Absorption SpectroscopyProtein Function Topic 4 Part 2 Biophysics

Absorption Spectroscopy/Protein Function Topic 4 Part 2 Biophysics
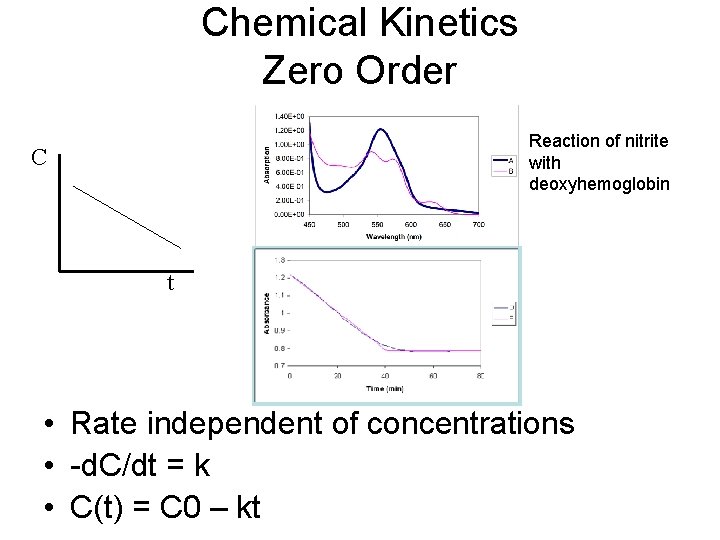
Chemical Kinetics Zero Order Reaction of nitrite with deoxyhemoglobin C t • Rate independent of concentrations • -d. C/dt = k • C(t) = C 0 – kt
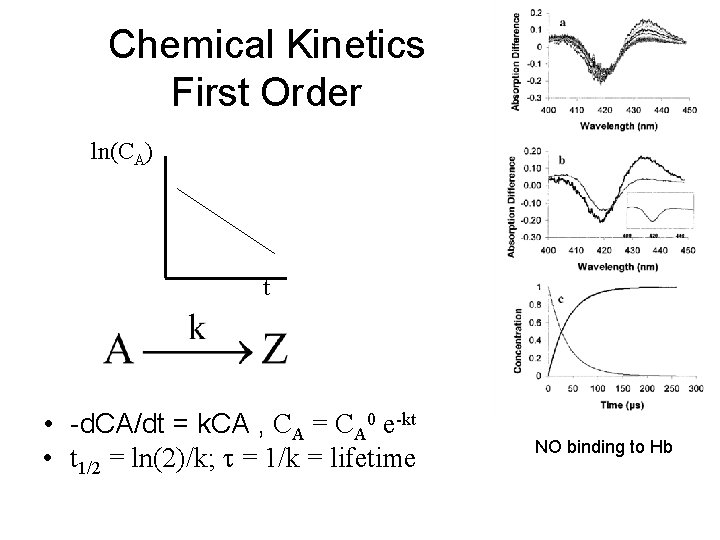
Chemical Kinetics First Order ln(CA) t • -d. CA/dt = k. CA , CA = CA 0 e-kt • t 1/2 = ln(2)/k; t = 1/k = lifetime NO binding to Hb

Chemical Kinetics Second Order • -d. CA/dt = -d. CB/dt = k. CACB • Make one species in excess so get pseudofirst order kinetics, kobs = k. CB so CA = CA 0 exp(-kobst)

Hemoglobin § Cooperative Binding of Oxygen §Linked to quaternary structure §Explained by MWC Model

