Absorption Kinetics of CO 2 in Novel Formulated

Absorption Kinetics of CO 2 in Novel Formulated 2 Amino-2 -Methyl-1 -Propanol and N-Methyl-4 Piperidinol Solvent TEERAWAT SEMA, T. Kiattinirachara, P. Na Ranong, P. Posoknistakul, R. Jiraratananon, P. Tontiwachwuthikul TMREES 2020 Conference June 25 -27, 2020 Email: teerawat. se@chula. ac. th
General Introduction Chemical Absorption Ø Affordable Ø Effective Ø Large amount of gas Ø Able to retrofit Ø Mature Ø Commercially proof

General Introduction KEY PARAMETERS for CO 2 Capture (chemical absorption): Process Design Packing Solvent The Easiest - The Cheapest - The Less Time Consuming way to improve the overall CO 2 capture performance is to change a solvent (highly effective solvent) FOCUSING ON SOLVENT
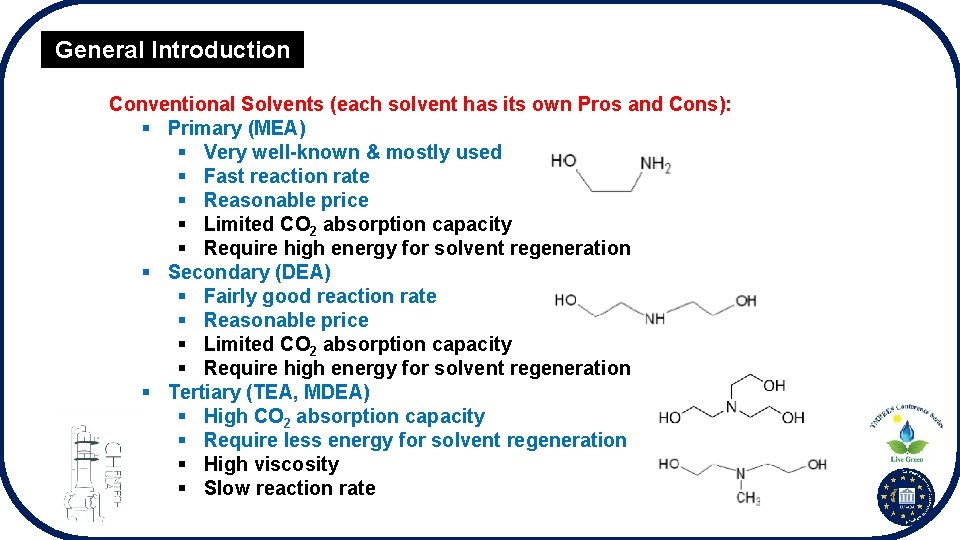
General Introduction Conventional Solvents (each solvent has its own Pros and Cons): § Primary (MEA) § Very well-known & mostly used § Fast reaction rate § Reasonable price § Limited CO 2 absorption capacity § Require high energy for solvent regeneration § Secondary (DEA) § Fairly good reaction rate § Reasonable price § Limited CO 2 absorption capacity § Require high energy for solvent regeneration § Tertiary (TEA, MDEA) § High CO 2 absorption capacity § Require less energy for solvent regeneration § High viscosity § Slow reaction rate

