Accuracy vs Precision Significant Figures Why Accuracy and












- Slides: 14

Accuracy vs. Precision & Significant Figures

Why Accuracy and Precision is Important Measurements must be made with standard instruments using standard procedures! If not we may get errors, limitations, and inaccuracies

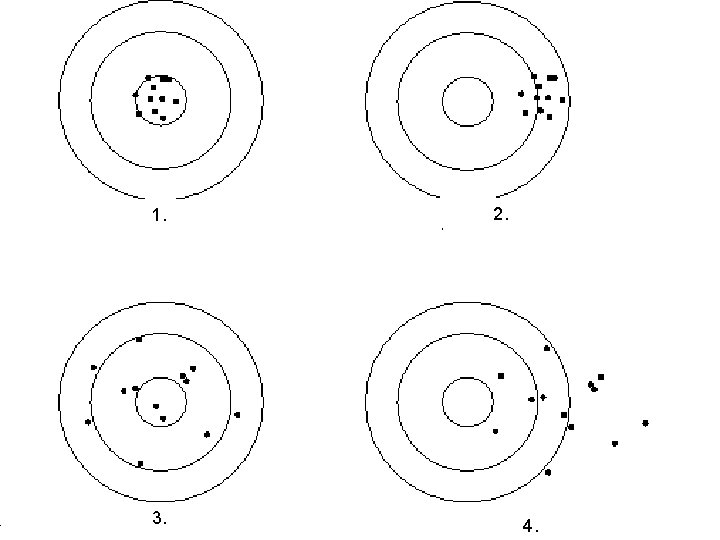
Accuracy §Accuracy is the extent to which a measurement approaches the true value of a quantity §Deals with how exact the measurement is to the true value §Ex: 35. 8 m. L 37. 2 m. L actual=36. 0 m. L (35. 8 m. L is more accurate)

Precision §Extent to which a series of measurements of the same quantity made in the same way agree with one another §Deals with how close the numbers agree §Ex: 110 g, 109 g, 111 g, 110 g

1. 3. 2. 4.

Significant Figures § Any digit in a measurement that is known with certainty plus one final digit, which is somewhat uncertain or estimated § AKA “Sig Figs” § EX: 11. 687 g (11. 68 is known) (0. 007 is the estimated digit)

Rules of Determining Sig Figs 1. Nonzero digits are always significant Ex: 47. 8 3 Sig Figs 2. Zeros Between nonzero digits are significant Ex: 40. 7 3 Sig Figs 3. Zeros in front of nonzero digits are not significant Ex: 0. 007 1 Sig Fig 4. Zeros at the end of a number and to the right of a decimal point are significant Ex: 85. 00 4 Sig Figs 5. Zeros at the end of a number with no decimal point are not significant Ex: 2000 1 Sig Fig 6. A decimal place after zeros means all numbers are significant Ex: 2000. 4 Sig Figs

How Many Sig Figs? § 31. 9 §. 000009 § 20. 5 § 3000 § 2013 § 6000. § 13. 175 § 50, 000 § 859. 00 §. 0000004 § 4000 § 300. § 18 § 10. 00 § 3

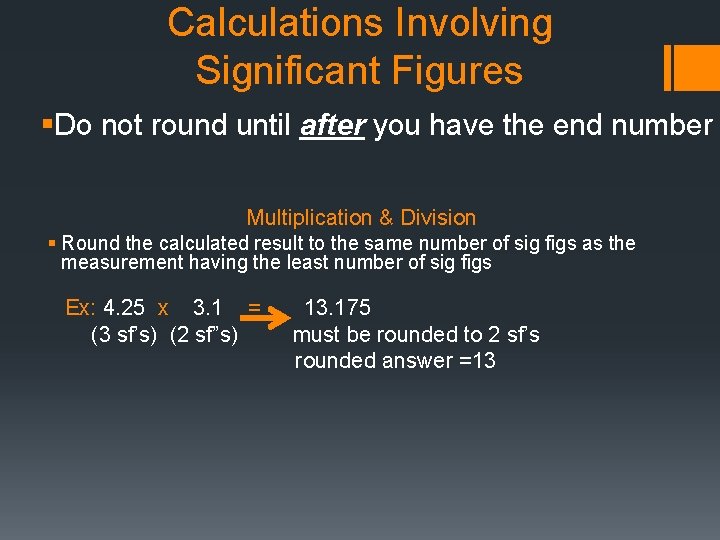
Calculations Involving Significant Figures §Do not round until after you have the end number Multiplication & Division § Round the calculated result to the same number of sig figs as the measurement having the least number of sig figs Ex: 4. 25 x 3. 1 = (3 sf’s) (2 sf”s) 13. 175 must be rounded to 2 sf’s rounded answer =13

Calculations Involving Significant Figures Addition and Subtraction § Answer can have no more digits to the right of the decimal point than there are in the measurement with the smallest number of digits to the right of the decimal point Ex: 3. 95 2. 879 213. 6 220. 429 round to 220. 4

Solve With the Correct Amount of Sig Figs ? a) 986. 72 / 5. 12 = 193 192. 71875 5 sf’s 3 sf’s 8 sf’s

Solve With the Correct Amount of Sig Figs b) 27. 343 6. 09 13. 1244 46. 56 46. 5574 ? 3 #’s after decimal 2 #’s after decimal 4 #’s after decimal 2 4 #’s after decimal

Scientific Notation § A way of writing very large or very small numbers in a way that is easier to calculate and takes up less space § Contains a number between 1 -10 and a power of 10 § Move decimal left = positive exponent § Move decimal right = negative exponent § EX: 60200000000000 = 6. 02 x 1023 0. 00000000785 = 7. 85 x 10 -17

Calculations with Scientific Notation § If numbers in scientific notation are multiplied exponents are added § Ex: (4. 2 x 102) (3. 9 x 105) = 16 x 107 § If numbers in scientific notation are subtracted exponents are subtracted § Ex (8. 2 x 107) / (4. 1 x 104) = 2. 0 x 103