On the Nature of Allosteric Transitions: A Plausible Model Jacques Monod, Jeffries Wyman, Jean-Pierre Changux J. Mol. Biol. 1965
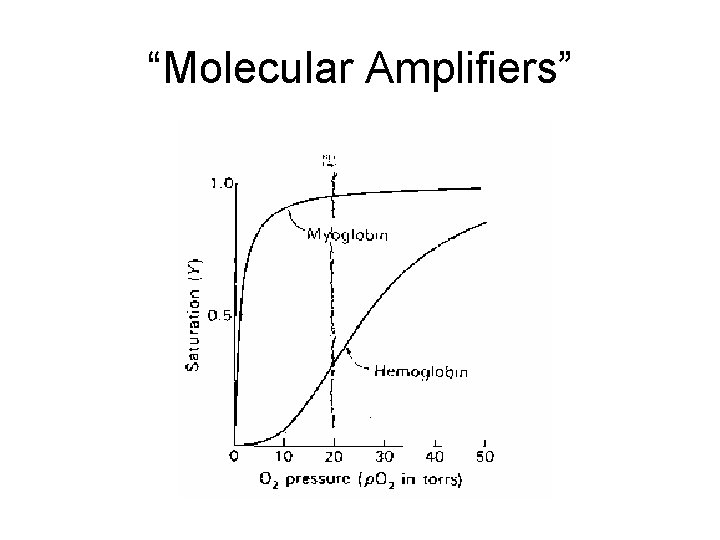
“Molecular Amplifiers”
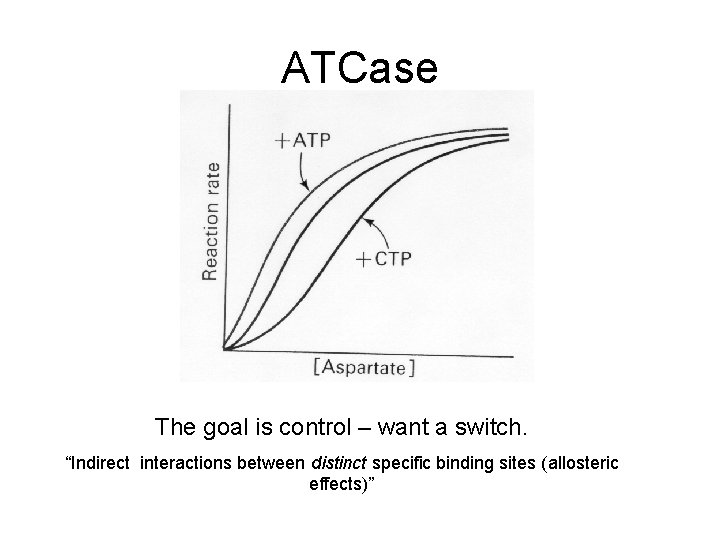
ATCase The goal is control – want a switch. “Indirect interactions between distinct specific binding sites (allosteric effects)”

Definitions and Generalizations • Homotrophic effects – identical ligands (eg. for Hb: O 2, CO, NO) • Heterotrophic effects –different ligands (eg. for Hb: DPG, IHP, Cl-, NO as SNO, NEM) • Most allosteric proteins are oligomers (several subunits or protomers) • Allosteric changes often involve quaternary stucture • Heterotrophic - positive or negative, Homotrophic – only positive (exception of Hg reductase? )

Model in English 1. Allosteric proteins are oligomers where the protomers are arranged symmetrically 2. There is one and only one identical ligandbinding site on each protomer 3. Tertiary structure of protomers affected by quaternary structure 4. There are two quaternary states (R and T) which dictate ligand affinities on all protomers 5. Transitions between states preserve symmetry

Model in Math • T 0 = L R 0, , L is the allosteric constant, (Big L = Big allostery) • Only also define Ø c defines relative affinities of quaternary states and a defines absolute affinity of one ØWhen L is small

Compare Hill Equation vs Q is a constant, n is the number of ligand sites, n is Hill coefficient

