Absorb UK Registry One year results of contemporary

Absorb UK Registry: One year results of contemporary bioresorbable vascular scaffold implantation Andreas Baumbach On behalf of the Absorb UK Steering group and investigators

Speaker's name: Andreas Baumbach I have the following potential conflicts of interest to report: • Institutional grant/research support: Abbott Vascular for the UK Registry • Receipt of honoraria or speaker fees: KSH, Microport, Sinomed, Medtronic, Astra Zeneca

ABSORB UK Registry Clin Trial Gov: NCT 01977534 Single Arm -, Post-market Registry in the United Kingdom Objective: to collect data for BVS in daily PCI practice in the UK. Evaluate implantation of BVS in a “real world” setting De novo lesions in previously untreated vessels Primary Endpoint Subjects: 1000 Sites: 24 Clinical follow-up Baseline 1 yr Endpoints: • Baseline: Patient and Lesion assessments • Outcome: MACE*, Ischemia driven TVF*, TLR*, ST* • Acute outcome: Device/Procedural success, In hospital assessments *Evaluated by independent CEC (Clinical Events Committee) 3 yr

Study Organisation Coordinating Centre - Bristol Heart Institute: Principle Investigator: Andreas Baumbach Research coordinator: Ruth Bowles R&D: Jake Harley Site Coordinator: Jo Roberts CEC coordinator: Vincy John Registry Sponsor – Abbott Vascular: Science: Susan Veldhof Biostats: Divine Ediebah Safety: Ravindra Kumar PM: Nadia Bouhdi MA: Solveig Grimstad Electronic Data Capture: Steering Committee: Azfar Zaman Jonathan Hill Mark de. Belder Adam de. Belder Adrian Banning Anthony Mathur Clinical Event Committee: Scott Garg Kanrath Balachandran Pierre Deharo Fawaz Bardooli Site Monitoring Clinical Endpoint Ltd. Nina Cooter (independent) Angiographic Core Lab (Events): Yale CRG, Alexandra Lansky 4

