Absorption spectroscopy In absorption spectroscopy the light incident

Absorption spectroscopy In absorption spectroscopy, the light incident on a sample under study is absorbed which results in reduction of intensity of the transmitted light. Incident radiation sample Transmitted radiation The amount of the light absorbed can be measured by absorbance which is linearly related to the concentration of the sample. This relation is given by the Beer’s Law, A = - log (I/I o) = abc A : absorbance a : absorptivity I : transmitted radiation b : pathlength Io: incident radiation c : concentration

Tunable diode laser absorption in combustion Tunable diode laser absorption spectroscopy (TDLAS) is a highly sensitive technique employed in concentration measurements of species, particularly in flames. The wavelength of the diode laser is tuned over the absorption lines of the species of interest and the intensity of transmitted light is measured. In our lab, a mid-infrared tunable diode laser is used as the light source. Combustion gases from the flame are extracted by a quartz microprobe into a cell of 18 m pathlength and the transmitted light from the cell is detected at a photodiode detector.
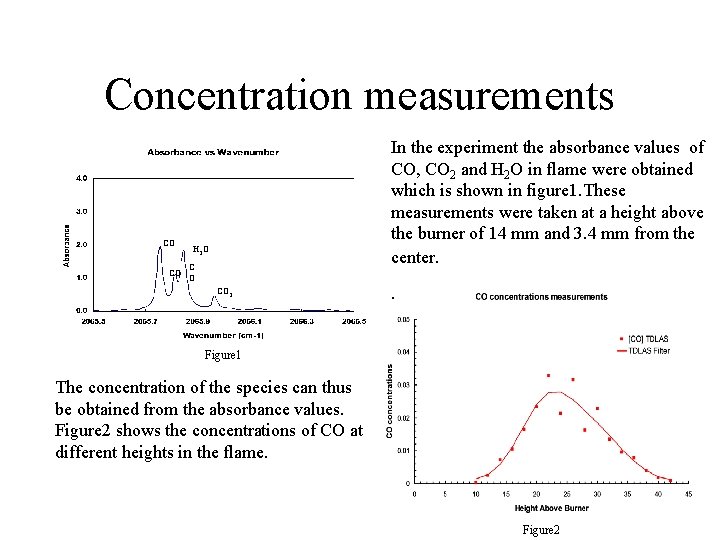
Concentration measurements CO CO In the experiment the absorbance values of CO, CO 2 and H 2 O in flame were obtained which is shown in figure 1. These measurements were taken at a height above the burner of 14 mm and 3. 4 mm from the center. H 2 O CO 2 . Figure 1 The concentration of the species can thus be obtained from the absorbance values. Figure 2 shows the concentrations of CO at different heights in the flame. Figure 2
- Slides: 3