Acid and Base Balance and Imbalance 1 p

Acid and Base Balance and Imbalance 1
![p. H Review • • • p. H = - log [H+] H+ is p. H Review • • • p. H = - log [H+] H+ is](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-2.jpg)
p. H Review • • • p. H = - log [H+] H+ is really a proton Range is from 0 - 14 If [H+] is high, the solution is acidic; p. H < 7 If [H+] is low, the solution is basic or alkaline ; p. H > 7 2

• Acids are H+ donors. • Bases are H+ acceptors, or give up OH- in solution. • Acids and bases can be: – Strong – dissociate completely in solution • HCl, Na. OH – Weak – dissociate only partially in solution • Lactic acid, carbonic acid 3

The Body and p. H • • • Homeostasis of p. H is tightly controlled Extracellular fluid = 7. 4 Blood = 7. 35 – 7. 45 < 6. 8 or > 8. 0 death occurs Acidosis (acidemia) below 7. 35 Alkalosis (alkalemia) above 7. 45 4
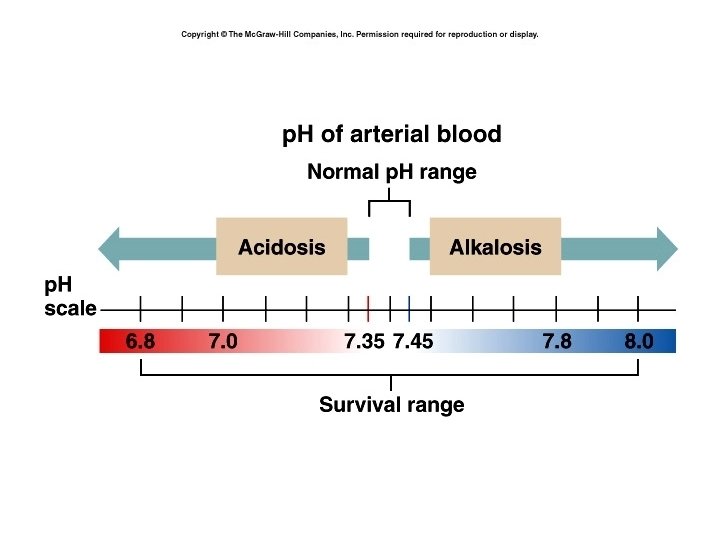
5

Small changes in p. H can produce major disturbances • Most enzymes function only with narrow p. H ranges • Acid-base balance can also affect electrolytes (Na+, K+, Cl-) • Can also affect hormones 6
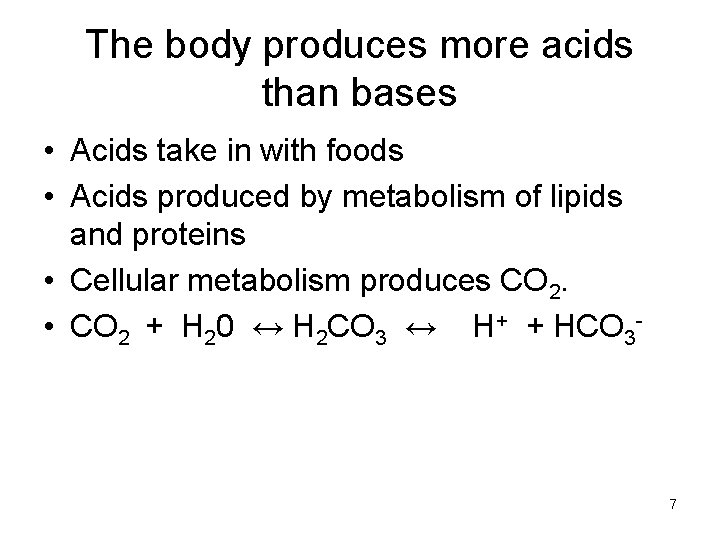
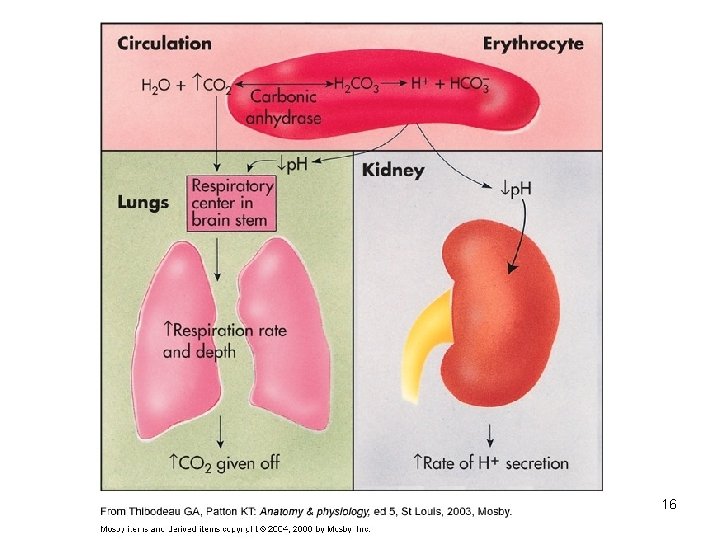
The body produces more acids than bases • Acids take in with foods • Acids produced by metabolism of lipids and proteins • Cellular metabolism produces CO 2. • CO 2 + H 20 ↔ H 2 CO 3 ↔ H+ + HCO 3 - 7

