosis gain of H+ or loss of HCO 3 Metabolic alkalosis gain of HCO 3 or loss of H+ Respiratory acidosis hypoventilation Respiratory alkalosis hyperventilation p. H HCO 3 Pa. CO 2 ↓ ↓ ↓ ↑ ↑ ↑ ↓ ↑ ↓

Case study: (1) • A 22 year-old woman presents with 4 hours of numbness in both hands and SOB • ABG: p. H 7. 48, p. CO 2 30 mm. Hg, p. O 2 86 mm. Hg • Na 140, Cl 110, HCO 3 22 • Step 1: Alkalosis • Step 2: Respiratory • Step 3: Acute. Drop in the p. CO 2 by 10 corresponds to a drop in the HCO 3 by 2 if acute

Respiratory Alkalosis – – – – Pain Drugs Sepsis Fever Thyrotoxicosis Pregnancy Overaggressive mechanical ventilation Hepatic failure Anxiety Hypoxemia Restrictive lung disease Severe congestive heart failure Pulmonary emboli

Respiratory acidosis • Is it acute or chronic? • Note that the PH is abnormal • Note the HCO 2 is with in normal • Remember: • Acute respiratory acidosis: – HCO 3 - increase by 1 m. Eq/l for every 10 mm. Hg increase in Pa. CO 2 • Chronic respiratory acidosis: – HCO 3 - increase by 3 -3. 5 m. Eq/l for every 10 mm. Hg increase in Pa. CO 2
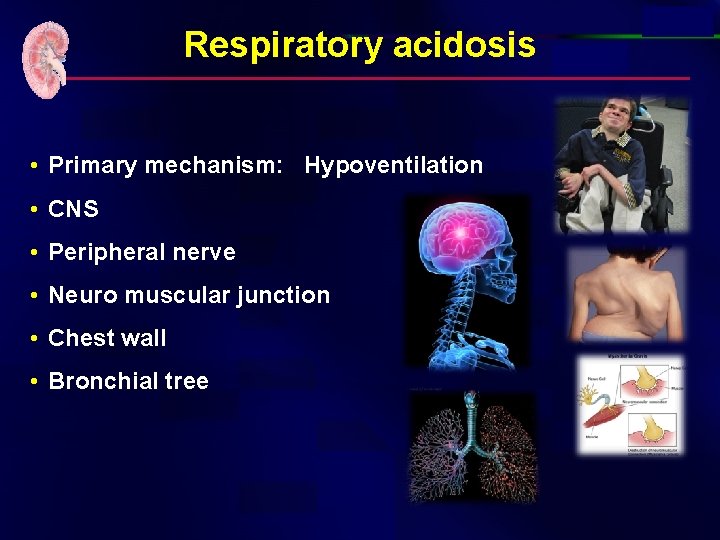
Respiratory acidosis • Primary mechanism: Hypoventilation • CNS • Peripheral nerve • Neuro muscular junction • Chest wall • Bronchial tree

Case study: (2) • p. H =7. 2, p. CO 2 = 60, HCO 2 = 24. • What it is the primary problem? Compensation? • Differential diagnosis? • Treatment ?

Case study: (3) • What do you expect the ABG in the following patients to be: • 24 years old male with acute SOB, and wheezes for 2 days. • Past hx: Bronchial asthma • 67 years old women, HTN, DM II, COPD presenting with cough and SOB
![Case study: (4) • p. H: 7. 25 • [HCO 3 -]: 20 m. Case study: (4) • p. H: 7. 25 • [HCO 3 -]: 20 m.](http://slidetodoc.com/presentation_image/0823124f41e155c0c25b958a4606787e/image-14.jpg)
Case study: (4) • p. H: 7. 25 • [HCO 3 -]: 20 m. Eq/L • Pa. CO 2: 52 mm. Hg • What it is the primary problem? Compensation? • Differential diagnosis?
![Case study: (5) • p. H: 7. 32 • [HCO 3 -]: 19 m. Case study: (5) • p. H: 7. 32 • [HCO 3 -]: 19 m.](http://slidetodoc.com/presentation_image/0823124f41e155c0c25b958a4606787e/image-15.jpg)
Case study: (5) • p. H: 7. 32 • [HCO 3 -]: 19 m. Eq/L • Pa. CO 2: 55 mm. Hg • What it is the primary problem? Compensation? • Differential diagnosis? • What other investigation you want to do?