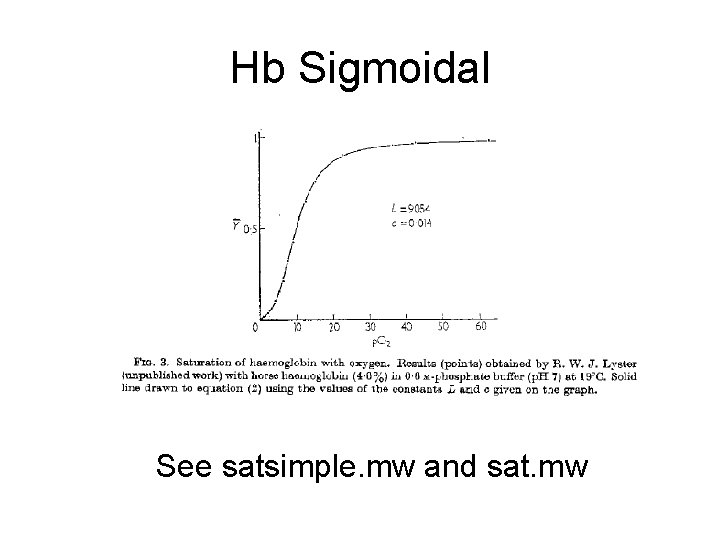
Hb Sigmoidal See satsimple. mw and sat. mw