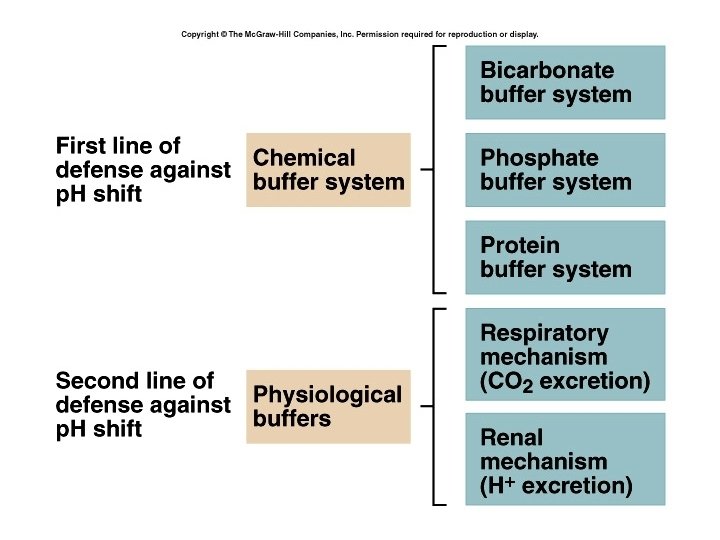
Control of Acids 1. Buffer systems Take up H+ or release H+ as conditions change Buffer pairs – weak acid and a base Exchange a strong acid or base for a weak one Results in a much smaller p. H change 8

Bicarbonate buffer • Sodium Bicarbonate (Na. HCO 3) and carbonic acid (H 2 CO 3) • Maintain a 20: 1 ratio : HCO 3 - : H 2 CO 3 HCl + Na. HCO 3 ↔ H 2 CO 3 + Na. Cl Na. OH + H 2 CO 3 ↔ Na. HCO 3 + H 2 O 9
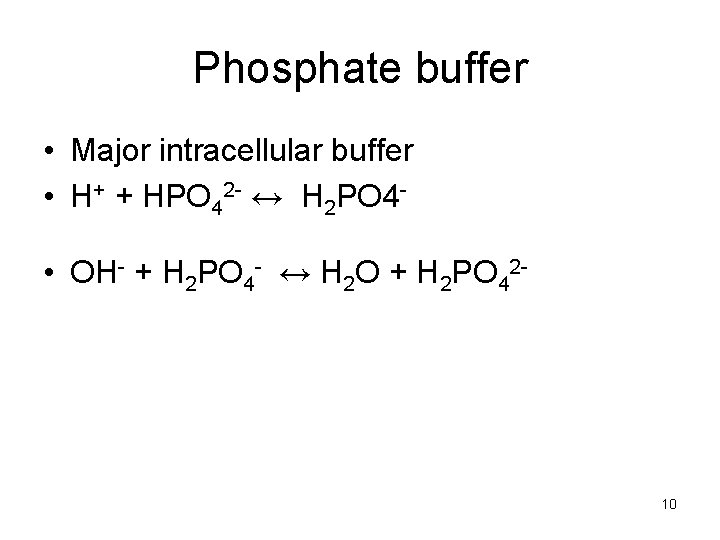
Phosphate buffer • Major intracellular buffer • H+ + HPO 42 - ↔ H 2 PO 4 • OH- + H 2 PO 4 - ↔ H 2 O + H 2 PO 42 - 10