Step 4: Is the respiratory disturbance acute or chronic? • Acute respiratory acidosis: – HCO 3 - increase by 1 m. Eq/l for every 10 mm. Hg increase in Pa. CO 2 • Chronic respiratory acidosis: – HCO 3 - increase by 3 -3. 5 m. Eq/l for every 10 mm. Hg increase in Pa. CO 2 • Acute respiratory alkalosis: – HCO 3 - decrease by 2 m. Eq/l for every 10 mm. Hg decrease in Pa. CO 2 • Chronic respiratory alkalosis: – HCO 3 - decrease by 4 -5 m. Eq/l for every 10 mm. Hg decrease in Pa. CO 2

Case study: (6) • A 70 year-old smoker presents with an acute onset of shortness of breath. • ABG: p. H 7. 30, p. CO 2 = 60 mm. Hg, p. O 2 60 mm. Hg • Na 135, Cl 100, HCO 3 30 • Step 1: Acidosis • Step 2: Respiratory • Step 3: Acute on chronic. p. CO 2 increased by 20, therefore the HCO 3 should increase by 2 if acute and 8 if chronic. Because the HCO 3 increased from 24 to 30 (6), an acute on chronic respiratory acidosis is present.
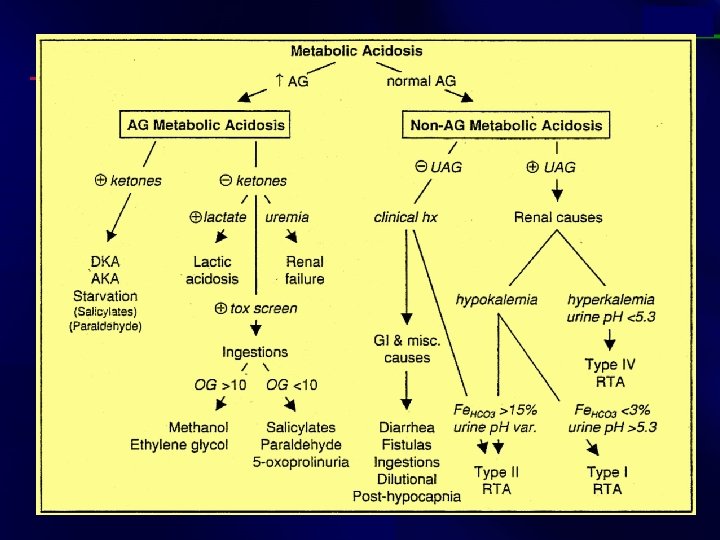

Step 5: For a metabolic acidosis, is there an increased anion gap? • Anion gap = [Sodium] - ([Chloride] + [Bicarbonate]) Or AG = [Na+] - ([Cl-] + [HCO 3 -]). • Normal AG 8 -16 • Serum Osmolality = (2 x (Na + K)) + (BUN ) + (glucose )

Case study: (7) • 32 year old man with depression and alcohol abuse presents with altered mental status. • ABG: p. H 6. 9, p. CO 2 29, p. O 2 100 • Na 140, Cl 101, HCO 3 5 • Step 1: Acidosis • Step 2: Metabolic • Step 3: p. CO 2 = 1. 5(HCO 3) + 8 = 15, but the patient’s p. CO 2 is higher than 15. Therefore, a respiratory acidosis is also present, possibly secondary to CNS depression. • Step 4: AG = 140 – (101 + 5) = 34