Patient Selection: Who would benefit the most? Favourable With caution? Avoid? • Young patients (<65) at risk for future interventions • Simple straightforwar d lesions • Prox. /Mid LAD • Multi vessel disease • Diabetes mellitus • STEMI • Bifurcations (jailing SB) • Long diffuse disease • SVG (size) • Ostial Lesions • Calcification • Tortuosity • ISR • CTO • • <2, 5 & >4, 0 mm Ø • • • Heavy calcification True bifurcations/2 stent strategy DAPT < 12 months Left Main Recommendation of the ABSORB UK Steering Committee 2014
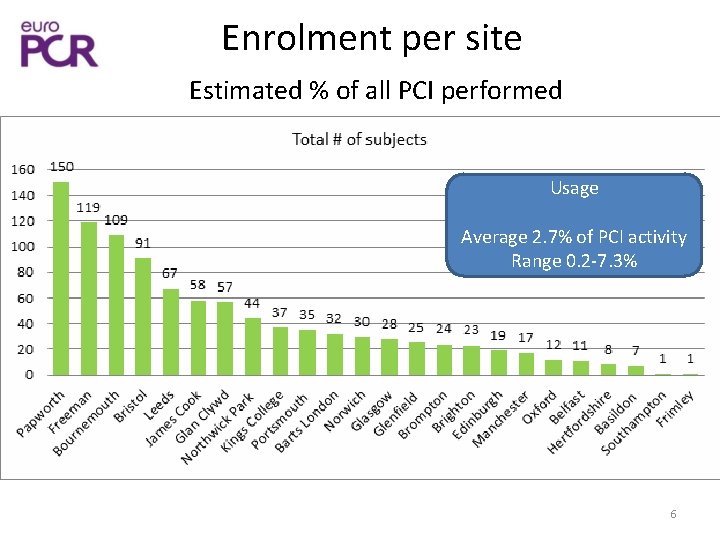
Enrolment per site Estimated % of all PCI performed Usage Average 2. 7% of PCI activity Range 0. 2 -7. 3% 6
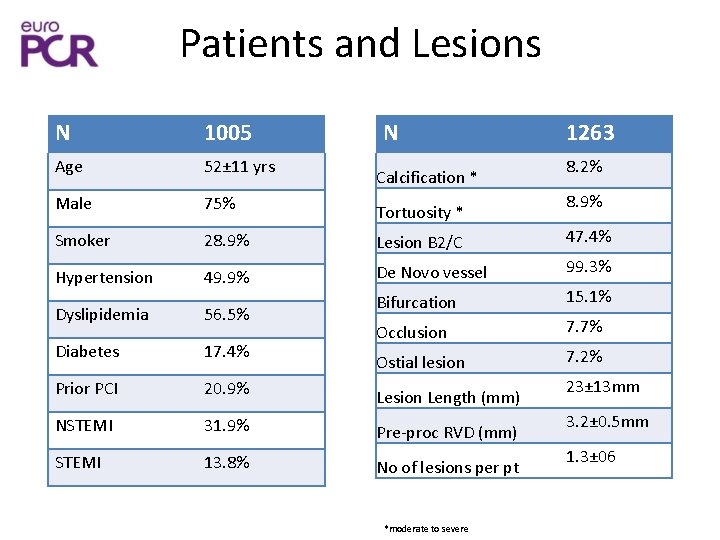
Patients and Lesions N 1005 N Age 52± 11 yrs Male 75% Tortuosity * Smoker 28. 9% Lesion B 2/C 47. 4% Hypertension 49. 9% De Novo vessel 99. 3% Bifurcation 15. 1% Occlusion 7. 7% Ostial lesion 7. 2% Calcification * Dyslipidemia 56. 5% Diabetes 17. 4% Prior PCI 20. 9% Lesion Length (mm) NSTEMI 31. 9% Pre-proc RVD (mm) STEMI 13. 8% No of lesions per pt *moderate to severe 1263 8. 2% 8. 9% 23± 13 mm 3. 2± 0. 5 mm 1. 3± 06
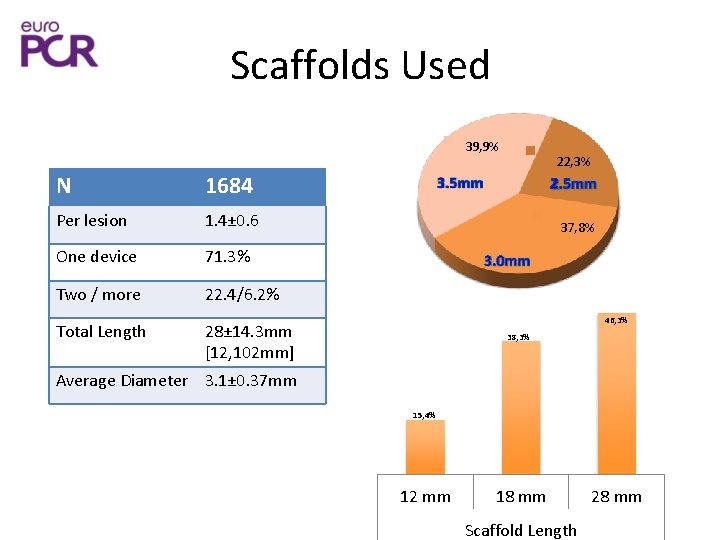
Scaffolds Used 39, 9% N 1684 Per lesion 1. 4± 0. 6 One device 71. 3% Two / more 22. 4/6. 2% Total Length 22, 3% 37, 8% 46, 3% 28± 14. 3 mm [12, 102 mm] 38, 3% Average Diameter 3. 1± 0. 37 mm 15, 4% 12 mm 18 mm Scaffold Length 28 mm