Protein Buffers • • Includes hemoglobin, work in blood and ISF Carboxyl group gives up H+ Amino Group accepts H+ Side chains that can buffer H+ are present on 27 amino acids. 11

2. Respiratory mechanisms • Exhalation of carbon dioxide • Powerful, but only works with volatile acids • Doesn’t affect fixed acids like lactic acid • CO 2 + H 20 ↔ H 2 CO 3 ↔ H+ + HCO 3 • Body p. H can be adjusted by changing rate and depth of breathing 12

3. Kidney excretion • • • Can eliminate large amounts of acid Can also excrete base Can conserve and produce bicarb ions Most effective regulator of p. H If kidneys fail, p. H balance fails 13

Rates of correction • Buffers function almost instantaneously • Respiratory mechanisms take several minutes to hours • Renal mechanisms may take several hours to days 14
15
16

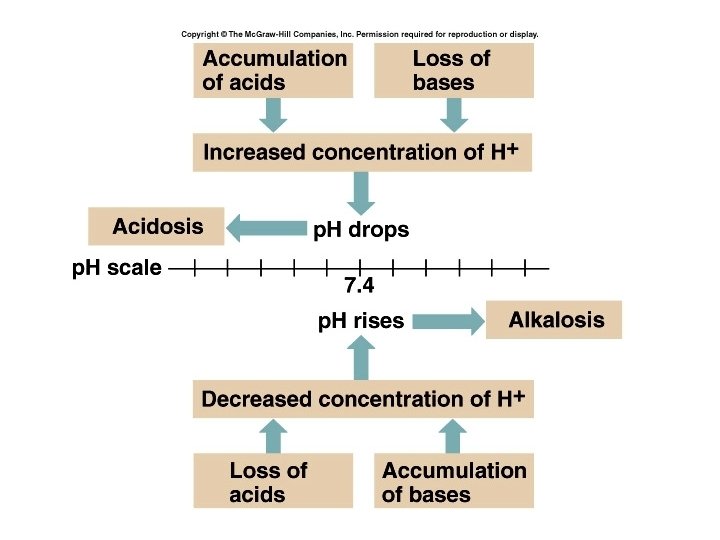
Acid-Base Imbalances • p. H< 7. 35 acidosis • p. H > 7. 45 alkalosis • The body response to acid-base imbalance is called compensation • May be complete if brought back within normal limits • Partial compensation if range is still outside norms. 17

Compensation • If underlying problem is metabolic, hyperventilation or hypoventilation can help : respiratory compensation. • If problem is respiratory, renal mechanisms can bring about metabolic compensation. 18

Acidosis • Principal effect of acidosis is depression of the CNS through ↓ in synaptic transmission. • Generalized weakness • Deranged CNS function the greatest threat • Severe acidosis causes – Disorientation – coma – death 19

Alkalosis • Alkalosis causes over excitability of the central and peripheral nervous systems. • Numbness • Lightheadedness • It can cause : – Nervousness – muscle spasms or tetany – Convulsions – Loss of consciousness – Death 20
21

Respiratory Acidosis • Carbonic acid excess caused by blood levels of CO 2 above 45 mm Hg. • Hypercapnia – high levels of CO 2 in blood • Chronic conditions: – Depression of respiratory center in brain that controls breathing rate – drugs or head trauma – Paralysis of respiratory or chest muscles – Emphysema 22

Respiratory Acidosis • Acute conditons: – Adult Respiratory Distress Syndrome – Pulmonary edema – Pneumothorax 23

Compensation for Respiratory Acidosis • Kidneys eliminate hydrogen ion and retain bicarbonate ion 24

Signs and Symptoms of Respiratory Acidosis • • • Breathlessness Restlessness Lethargy and disorientation Tremors, convulsions, coma Respiratory rate rapid, then gradually depressed • Skin warm and flushed due to vasodilation caused by excess CO 2 25