Heterotrophic effectors Affect L Activators decrease L (push to R) and Inhibitors increase it
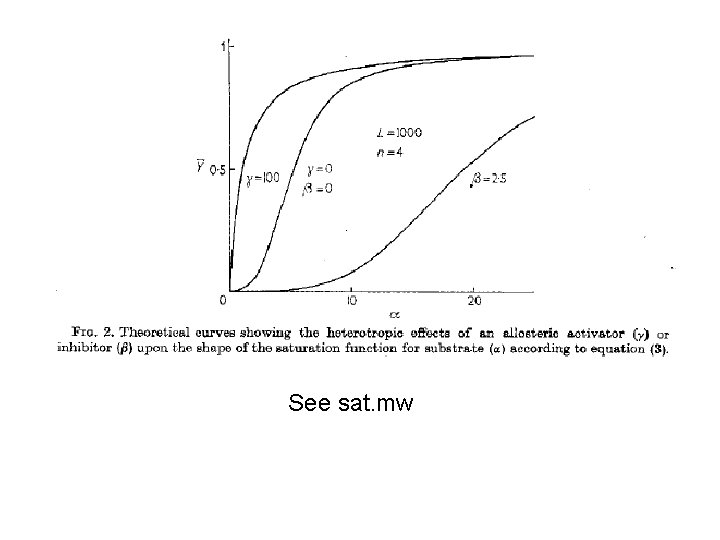
See sat. mw
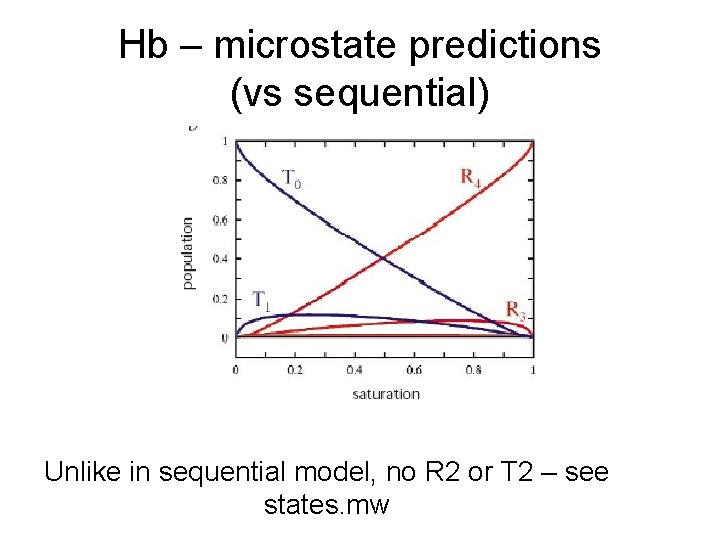
Hb – microstate predictions (vs sequential) Unlike in sequential model, no R 2 or T 2 – see states. mw
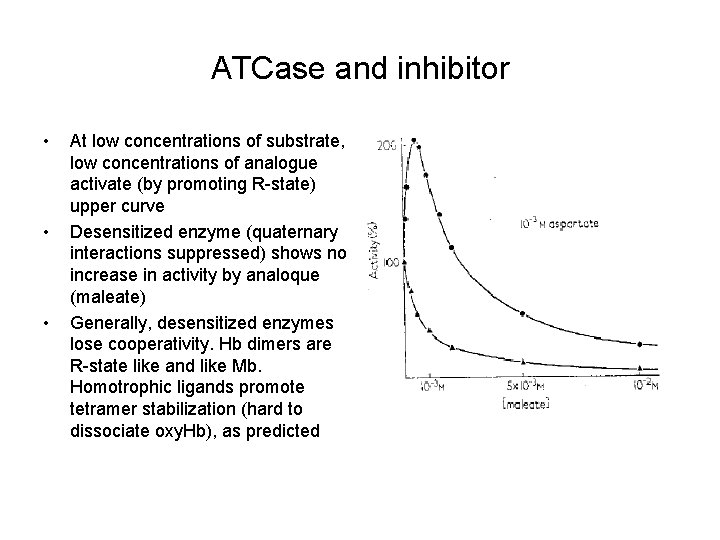
ATCase and inhibitor • • • At low concentrations of substrate, low concentrations of analogue activate (by promoting R-state) upper curve Desensitized enzyme (quaternary interactions suppressed) shows no increase in activity by analoque (maleate) Generally, desensitized enzymes lose cooperativity. Hb dimers are R-state like and like Mb. Homotrophic ligands promote tetramer stabilization (hard to dissociate oxy. Hb), as predicted
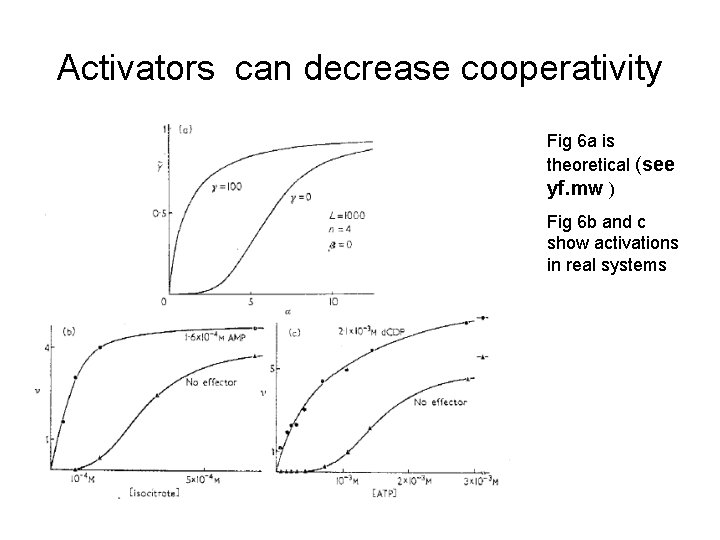
Activators can decrease cooperativity Fig 6 a is theoretical (see yf. mw ) Fig 6 b and c show activations in real systems

Confirmations of MWC ATCase • Model predicts fraction in R-state > fraction ligand bound. Schachman lab (1966) shows this using sedimentation to examine quaternary state (size) and spectroscopy for ligation. • They also showed (like Gerhart lab) low concentration of inhibitor activate ATCase

Confirmations - Hb • MWC’s prediction of concomitant changes in tertiary structure in protomers with known symmetry of tetramer confirmed by more refined X-ray structures. • Perutz provides mechanism of allosteric transitions • Szabo and Karplus show quantitative agreement of MWC/perutz model with equilibrium data (Eg Lc 4 constant after all salt bridges broken). • Equilibrium oxygen binding to Hb trapped in T-state crystal non-cooperative (Eaton lab). • CO rebinding following photolysis of Hb. CO (R-state) much faster than CO binding to Hb (T-state) – Gibson.

- Slides: 22