Access to Essential Medicines and Supplies Supporting national













- Slides: 16

Access to Essential Medicines and Supplies Supporting national capacities Bonface Fundafunda Supply Division UNICEF

UNICEF Priorities 2002 -5 and The Supply Function Immunisation Plus HIV/AIDS • School-in-a-Box Kit • WES Girls’ Education • Vaccine campaigns • Vaccine security • GAVI • Vaccine safety • Polio eradication Child Protection • Technical support • Roll Back Malaria • ARVs • Essential medicines • PMTCT • Micronutrients • Test kits • Water & Sanitation • Emergency response Early Childhood

Products • BASIC • Medicines: Antiretrovirals (ARVs), Opportunistic Infections (OIs), Sexually Transmitted Infections (STIs), Antimalarials • Test Kits: HIV, STIs, Malaria • Micronutrients • SPECIAL • High Protein energy foods - helping to reduce nutritional vulnerabilities • Blended foods for children (UNIMIX) • Home/Community packs - for improved access/adherence Under development

The UNICEF’s ARV supply strategy is based on guiding principles: • Support global policies to ensure equitable, sustainable availability and access to basic essential drugs • Follow WHO guidelines incl. Model List of Essential Medicines • Focus on primary health care centre – hard to reach populations • Assure technically sound procurement • Assure cost – effectiveness through international competitive bidding

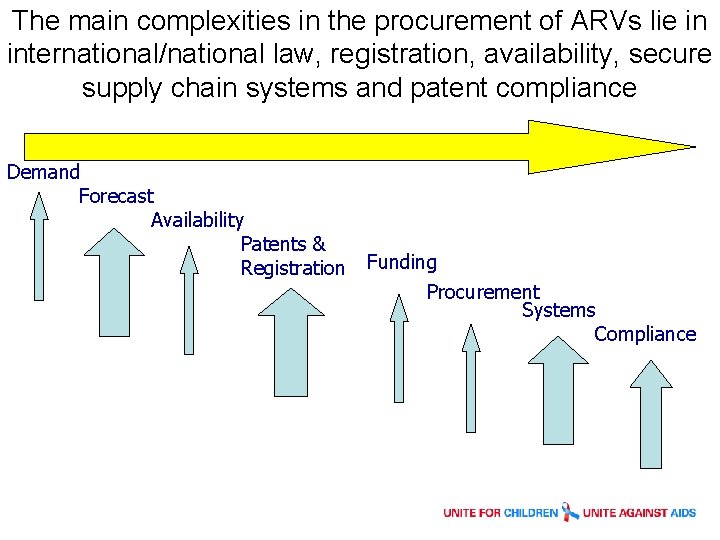
The main complexities in the procurement of ARVs lie in international/national law, registration, availability, secure supply chain systems and patent compliance Demand Forecast Availability Patents & Registration Funding Procurement Systems Compliance

UNICEF supports countries’ supply chain management by combining its field operations with global procurement Demand Creation Product Selection Monitoring Effective Use Receipt, Storage, Distribution Forecasting Field Offices Financing Supply Division Quality Product Procurement Assurance Supplier Agreements

The procurement of ARVs is complex and requires appropriate procurement, supply and logistics capacity at global and local levels • ARV demand has no history and depends on patient testing and acceptance Continuous monitoring • Secure delivery to treatment sites is essential – value of ARVs/ poorly-paid health workers and local logistics personnel Logistics arrangements • Zero tolerance on ARV stock-outs is required to avoid any interruption in treatment Copenhagen stockpile, $1. 6 m • Patent and registration situation needs to be clear in each country Patents review • The ARV supply must be accompanied by timely availability of test kits and supporting lab equipment and supplies Comprehensive supply plans

INCREASED SUPPLIER BASE IN 2006 • WHO pre-qualification ↑ • FDA approval ↑ INCREASED COMPETITION • FDC d 4 T + 3 TC + NVP 30 mg $130. 2 per patient/year • FDC d 4 T + 3 TC + NVP 40 mg $138. 12 per patient/year

INCREASED COMPLEXITY • Different specifications Material specifications (eg, child-proof caps under FDA) • Volume discounts • Lead times • Careful monitoring of contract awards (paying attention to lead- times, discounts and actual performance)

UNICEF has 35 one-to- two year purchase arrangements for ARVs and diagnostics 17 18 Originators, generics, distributors Diagnostics ARVs

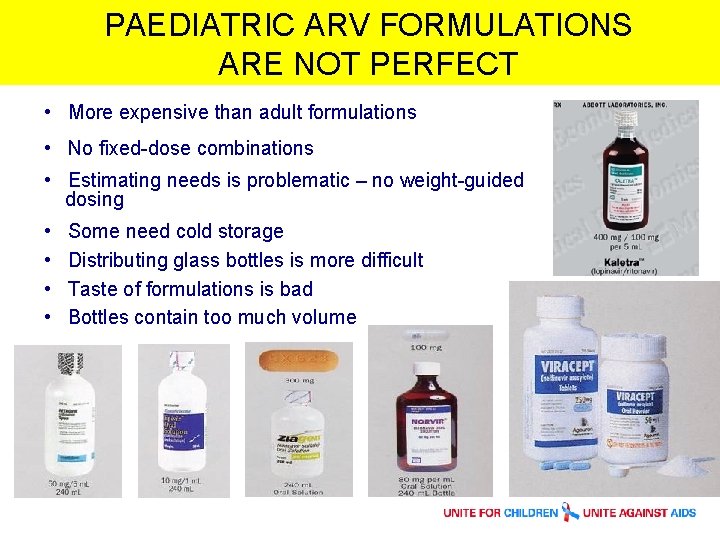
PAEDIATRIC ARV FORMULATIONS ARE NOT PERFECT • More expensive than adult formulations • No fixed-dose combinations • Estimating needs is problematic – no weight-guided dosing • • Some need cold storage Distributing glass bottles is more difficult Taste of formulations is bad Bottles contain too much volume

Transparency in prices is provided through Sources and Prices Report www. unicef. org/supply

Common supply-related problems • Relatively new health problem; medicines new, patented and expensive • Old medicines useful for opportunistic infections out of production (pyrimethamine, sulfadiazine) • Regulatory/Legal impediments – registration, Intellectual Property protection • Quantities may be too small to motivate manufacturers – important to consolidate • Availability of Pediatric dosage forms severely restricted • Programme context not always understood by procurers – inadequate feedback with procurer • Quality requirement and Assurance time consuming

Common supply-related problems - contd. • Lead time in appropriate in some situations – e. g. gender based violence • Existing weak health infrastructure impedes efficiency • HIV/AIDS still emotive and politically charged subject • Ordering of non-essential/non-standard treatment guideline items • Funding still a problem

Conclusion LEGISLATIVE, REGULATORY AND COMMERCIAL CONTEXT: ensure full compliance in advance SUPPLIER INTEGRITY: pre qualify suppliers, manage tender process transparently, monitor performance PRODUCT AND PROCESS QUALITY: monitor and evaluate In Country Logistics: reliable and functioning system is imperative to assure full access INFORMATION SYSTEM: maintain information integrity and flow for action

Supply