Treatment of Respiratory Acidosis • Restore ventilation • IV lactate solution • Treat underlying dysfunction or disease 26

Respiratory Alkalosis • • Carbonic acid deficit p. CO 2 less than 35 mm Hg (hypocapnea) Most common acid-base imbalance Primary cause is hyperventilation 27

Respiratory Alkalosis • Conditions that stimulate respiratory center: – Oxygen deficiency at high altitudes – Pulmonary disease and Congestive heart failure – caused by hypoxia – Acute anxiety – Fever, anemia – Early salicylate intoxication – Cirrhosis – Gram-negative sepsis 28

Compensation of Respiratory Alkalosis • Kidneys conserve hydrogen ion • Excrete bicarbonate ion 29

Treatment of Respiratory Alkalosis • Treat underlying cause • Breathe into a paper bag • IV Chloride containing solution – Cl- ions replace lost bicarbonate ions 30

Metabolic Acidosis • Bicarbonate deficit - blood concentrations of bicarb drop below 22 m. Eq/L • Causes: – Loss of bicarbonate through diarrhea or renal dysfunction – Accumulation of acids (lactic acid or ketones) – Failure of kidneys to excrete H+ 31

Symptoms of Metabolic Acidosis • • Headache, lethargy Nausea, vomiting, diarrhea Coma Death 32

Compensation for Metabolic Acidosis • Increased ventilation • Renal excretion of hydrogen ions if possible • K+ exchanges with excess H+ in ECF • ( H+ into cells, K+ out of cells) 33
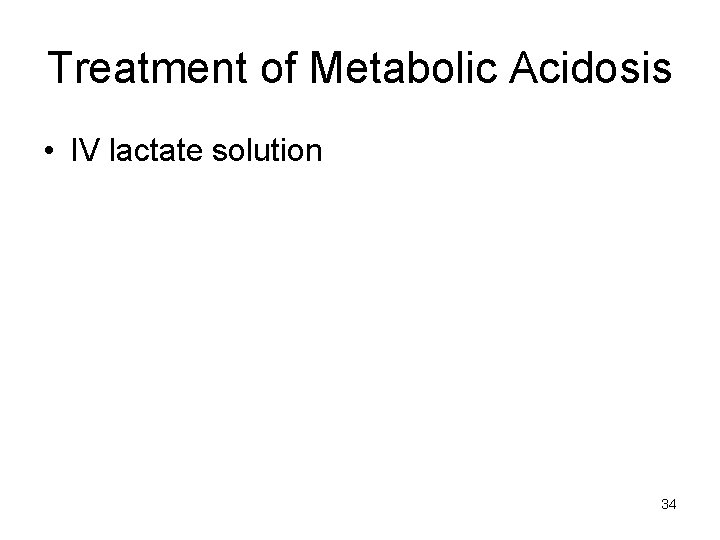
Treatment of Metabolic Acidosis • IV lactate solution 34

Metabolic Alkalosis • Bicarbonate excess - concentration in blood is greater than 26 m. Eq/L • Causes: – Excess vomiting = loss of stomach acid – Excessive use of alkaline drugs – Certain diuretics – Endocrine disorders – Heavy ingestion of antacids – Severe dehydration 35

Compensation for Metabolic Alkalosis • Alkalosis most commonly occurs with renal dysfunction, so can’t count on kidneys • Respiratory compensation difficult – hypoventilation limited by hypoxia 36

Symptoms of Metabolic Alkalosis • • • Respiration slow and shallow Hyperactive reflexes ; tetany Often related to depletion of electrolytes Atrial tachycardia Dysrhythmias 37

Treatment of Metabolic Alkalosis • Electrolytes to replace those lost • IV chloride containing solution • Treat underlying disorder 38