Increased anion gap metabolic acidosis • Methanol other alcohols, and ethylene glycol intoxication Uremia (renal failure) Lactic acidosis Ethanol Paraldehyde and other drugs Aspirin Ketones (starvation, alcoholic and diabetic ketoacidosis)

Case study: (8) • A 68 year old man who recently took antibiotics for a skin infection presents with 10 episodes of watery diarrhea per day for the last 5 days. • ABG: p. H 7. 34, p. CO 2 34, p. O 2 80 • Na 135, Cl 108, HCO 3 18 • Step 1: Acidosis • Step 2: Metabolic • Step 3: p. CO 2 = 1. 5(HCO 3) + 8 = 35 • Step 4: AG = 135 – (108 + 18) = 9 • NAGMA (non anion gap metabolic acidosis)

Is the respiratory system compensating adequately for a metabolic disturbance • Metabolic acidosis: – PCO 2 decreases by 1 mm. Hg for every 1 m. Eq/l decrease in HCO 3 OR – PCO 2 = [1. 5 x (serum HCO 3)] + 8 (± 2) • Metabolic alkalosis: • PCO 2 increases by 0. 6 mm. Hg for every 1 m. Eq/l increases in HCO 3 OR • ↑PCO 2 = 0. 6 x ↑HCO 3 (± 2)

Case study: (9) • p. H =7. 1, p. CO 2 = 42, HCO 3 - = 12.

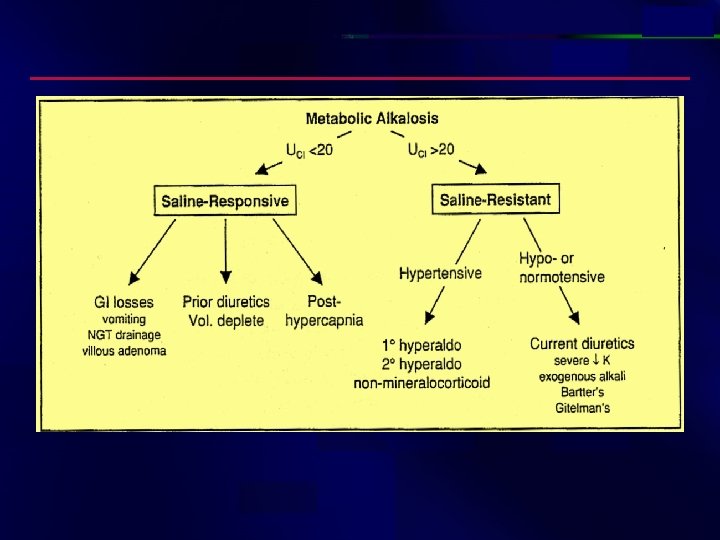
Case study: (10) • HCO 3= 39; p. CO 2 = 40; p. H = 7. 6; K = 3. 0. The patient has been vomiting. • p. H = 7. 6 and K = 3. 0

Case study: (11) • p. H =7. 58, p. CO 2 = 27, HCO 3 - = 24. • What is the primary cause of the condition? • Hyperventilation - excess loss of CO 2 gas

Case study: (12) • 56 yo M with Hx of COPD is admitted with 1 -wk Hx of dyspnea, productive cough and diarrhea (Na) 125, (Cl) 103 , (BUN) 42, (Glucose) 100, (K) 3. 5, (HCO 3 -) 10, (Creat) 1. 4 • ABG 7. 14 p. CO 2 30 p. O 2 50 • What is the predominant acid base disorder ? Acidosis

Case study: (12) continue • What p. CO 2 is expected with normal respiratory compensation ? 40 – (1. 2 * (24 -10)) = 23. 2, this is not full compensation b/c p. CO 2 is 30 – indicates an underlying primary respiratory acidosis, suggested by the Hx of COPD, dyspnea, and productive cough (lungs not able to appropriately compensate) • What is the Anion Gap ? 125 – (103+10) = 12 – normal AG etiology is either diarrhea or RTA – most likely diarrhea b/c of the history

