Absorption Spectra of Iodine Vapor TA Paul Dent

Absorption Spectra of Iodine Vapor TA: Paul Dent pwdent@syr. edu

Why study I 2? Gaseous iodine is purple and therefore absorbs yellow light in the visible region. This absorption causes an electronic transition from the ground to an excited state. The vibrational levels of the ground state are low frequency. At room temperature several of the ground vibrational states are populated. Absorptions from a variety of initial states can be measured. Image from: http: //chemistry. about. com/od/elementgroups/ig/Halogens-Photo-Gallery/Iodine-Crystal. -duz. htm

Potential Energy Diagram of I 2

Excited State Well Characterization The absorption spectra yields information about the excited state well of I 2 The following parameters can be determined ν e′ (the harmonic vibrational frequency) xe’ (the anharmonicity correction factor) D’o (the dissociation energy) D’e (the well depth) νc’ (the vibrational quantum number at the continuum limit) All of these parameters can be calculated through the information from a Birge-Sponer Plot

Expected Vibrational Transitions must obey the Franck-Condon Principle Excitation is “vertical” because r does not change Small overlap between v”=0 ground state and v’=0 excited state The maximum in the Franck-Condon overlap is near the dissociation limit Energies above the dissociation limit correspond to the “free” relative motion of two I atoms The excited state vibrational levels get closer together as they approach the dissociation limit
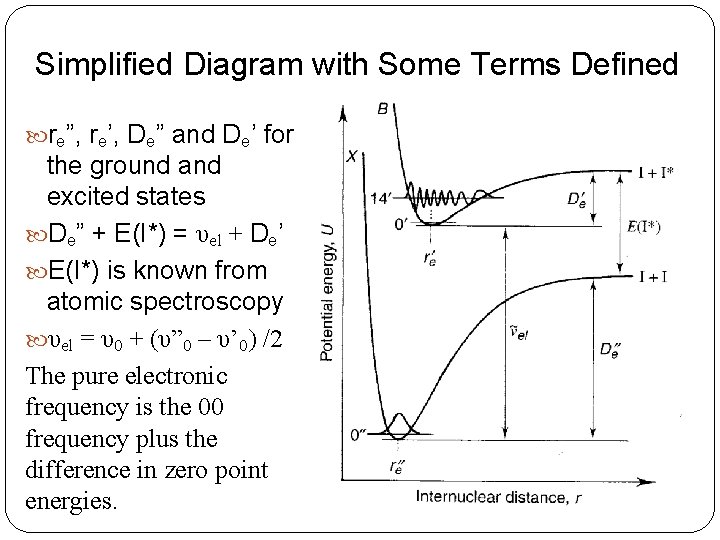
Simplified Diagram with Some Terms Defined re”, re’, De” and De’ for the ground and excited states De” + E(I*) = υel + De’ E(I*) is known from atomic spectroscopy υel = υ0 + (υ” 0 – υ’ 0) /2 The pure electronic frequency is the 00 frequency plus the difference in zero point energies.
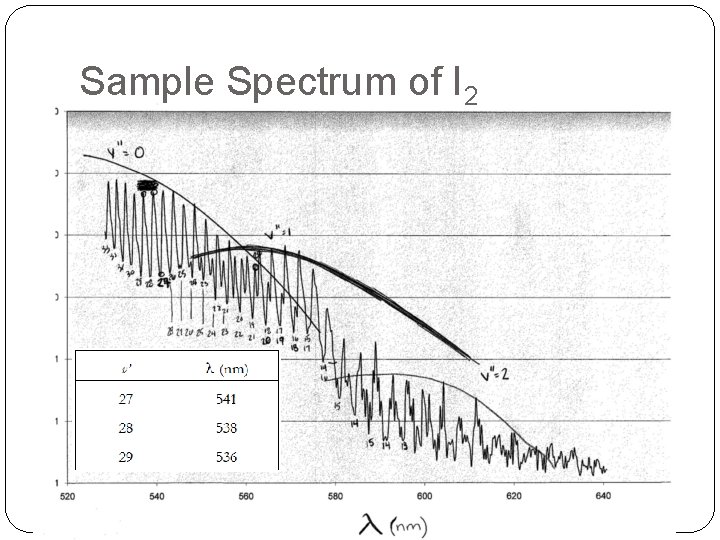
Sample Spectrum of I 2
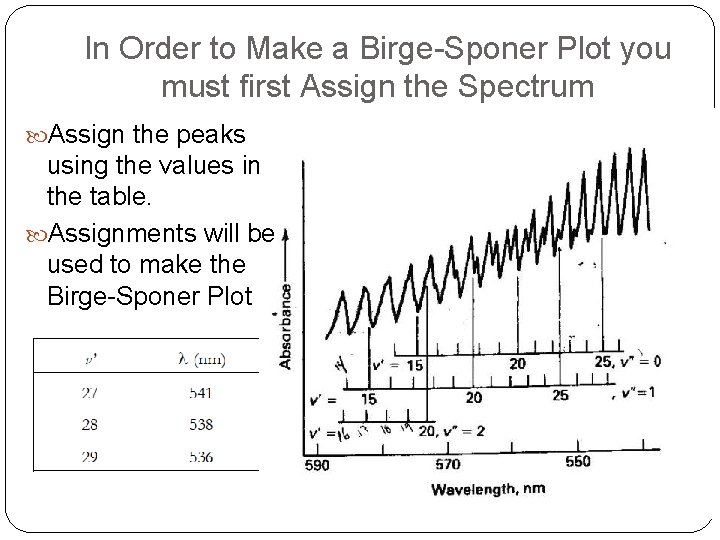
In Order to Make a Birge-Sponer Plot you must first Assign the Spectrum Assign the peaks using the values in the table. Assignments will be used to make the Birge-Sponer Plot