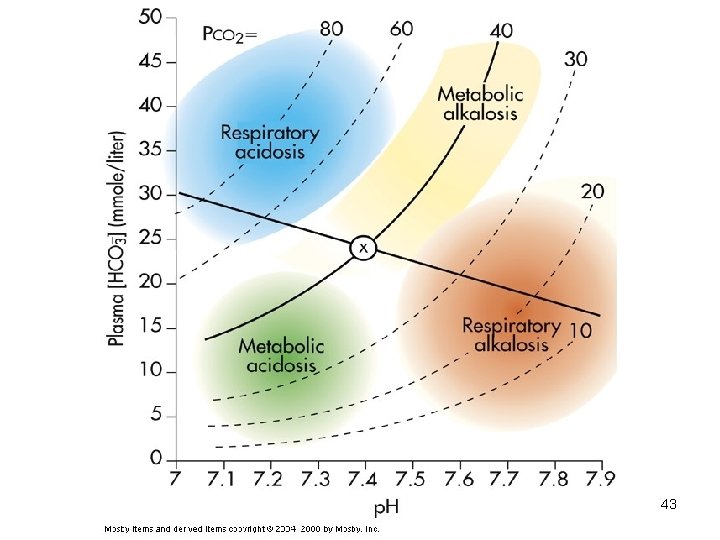
Diagnosis of Acid-Base Imbalances 1. Note whether the p. H is low (acidosis) or high (alkalosis) 2. Decide which value, p. CO 2 or HCO 3 - , is outside the normal range and could be the cause of the problem. If the cause is a change in p. CO 2, the problem is respiratory. If the cause is HCO 3 - the problem is metabolic. 39

3. Look at the value that doesn’t correspond to the observed p. H change. If it is inside the normal range, there is no compensation occurring. If it is outside the normal range, the body is partially compensating for the problem. 40

Example • A patient is in intensive care because he suffered a severe myocardial infarction 3 days ago. The lab reports the following values from an arterial blood sample: – p. H 7. 3 – HCO 3 - = 20 m. Eq / L ( 22 - 26) – p. CO 2 = 32 mm Hg (35 - 45) 41

Diagnosis • Metabolic acidosis • With compensation 42
43

• • • • Blood Sodium 135 -145 m. Eq/L Potassium 3. 5 -5. 0 m. Eql. L Chloride 95 -105 m. Eq/L Bicarbonate 24 -26 m. Eq/L Osmolality 280 -295 m. Eq. L Osmolal gap < 10 m. Osm/L Anion gap 9 -16 m. Eql. L Urea nitrogen 10 -20 mgldl Arterial blood gas analysis p. H 7. 35 -7. 45 PCOZ 35 -45 mm Hg Poz 90 -100 mm Hg (declines with age) 44
![Disorder Expected Compensation Metabolic acidosis PCO 2 = 1. 5 × [HCO 3 -] Disorder Expected Compensation Metabolic acidosis PCO 2 = 1. 5 × [HCO 3 -]](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-45.jpg)
Disorder Expected Compensation Metabolic acidosis PCO 2 = 1. 5 × [HCO 3 -] + 8 ± 2 Metabolic alkalosis PCO 2 increases by 7 mm Hg for each 10 m. Eq/L increase in the serum [HCO 3 -] Respiratory acidosis Acute [HCO 3 -] increases by 1 for each 10 mm Hg increase in the PCO 2 Chronic [HCO 3 -] increases by 3. 5 for each 10 mm Hg increase in the PCO 2 Respiratory alkalosis Acute [HCO 3 -] falls by 2 for each 10 mm Hg decrease in the PCO 2 Chronic [HCO 3 -] falls by 4 for each 10 mm Hg decrease in the PCO 2 45

Formulas Women: Total body water (TBW) =. 5 X Body weight (kg) Men: Total body water (TBW) =. 6 X Body weight (kg) Osmolal gap = OSM, measured) - OSM, ca~culatcd) Anion gap (AG) = UA - UC = [Na'] - ([CI-] + [HCO, -]) 46

• OSM GAP = OSM(measured) - OSM(cal) • Values of greater than 10 m. Osm/L are abnormal and suggest the presence of an exogenous substance • AG is normally 9 -16 m. Fq/L. 47