General Introduction Why New Solvents? § The conventional solvents, which have been using recently, do not yet show an ultimate performance. § Each conventional solvent has its Pros & Cons. § The ultimate/promising solvents should have existed. § The better (than those conventional) solvents can be found/designed/synthesized. § Actually, it depends on “How can we use the new solvents properly”.
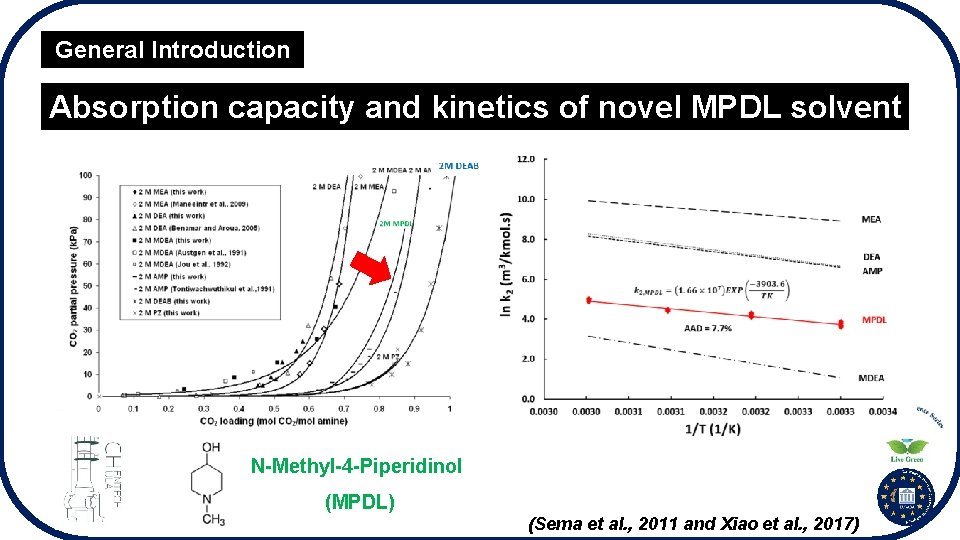
General Introduction Absorption capacity and kinetics of novel MPDL solvent N-Methyl-4 -Piperidinol (MPDL) (Sema et al. , 2011 and Xiao et al. , 2017)
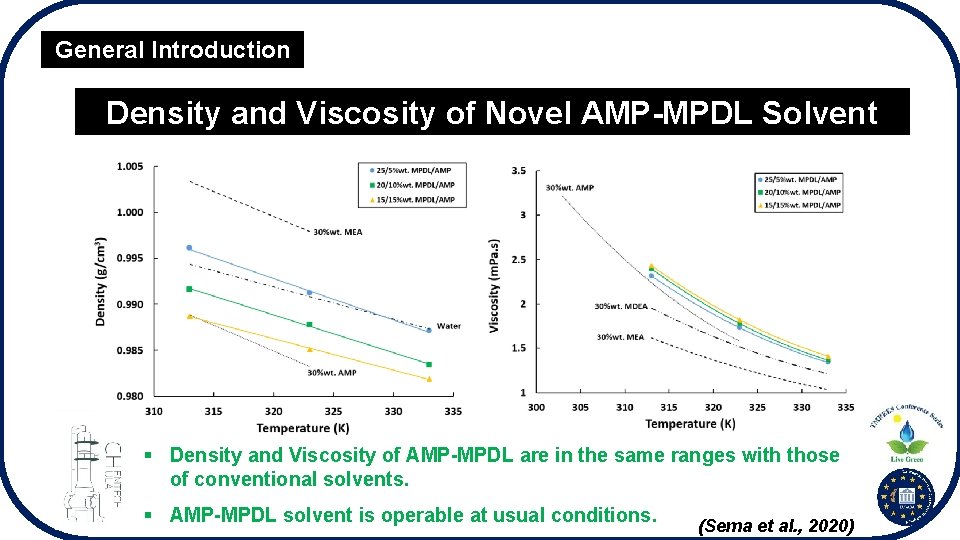
General Introduction Density and Viscosity of Novel AMP-MPDL Solvent § Density and Viscosity of AMP-MPDL are in the same ranges with those of conventional solvents. § AMP-MPDL solvent is operable at usual conditions. (Sema et al. , 2020)

General Introduction Absorption Performance of Novel MPDL I. Both absorption capacity and kinetics of MPDL are higher than those of MDEA. II. Absorption kinetics of MPDL is too low to use as a SINGLE solvent. III. MPDL is considered to blend with AMP to improve its kinetics drawback. IV. Absorption kinetics well represents the performance of CO 2 absorption. It also requires for designing the absorption column and the operating conditions.

OBJECTIVE Overall objective: To determine the absorption kinetics performance of novel formulated MPDL-AMP solvent.