Birge-Sponer Plot For a harmonic oscillator G(v) = (v +1/2)υ’e And so = υ [(v+1)-v] =υ For a Morse oscillator G(v) = υe(v+1/2) – υexe(v+1/2)2 And so = υe’ 2υe’xe’(v’+1) Slope = -2 νe’xe’ ∆ν = νe’-2 νe’xe’ When X=1 or v’=0
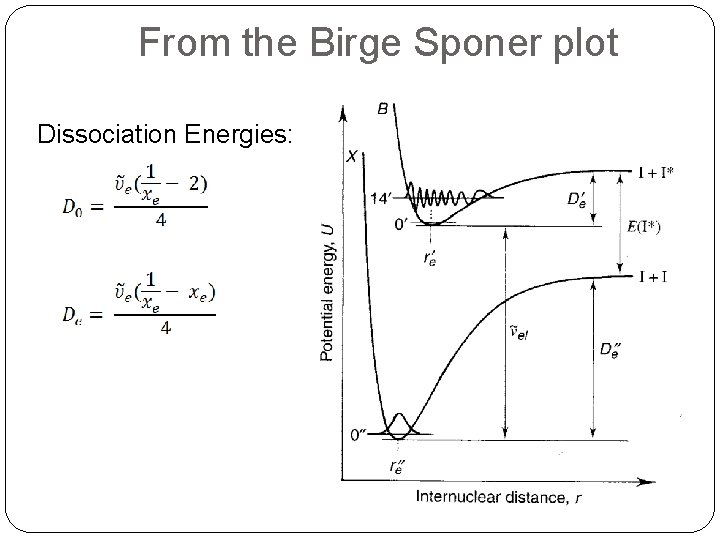
From the Birge Sponer plot Dissociation Energies:

Items To Include In Report The spectrum of I 2 absorption with wavelength as the x-axis A Birge-Sponer Plot made from the values assigned in the absorption spectrum Solve for all quantities using the Birge. Sponer Plot Answer all of the questions and make the appropriate calculations in the provided handout Start early so you can find me to ask
- Slides: 11