Common Causes of Metabolic Acidosis Increased Anion Gap LA MUD PIE Increased anion gap (Mnemonic) Diabetic ketoacidosis Lactate (sepsis, ischemia, etc. ) L-lactic acidosis Aspirin D-lactic acidosis Methanol Alcoholic ketoacidosis Uremic acidosis (advanced renal failure) Uremia Diabetic ketoacidosis Salicylate intoxication Paraldehyde, Propylene glycol Ethylene glycol intoxication Isopropyl alcohol, INH Methanol intoxication Ethylene glycol (antifreeze, low calcium) Paraldehyde intoxication Normal anion gap Mild to moderate renal failure Gastrointestinal loss of HCO 3 - (acute diarrhea) Type I (distal) renal tubular acidosis Type I 1 (proximal) renal tubular acidosis Qpe IV renal tubular acidosis Dilutional acidosis Treatment of diabetic ketoacidosis (ketones lost in urine) 48

Diabetic ketoacidosis (DKA) Patients with severe diabetic ketoacidosis typically present with High anion gap metabolic acidosis Severe acidemia (p. H < 7. 15) Hyperglycemia ECFV depletion Potassium depletion despite serum potassium concentrations that may be normal or elevated. 49

Ethylene glycol intoxication may result from drinking antifreeze or radiator fluid. There is usually, but not always, a background of chronic alcoholism. Ethylene glycol is metabolized by alcohol dehydrogenase to a number of very toxic compounds which produce: A high anion gap acidosis. Acute central nervous system dysfunction: ataxia, confusion, seizures, and coma Acute renal failure Calcium oxalate crystals in the urine (one of the toxic compounds is oxalic acid) 50

A 40 -year-old man is admitted with shallow, rapid respirations. His serum chemistries are: sodium 142 rn. Eq/L, potassium 3. 6 rn. Eq/L, chloride 100 m. Eqn, bicarbonate 12 m. Eq/L. Arterial blood gas: p. H 7. 28, PCO 26, HC 0312. What is your differential diagnosis? 51

A 60 -year-old alcoholic woman is admitted with rapid respiration, tachycardia, and a blood pressure of 90/160. Her chemistries are: sodium 142 m. Eq. L, potassium 3. 6 m. Eq. L, chloride 100 m. Eq. L, bicarbonate 12 m. Eq. L, glucose 180 mgldl, BUN 28 mgldl. Arterial blood gas: p. H 7. 28, PCO 2~6, HC 03 - 12. What is your differential diagnosis, and what do you do to make a diagnosis? 52

A 50 -year-old man is admitted with rapid respiration, tachycardia, and a blood pressure of 90160. His chemistries are: sodium 142 m. Eqn, potassium 3. 6 m. Eqn, chloride 100 m. Eq. L, bicarbonate 12 m. Eqn, glucose 180 mgldl, BUN 28 mgldl. Arterial blood gas: p. H 7. 28, Pcoz 26, HC 0312. Urinalysis: calcium oxalate crystals. What is your differential diagnosis, and what do you do to make a diagnosis? 53

A 15 -year-old boy is admitted with severe diarrhea and the following laboratory data: sodium 142 m. Eq/L, potassium 3. 6 m. Eqn, chloride 1 15 m. Eq/L, bicarbonate 12 m. Eq. L, creatinine 1. 1 mg/dl. Urine ketones negative. Arterial blood gas: p. H 7. 12, PCO 3~8, HC 03 - 12. What acid-base disorders are present? 54

Causes of Metabolic Alkalosis ECFV depletion--chloride depletion syndrome (saline-responsive) Vomiting/nasogastric suction Diuretic therapy Posth ypercapnea Chronic diarrheallaxative abuse Severe potassium depletion from any cause (saline-resistant) Mineralocorticoid excess syndromes (saline-resistant) Primary hyperaldosteronism Cushing's syndrome Ectopic ACTH Secondary hyperaldosteronism Renovascular disease Malignant hypertension Congestive heart failure (with diuretic therapy) Cirrhosis (with diuretic therapy) Gitelman's syndrome (saline-resistant) Bartter's syndrome (saline-resistant) 55
![PCO 2= 40 +. 7 X ([HC 03 (measured)] - [HC 03 (normal)]) 56 PCO 2= 40 +. 7 X ([HC 03 (measured)] - [HC 03 (normal)]) 56](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-56.jpg)
PCO 2= 40 +. 7 X ([HC 03 (measured)] - [HC 03 (normal)]) 56

