Achondroplasia Dwarfism Kelly La Barre Clinical Features l
Achondroplasia: Dwarfism Kelly La. Barre

Clinical Features l l Achondroplasia literally means “without cartilage formation. ” Presents clinically as a long narrow trunk with short extremities, large head with frontal bossing, hypoplasia of the midface, and trident configuration of the hands. An autosomal dominant disorder; a majority of cases are sporadic, the result of a de novo mutation. A de novo mutation is a new mutation that occurs in a germ cell and is then passed on to an offspring.
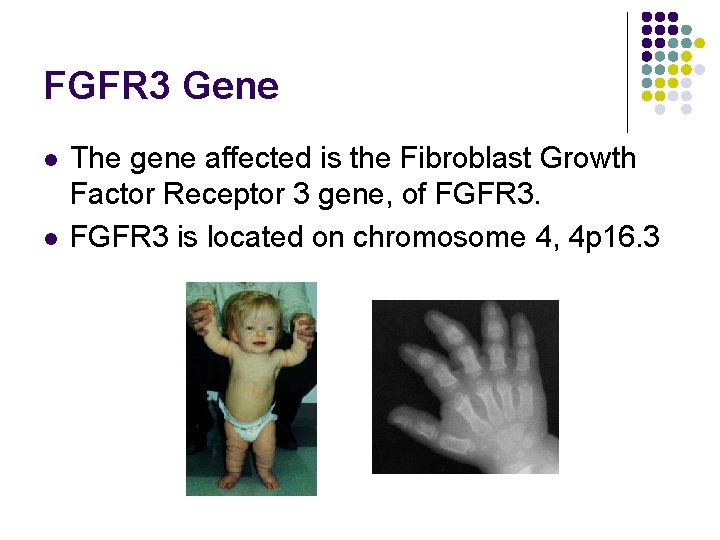
FGFR 3 Gene l l The gene affected is the Fibroblast Growth Factor Receptor 3 gene, of FGFR 3 is located on chromosome 4, 4 p 16. 3

Mutation l l There is a G to A transition at nucleotide 1138 of the coding sequence resulting in a gly 380 to arg substitution. Glycine is a basic side chain with its positive charge stabilized by resonance. Arginie is a nonpolar side chain consisting of a single hydrogen atom. GGG changes to AGG in the majority of the cases of achondroplasia.
Protein Function l l l These proteins play a role in several important cellular processes, including regulation of cell growth and division, determination of cell type, formation of blood vessels, wound healing, and embryo development. FGFR 3 is a transmembrane protein. The FGFR 3 protein is involved in the development and maintenance of bone and brain tissue. Researchers believe that this receptor regulates bone growth by limiting the formation of bone from cartilage in the long bones.

Conserved Domains Most of the sections of the conserved domains are tyrosine kinases, which are enzymes that can transfer a phosphate group from ATP to a tyrosine residue in a protein. This is important because FGFR 3 is a transmembrane protein that can interact outside the cell to create a cascade of events inside. The remainder of the conserved domains are immunoglobulin chains.
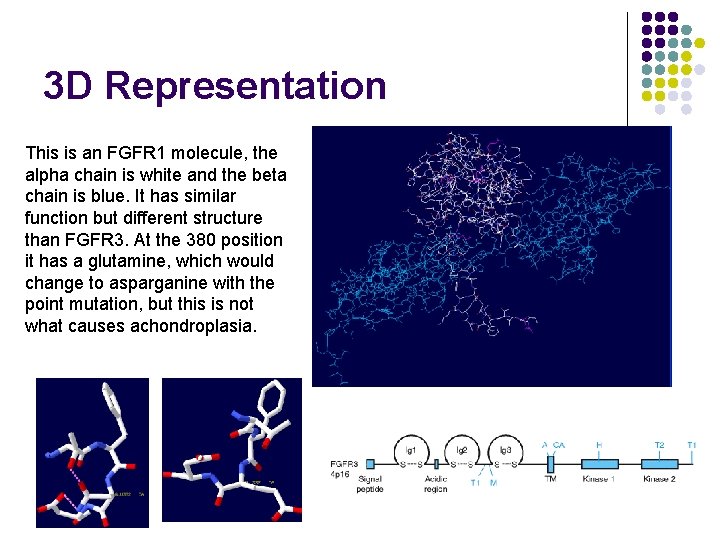
3 D Representation This is an FGFR 1 molecule, the alpha chain is white and the beta chain is blue. It has similar function but different structure than FGFR 3. At the 380 position it has a glutamine, which would change to asparganine with the point mutation, but this is not what causes achondroplasia.
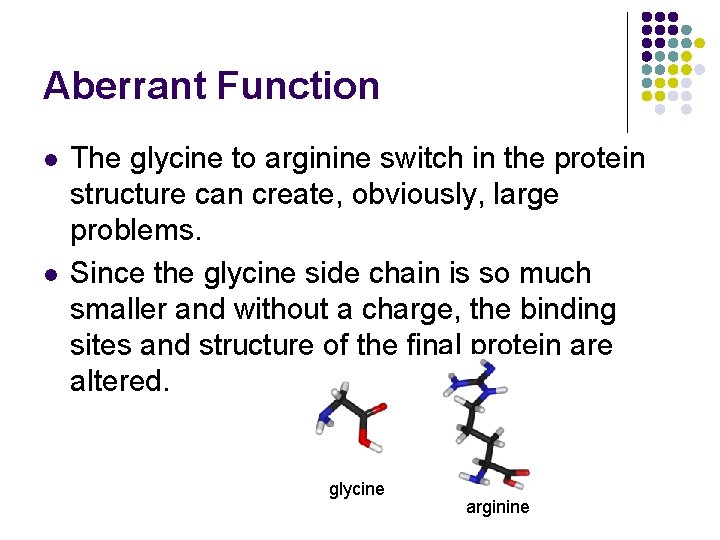
Aberrant Function l l The glycine to arginine switch in the protein structure can create, obviously, large problems. Since the glycine side chain is so much smaller and without a charge, the binding sites and structure of the final protein are altered. glycine arginine

Why? The normal function of FGFR 3 is to slow down the formation of bone by inhibiting the proliferation of chondrocytes, the cells that produce cartilage. The mutation increases the activity of FGFR 3, severely limiting bone growth. The mutant receptors actually work better than the wild-type.

References l l l Gene. Reviews Editor-in-chief: Pagon, Roberta A. Associate editors: Cassidy, Suzanne B. ; Bird, Thomas C. ; Dinulos, Mary Beth; Feldman, Gerald L. ; Smith, Richard J. H. ; Dolan, Cynthia R. Technical editor: Baskin, Patricia K. Seattle (WA): University of Washington; 1993 -2006 Genes and disease. Bethesda (MD): National Library of Medicine (US), NCBI. Human Molecular Genetics 2 2 nd ed. Strachan, Tom and Read, Andrew P. New York and London: Garland Science; c 1999 http: //www. ncbi. nlm. nih. gov/BLAST/ http: //www. ncbi. nlm. nih. gov/entrez/query. fcgi? db=OMIM RCSB Protein Data Bank : http: //www. rcsb. org/pdb/home. do
- Slides: 10