Accuracy and Precision Remember these definitions Accuracy a

Accuracy and Precision

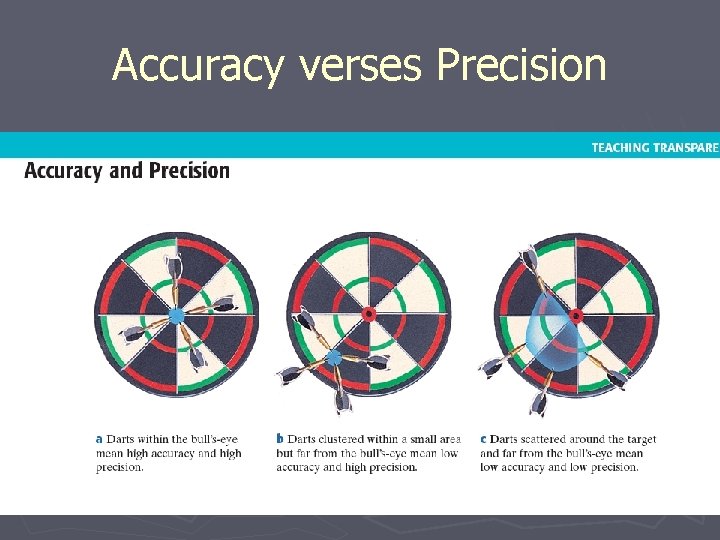
Remember these definitions? ► Accuracy – a description of how close a measurement is to the true value of the quantity measured ► Precision – the exactness of a measurement
Accuracy verses Precision

Is it Accurate, Precise, Both or Neither? ► Known Density = 3. 11 g/m. L § Test Results 3. 77, 3. 81, 3. 76, 3. 80 § Precise, not accurate § Test Results 3. 01, 3. 89, 3. 50, 5. 99 § Neither § Test Results 3. 04, 3. 20, 3. 13, 3. 07 § Accurate, not precise § Test Results 3. 11, 3. 12, 3. 10 § Both

What are some reasons for accuracy or precision being off? ►Error § Human Error ►Parallax Error – angle error ►Mathematical Error § Instrument Error

How do we represent error? ► Error is the difference between the actual (or accepted) value and the experimental value ► Percent Error = Accepted – Experimental Accepted x 100

Example Problem ► Working in the laboratory, a student finds the density of a piece of pure aluminum to be 2. 85 g/cm 3. The accepted value for the density of aluminum is 2. 699 g/cm 3. What is the student's percent error? % error = |2. 699 g/cm 3 – 2. 85 g/cm 3| x 100 2. 699 g/cm 3 = 5. 60 %

Another Example Problem ► A student takes an object with an accepted mass of 200. 00 grams and masses it on his own balance. He records the mass of the object as 196. 5 g. What is his percent error? % error = |200. 00 g – 196. 5 g| x 100 200. 00 g = 1. 75 %

Which leads us to the lab ► Density can be measured in two ways. ► If it is a liquid: § Find the mass of the liquid § Find the volume of the liquid § Divide the mass by the volume ► If it is a solid § Find the mass of the solid § Submerge the solid in a liquid and record the difference in the volume of the liquid (final – initial) § Divide the mass by the volume difference
- Slides: 9