A 40 -year-old man is admitted with the following chemistries: sodium I 140 m. Eq/L, chloride 86 m. Eq/L, bicarbonate 40 m. Eq/L, potassium 3. 0 m. Eq/L, glucose 120 mg/dl, BUN 32 mg/dl, Cr 1. 4 mg/dl. Arterial blood gas: p. H 7. 52, P 02 --51 mrn Hg, HCO 3 - 40 m. Eq/L. What is your general approach to this patient? 57
![Respiratory alkalosis: Acute: [HCOs-] decreases by 2 m. Eqn for every 10 mm Hg Respiratory alkalosis: Acute: [HCOs-] decreases by 2 m. Eqn for every 10 mm Hg](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-58.jpg)
Respiratory alkalosis: Acute: [HCOs-] decreases by 2 m. Eqn for every 10 mm Hg decrease in PCOZ Chronic: [HC 03 -] decreases by 5 m. Eq. L for every 10 mm Hg decrease in PC 02 Step 3: Calculate the anion gap. AG = [Na+] - ([Cl-] + [HCOs-I) The normal AG is 9 -16 m. Eq/L. If AG > 20 m. Eqn, high AG acidosis is probably present. If AG > 30 m. Eq/L, high AG acidosis is almost certainly present. 58
![A patient presents with: p. H 7. 15, calculated [HC 03 -] 6 m. A patient presents with: p. H 7. 15, calculated [HC 03 -] 6 m.](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-59.jpg)
A patient presents with: p. H 7. 15, calculated [HC 03 -] 6 m. Eq/L, PCOZ 1 8 mm Hg, sodium 135 m. Eq/L, chloride 114 m. Eq/L, , potassium 4. 5 m. Eq. L, serum [HC 03 -] 6 m. Eq/L. 59

A patient presents with: p. H 7. 49, [HC 03 - 35, PCO 2 -- 4 8, AG 16. 60
![A patient presents with: p. H 7. 68, PCO 2 35, [HC 03 -] A patient presents with: p. H 7. 68, PCO 2 35, [HC 03 -]](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-61.jpg)
A patient presents with: p. H 7. 68, PCO 2 35, [HC 03 -] 40, AG 18. 61

A previously well patient presents with 30 minutes of respiratory distress and p. H 7. 26, Pc 02 60, [HC 03 -] 26, AG 14. 62
![Apatient presents with: p. H 7. 45, PCOZ 65 , [HC 03 -] 44, Apatient presents with: p. H 7. 45, PCOZ 65 , [HC 03 -] 44,](http://slidetodoc.com/presentation_image/539f6b31e4f1d94fc7ca9c2557916a3d/image-63.jpg)
Apatient presents with: p. H 7. 45, PCOZ 65 , [HC 03 -] 44, AG 14. Short of breath for 3 days. 63

A patient presents with diabetic ketoacidosis: p. H 6. 95, PCOZ 28, [HC 03 -] 6, AG 32. 64

A patient with recurrent episodes of small bowel obstruction presents with severe abdominal pain and vomiting: p. H 7. 33, PCO 3~5 , [HC 03 -] 18, AG 33. Urine dipstick negative for ketones. The blood pressure is 82154 and the heart rate 116. 65

A 21 -year-old diabetic patient presents with vomiting and p. H 7. 75, PCO 2~4 , [HC 03 -] 32, AG 30. The urine is strongly positive for ketones and serum ketones are strongly positive. 66

A 50 -year-old 70 kg alcoholic man presents with 4 days of nausea, vomiting, and mild abdominal pain following a week-long drinking binge. He is unable to take anything by mouth. His mucous membranes are dry, and his vital signs reveal an orthostatic blood pressure drop with a rise in pulse. The following laboratory data are obtained: Na 134 m. Eqn, K 3. 1 m. Eq/L, [HC 03 -] 20 m. Eq/L, C 1 80 m. Eq/L, glucose 86 mg/dl, BUN 52 mg/dl, Cr 1. 4 mg/dl, amylase pending, serum ketones: high positive reading. ABG: p. H 7. 32, PCO 4~0 mm Hg, [HC 03 -] 20 m. Eq/L. Urine sodium 7 m. Eq. L (low). Urine ketones: high reading. What is your diagnosis, and what do you do? 67
- Slides: 67