Acid and Base Reactions Chapter 4 Acids Substances

Acid and Base Reactions Chapter 4

Acids: • Substances that increase the concentration of H+ when dissolved in water (Arrhenius). • Proton donors (Brønsted–Lowry).

Acids There are only seven strong acids: • • Hydrochloric (HCl) Hydrobromic (HBr) Hydroiodic (HI) Nitric (HNO 3) Sulfuric (H 2 SO 4) Chloric (HCl. O 3) Perchloric (HCl. O 4)

Bases: • Substances that increase the concentration of OH− when dissolved in water (Arrhenius). • Proton acceptors (Brønsted–Lowry).

Bases The strong bases are the soluble salts of hydroxide ion: • • Alkali metals Calcium Strontium Barium

Acid-Base Reactions In an acid-base reaction, the acid donates a proton (H+) to the base.

Neutralization Reactions Generally, when solutions of an acid and a base are combined, the products are a salt and water. HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l)

Neutralization Reactions When a strong acid reacts with a strong base, the net ionic equation is… HCl (aq) + Na. OH (aq) Na. Cl (aq) + H 2 O (l) H+ (aq) + Cl- (aq) + Na+ (aq) + OH-(aq) Na+ (aq) + Cl- (aq) + H 2 O (l) H+ (aq) + Cl- (aq) + Na+ (aq) + OH- (aq) Na+ (aq) + Cl- (aq) + H 2 O (l)
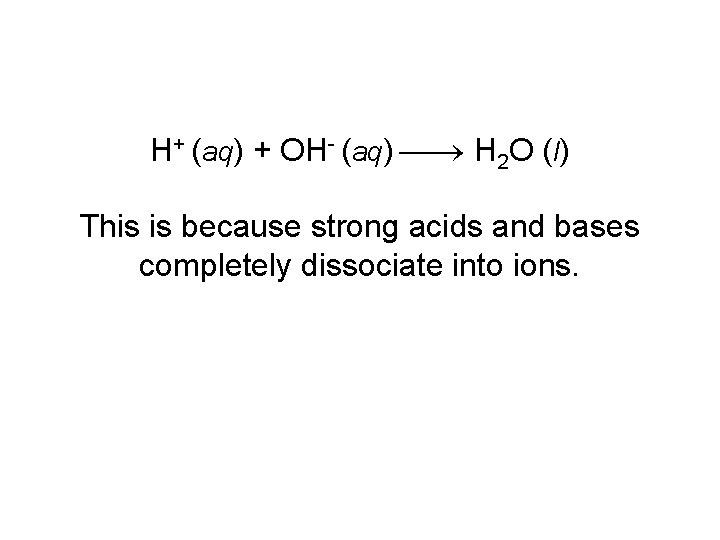
H+ (aq) + OH- (aq) H 2 O (l) This is because strong acids and bases completely dissociate into ions.

Reactions involving weak acids or bases • Since weak acids and bases are weak electrolytes (they only partially dissociate in solution) they will be written as a compound on the reactant side Example: • HF (aq) + Na. OH (aq) H 2 O (l) + Na. F (aq) • NH 3 (aq) + HI (aq) NH 4 I (aq)

Gas-Forming Reactions • These metathesis reactions do not give the product expected. • The expected product decomposes to give a gaseous product (CO 2 or SO 2). – Typically happens when carbonate or sulfite (SO 3) are reactants Ca. CO 3 (s) + HCl (aq) Ca. Cl 2 (aq) + CO 2 (g) + H 2 O (l) Na. HCO 3 (aq) + HBr (aq) Na. Br (aq) + CO 2 (g) + H 2 O (l) Sr. SO 3 (s) + 2 HI (aq) Sr. I 2 (aq) + SO 2 (g) + H 2 O (l)
- Slides: 11