Acid and Base Review Game Name the Acid

Acid and Base Review Game
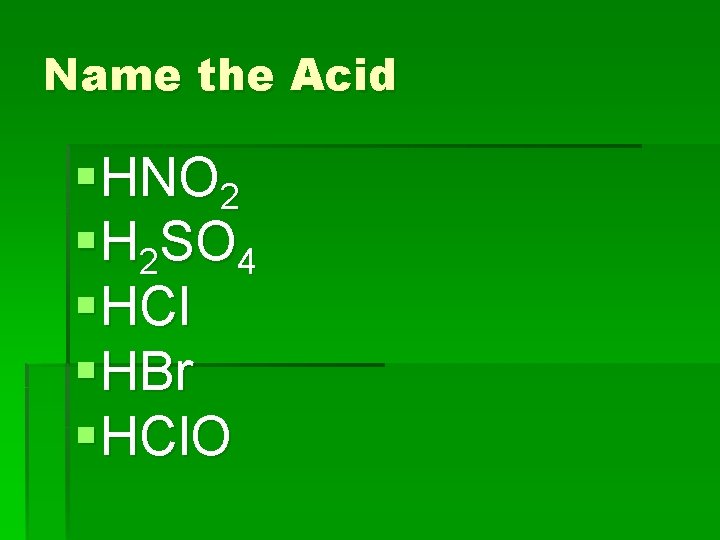
Name the Acid § HNO 2 § H 2 SO 4 § HCl § HBr § HCl. O
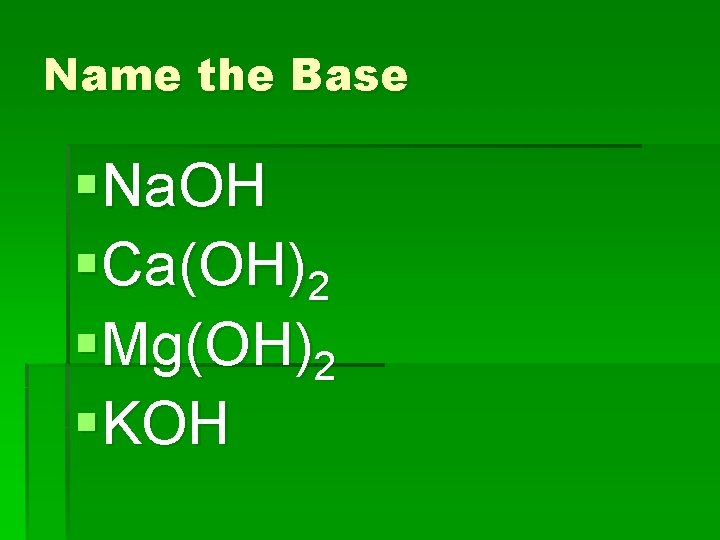
Name the Base §Na. OH §Ca(OH)2 §Mg(OH)2 §KOH

Write the Formula §Nitric Acid §Hydroiodidic Acid §Phosphoric Acid §Acetic Acid

Write the Formula §Sodium carbonate §Barium hydroxide §Lithium hydroxide

Arrhenius §What is the definition of an acid? §What is the definition of a base?
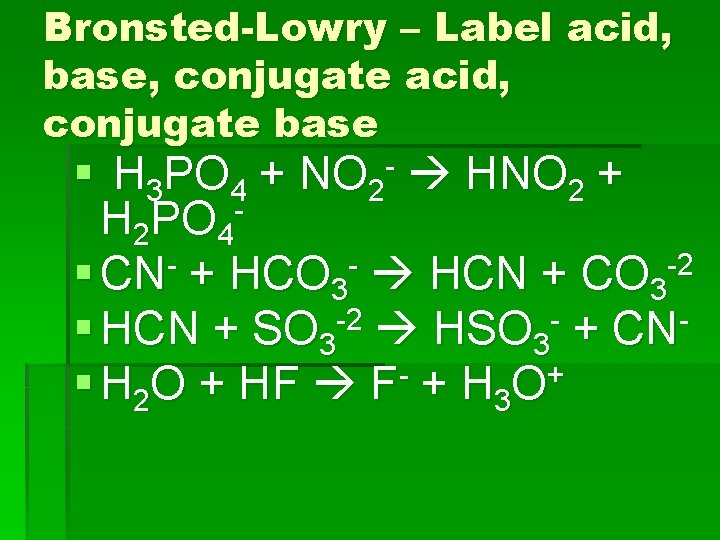
Bronsted-Lowry – Label acid, base, conjugate acid, conjugate base § H 3 PO 4 + HNO 2 + H 2 PO 4 -2 § CN + HCO 3 HCN + CO 3 § HCN + SO 3 -2 HSO 3 - + CN§ H 2 O + HF F- + H 3 O+ NO 2

p. H § If a soap has a hydrogen ion concentration of -6 2 x 10 M, what is the p. H of the solution? The p. OH? Is it an acid or a base?

p. H § What is the hydrogen ion concentration and hydroxide ion concentration of a solution with a p. H of 2. 3? Is the substance an acid or base?

p. H § What is the hydrogen ion concentration of a sample of phosphoric acid that has a p. H of 4. 9? What is the concentration of phosphoric acid?

Neutralization Reaction § What is the neutralization reaction for nitrous acid reacting with potassium hydroxide?

Neutralization Reaction § Write the complete balanced equation for the neutralization of phosphoric acid with calcium hydroxide

Concentration § How many grams of copper sulfate pentahydrate will be needed to make 75 m. L of a 0. 250 M solution?

Concentration § How many kilograms of sucrose C 12 H 22 O 11 will be needed to make 3. 50 L of a 1. 15 M solution?

Concentration § What is the molar concentration of a solution that contains 0. 0750 moles of Na. HCO 3 in a volume of 115 m. L

Concentration § A solution has 3. 0 moles of solute in 2 L of solution, what is its molar concentration? How many moles would there be in 350 m. L of solution?

Making Solution § Describe in your own words how you would prepare 1. 00 L of a 0. 85 M solution of formic acid HCO 2 H?

Molarity § What is the molarity of a sulfuric acid solution which contains 5. 4 grams of sulfuric acid in 250 m. L of water? What is the normality?

Molarity § What is the molarity of a potassium hydroxide solution which contains 0. 94 moles of potassium hydroxide in 450 m. L of water? What is the normality?

Titration § In the titration of 35 m. L of liquid drain cleaner containing Na. OH, 50 m. L of 0. 4 M HCl must be added to reach the equivalence point. What is the molarity of the base in the cleaner?

Titration § Calculate how many milliliters of 0. 25 M Ba(OH)2 must be added to titrate 46 m. L of 0. 40 M HCl. O 4

Titration § A 15. 5 m. L sample of 0. 215 M KOH was titrated with an acetic acid solution, It took 21. 2 m. L of the acid to reach the equivalence point. What is the molarity of the acetic acid?

Titration § A 20 m. L sample of an HCl solution was titrated with 27. 4 m. L of a standard solution of Ba(OH)2. The concetnration of the standard is 0. 0154 M. What is the molarity of the HCl?

Buffer §What is a buffer?

Buffer § What do buffers do? Where can you find them?
- Slides: 25