EXPERIMENTAL SECTION
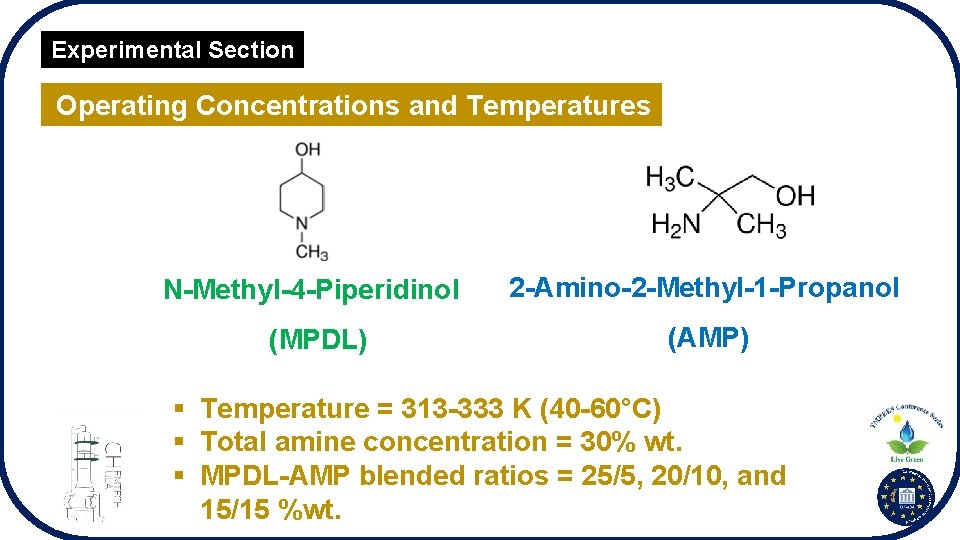
Experimental Section Operating Concentrations and Temperatures N-Methyl-4 -Piperidinol 2 -Amino-2 -Methyl-1 -Propanol (MPDL) (AMP) § Temperature = 313 -333 K (40 -60°C) § Total amine concentration = 30% wt. § MPDL-AMP blended ratios = 25/5, 20/10, and 15/15 %wt.
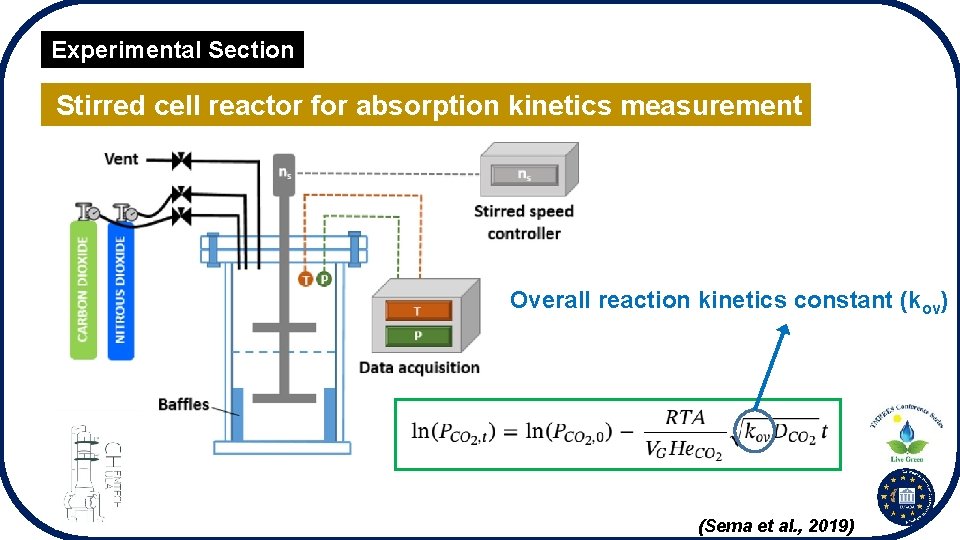
Experimental Section Stirred cell reactor for absorption kinetics measurement Overall reaction kinetics constant (kov) (Sema et al. , 2019)

