Accuracy and Precision in the Lab Precision and

Accuracy and Precision in the Lab

Precision and Accuracy Errors in Scientific Measurements Precision - Refers to reproducibility or “How close the measurements are to each other. ” Accuracy - Refers to how close a measurement is to the real or true value. Systematic error - produces values that are either all higher or all lower than the actual value. Random Error - in the absence of systematic error, produces some values that are higher and some that are lower than the actual value.
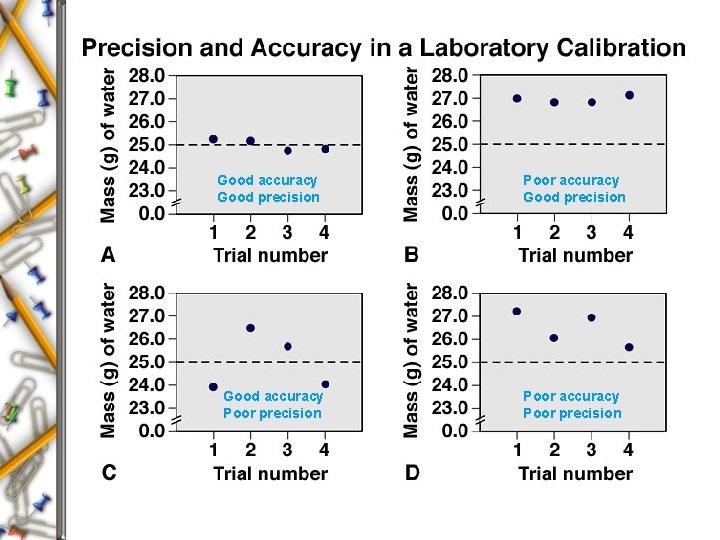
Good accuracy Good precision Good accuracy Poor precision Poor accuracy Good precision Poor accuracy Poor precision

Balances 0. 1 mg precision Accuracy determined by calibration

Graduated Cylinder Estimate the volume in the graduated cylinder… Most would read between 19. 1 to 19. 3 m. L No one would properly read the volume as high as 19. 5 m. L or as low as 19. 0 m. L. No one could reasonably read the volume to the 1/100 s digit. Correct way to read a graduated cylinder

Graduated Cylinder Incorrect way to read a graduated cylinder
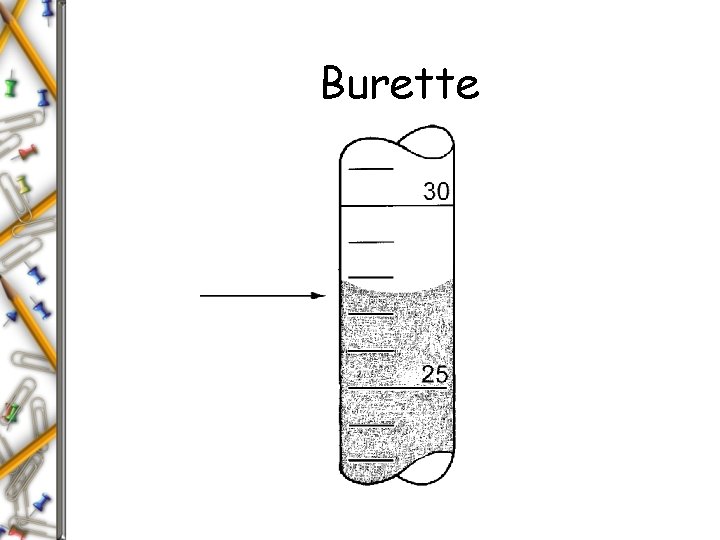
Burette

Spectronic-20 Estimate the reading on the upper scale… Most would read between 35. 4 to 35. 6 %T No one would properly read the scale as high as 36. 0 or as low as 35 %T.

The Number of Significant Figures in a Measurement Depends Upon the Measuring Device A linearly calibrated instrument can usually be read to 1 digit more precision than the engraved calibration

Significant Digit Rules and Conventions A significant figure (also called a significant digit) in a measurement is one which is known to some level of precision. The rules presented here are simplifications of a more complete statistical analysis and should be used to imply a certain confidence in a written numerical value. These rules are not infallible. To avoid round-off errors when making multiple-step calculations, carry one or two extra significant figures in the intermediate calculations. Round off the answer to the appropriate number of significant figures at the very end.

Rules: All nonzero digits in a reported value are significant. 422 g has 3 significant figures (SFs) Zeroes between nonzero digits are significant. 2003 miles has 4 SFs Trailing zeroes after the decimal point are significant. -2. 10 J has 3 SFs 0. 110 g has 3 SFs Leading zeroes after the decimal point are not significant. 0. 00214 g has 3 SFs Trailing zeroes in a number without a decimal point lead to ambiguity and are usually assumed not to be significant. Eliminate the ambiguity by converting to scientific notation (exponential notation). 96, 500 C (3, 4, or 5 SFs) might be 9. 650 x 104 C (4 SFs)

Handling Significant Figure in Calculations Addition/Subtraction: The number of decimal digits in the final answer is the same as the minimum number of decimal digits in any measurement. +

Multiplication/Division: The total number of significant figures in the final answer is equal to the minimum number of significant figures in any measurement. Area = Length x Width Length = 12. 5 cm Width = 2. 0 cm X

Logarithms: The number of decimal digits in the answer is equal to the number of significant digits in the measurement. Log 54 (2 SFs) = 1. 73 (2 decimal digits) Powers of 10 and antilog: The number of significant figures in the answer is equal to the number of decimal digits in the measurement. 10 -2. 53 (2 decimal digits) = 0. 0030 (2 SFs)

Review • Precision refers to the reproducibility of multiple measurements • Accuracy refers to how close a measurement or average of measurements is to the real or true value. • Errors can be systematic (unidirectional and can be eliminated) or random (bidirectional and normal) • A linearly calibrated instrument can usually be read to 1 digit more precision than the engraved calibration • Presenting measurements and calculated results with appropriate significant digits is a way to display the estimated precision of the values. • The rules of significant figure calculations are merely approximations of a much more rigorous statistical analysis and must be used carefully to avoid introducing unexpected and possibly undetected errors.
- Slides: 15