Case study: (13) • 32 y/o male present w/ 2 d Hx of intractable vomiting. ; p. H 7. 51, p. CO 2 41 Na 132, Cl 90 32 K 3. 4 HCo 2= 33 creatinine 1. 6 • What is the predominant acid-base disorder? Alkalosis

Etiologies of Metabolic Alkalosis Saline – responsiv e GI loss of H+ : vomiting, NGT drainage, villous adenoma Diuretic use posthypercapina Saline – resistant Hypertensive (mineralocorticoid excess) 10 hyperaldosteronism (eg. Conn's) 20 hyperaldosteronism (eg, renovascular dis. Renninsecreting tumor) Non-aldo (eg. Cushing's, Liddle's, exogenousmineralocorticoids) Normotensive Severe hypokalemia Exogenous alkali load Bartter's syndrome, Gitelman's syndrome

Case study: (13) contunue • What p. CO 2 is expected w/ normal respiratory compensation? = 40 + (32 – 24) * (~ 0. 6 0. 7) = 44. 8 45. 6 mm. Hg; since the measured p. CO 2 < 44. 8 45. 6, there is also a primary respiratory alkalosis (inappropriate hyperventilation) • Tx: Isotonic saline to correct for volume depletion –

Case study: (14) • A 58 - year old man presents to the Emergency Department with abdominal pain and hypotension. Investigation reveal the following: • Na 140 PH 6. 8 PCO 2 K 4 36 Cl 90 HCO 3 = 5 PO 2 7 • Analyze the acid-base disorder(s) seen in the patient. •

Case study: (14) continue • Acidemia • Anion Gap = 45

Case study: (14) • A 42 -year-old man to the ER after he was found unconscious. • O/E – – – 120/80, HR 110/min, RR 28/min, and Temp 37°C. Unresponsive Pupils minimally reactive to light Bibasilar crackles Deep tendon reflexes were brisk and symmetric

Case study: (14) continue • Lab: • p. H 7. 1 • Pa. CO 2, 35 mm Hg • Pa. O 2, 90 mm Hg at room air • Na 145 m. Eq/L; K 5 m. Eq/L; Cl 97 m. Eq/L; HCO 3 12 m. Eq/L • blood urea nitrogen, 10 mmol/l; creatinine, 140 umol/l; and glucose, 6. 0 mmol/l. •

Case study: (14) continue • Acidemic the change in Pa. CO 2 equaled about 14 mm Hg (ie, 1. 2 x 12). • Therefore, the expected Pa. CO 2 was 26 mm Hg (ie, 40 14 ). • the measured Pa. CO 2 of 35 mm Hg was much higher than expected

Case 11 continue • Concomitant respiratory acidosis • AG= 145 – (97+12) = 36 • Measured osmolality= 350 • Calculated osmolality = (2 x 145)+6+10= 306 • Osmole Gap=44 • Causes of increased Gap: – Methanol – Ethylene glycol – Ethanol – Isopropyl alcohol

Case study: (14) continue – lactate, 1 m. Eq/L – ketones, negative – Salicylate, negative – Urinalysis showed calcium oxalate crystals – +ve ethylene glycol • Final Dx – High AG Met acidosis – Ethylene Glycol – Respiratory Acidosis – Decreased level of consciousness – Metabolic alkalosis due to vomiting

Summary • First, does the patient have an acidosis or an alkalosis – Look at the p. H • Second, what is the primary problem – metabolic or respiratory – Look at the p. CO 2 – If the p. CO 2 change is in the opposite direction of the p. H change, the primary problem is respiratory

Summary • Third, is there any compensation by the patient - do the calculations – For a primary respiratory problem, is the p. H change completely accounted for by the change in p. CO 2 • if yes, then there is no metabolic compensation • if not, then there is either partial compensation or concomitant metabolic problem
- Slides: 42