Results and Discussion
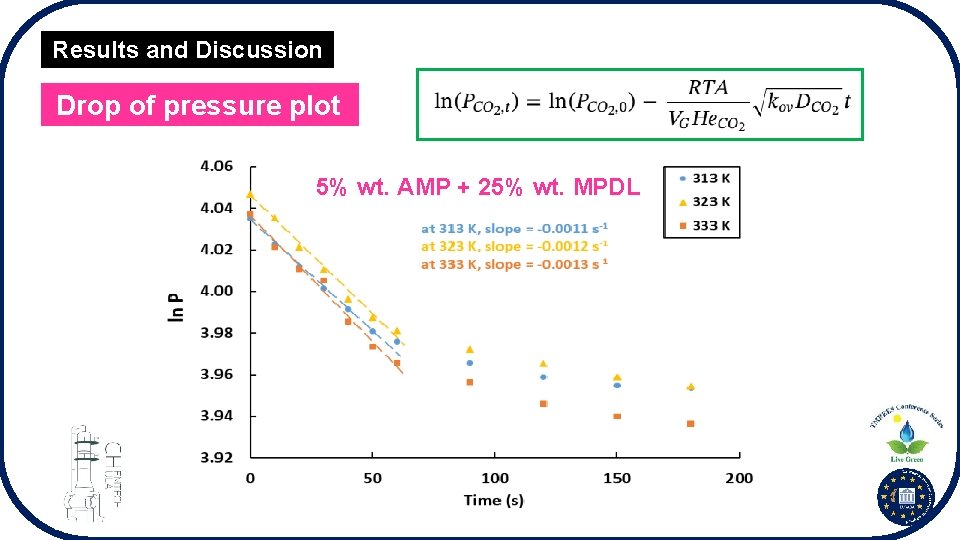
Results and Discussion Drop of pressure plot 5% wt. AMP + 25% wt. MPDL
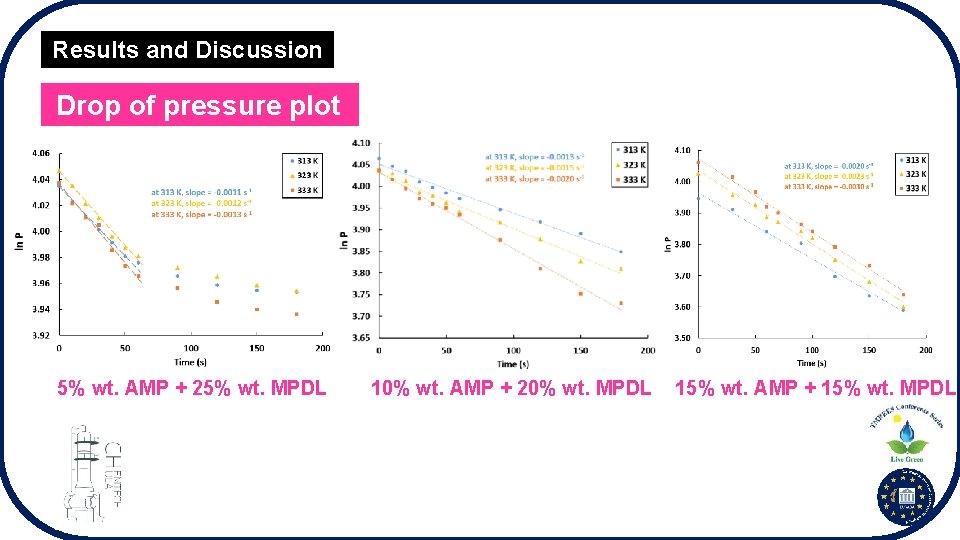
Results and Discussion Drop of pressure plot 5% wt. AMP + 25% wt. MPDL 10% wt. AMP + 20% wt. MPDL 15% wt. AMP + 15% wt. MPDL
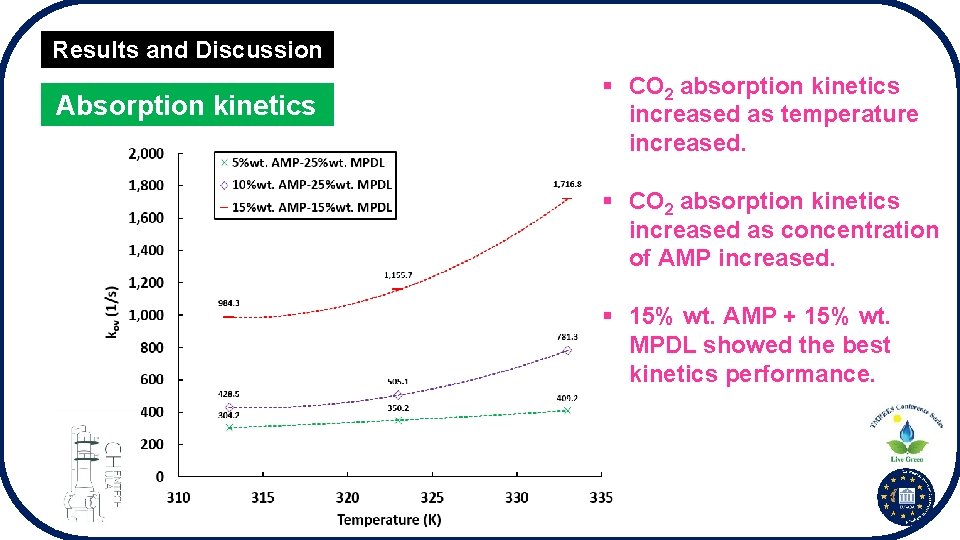
Results and Discussion Absorption kinetics § CO 2 absorption kinetics increased as temperature increased. § CO 2 absorption kinetics increased as concentration of AMP increased. § 15% wt. AMP + 15% wt. MPDL showed the best kinetics performance.
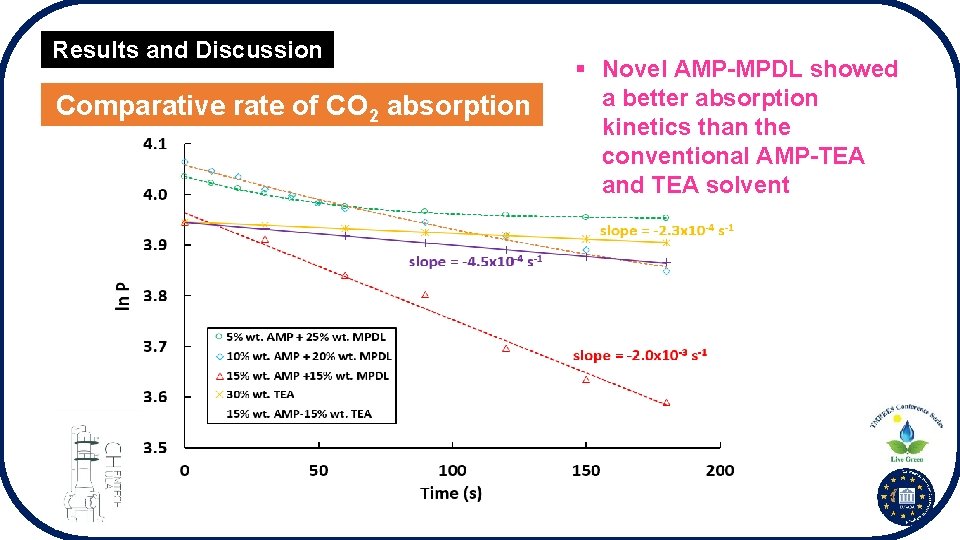
Results and Discussion Comparative rate of CO 2 absorption § Novel AMP-MPDL showed a better absorption kinetics than the conventional AMP-TEA and TEA solvent
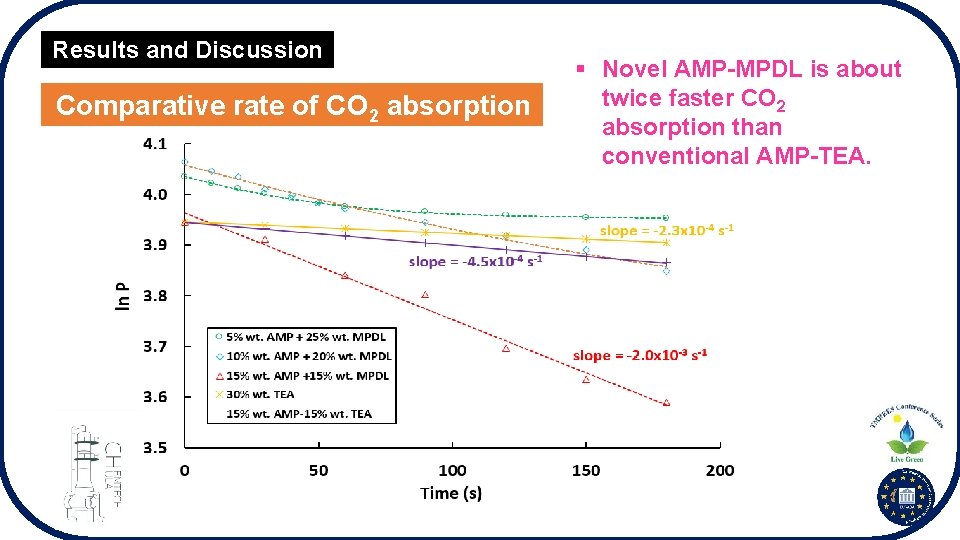
Results and Discussion Comparative rate of CO 2 absorption § Novel AMP-MPDL is about twice faster CO 2 absorption than conventional AMP-TEA.

Conclusions § Absorption kinetics of novel AMP-MPDL solvent increased as temperature and/or concentration of AMP increased. § Novel AMP-MPDL can be considered as POTENTIAL ALTERNATIVE SOLVENT for CO 2 capture. § Novel AMP-MPDL solvent is twice faster than conventional AMP-TEA solvent. § Further investigations on its absorption capacity and solvent regeneration ability should be considered.

Acknowledgement: • Grants for Development of New Faculty, Ratchadaphiseksomphot Endowment Fund, Chulalongkorn University, Thailand. • Thailand Research Fund and Office of the Higher Education Commission (Research Grant for New Scholar: MRG 6280187) • Mahidol University, Thailand. Contact: TEERAWAT SEMA, Ph. D. Department of Chemical Technology, Faculty of Science, Chulalongkorn University, Bangkok, Thailand Email: teerawat. se@chula. ac. th
- Slides: 20