Implantation Technique • Radial access in 89. 3% • Use of imaging (IVUS or OCT) in 50% • Predilatation (median balloon size of 3. 0 mm) in 97. 5% • Postdilatation (NC balloon, mean 17 atm) in 94. 9% • ‘PSP’ (predilatation, sizing according to reference diameter, high pressure postdilatation >16 atm) in 65. 8% • Clinical procedure success 97. 3%
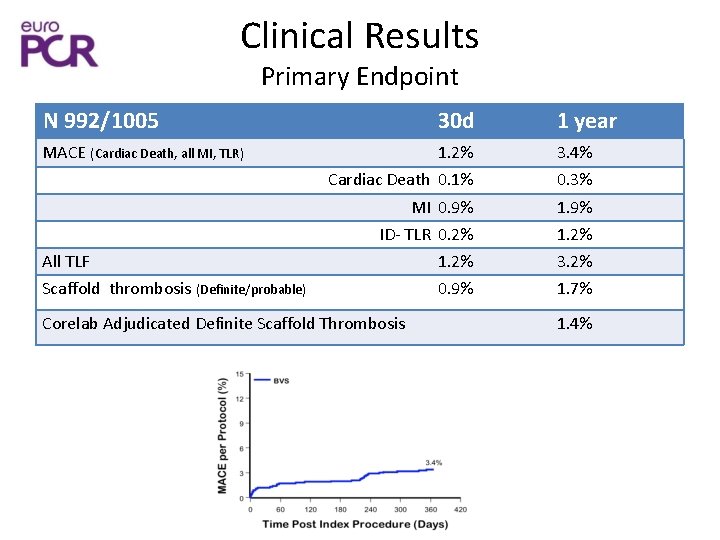
Clinical Results Primary Endpoint N 992/1005 MACE (Cardiac Death, all MI, TLR) All TLF Scaffold thrombosis (Definite/probable) 30 d 1 year 1. 2% Cardiac Death 0. 1% 3. 4% 0. 3% MI 0. 9% ID- TLR 0. 2% 1. 2% 0. 9% 1. 2% 3. 2% 1. 7% Corelab Adjudicated Definite Scaffold Thrombosis 1. 4%
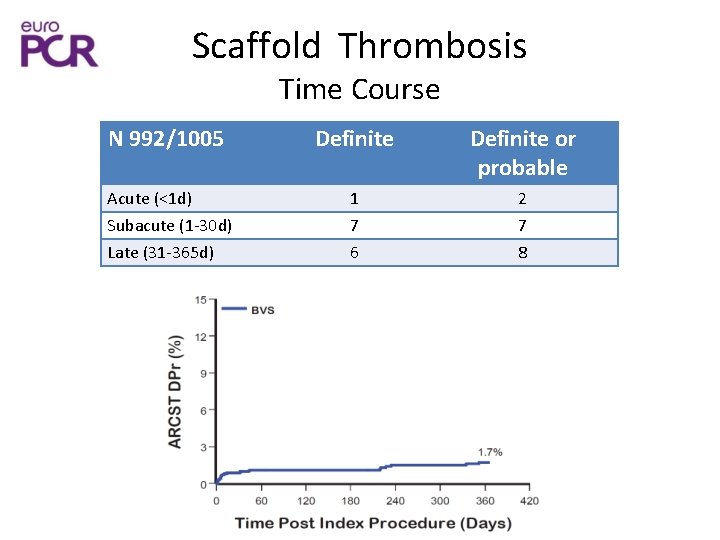
Scaffold Thrombosis Time Course N 992/1005 Definite or probable Acute (<1 d) 1 2 Subacute (1 -30 d) Late (31 -365 d) 7 6 7 8
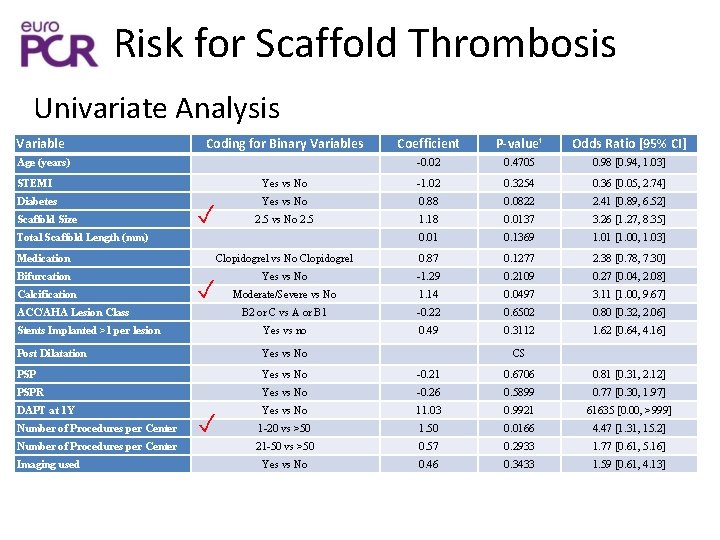
Risk for Scaffold Thrombosis Univariate Analysis Variable Coding for Binary Variables Age (years) Coefficient P-value¹ Odds Ratio [95% CI] -0. 02 0. 4705 0. 98 [0. 94, 1. 03] STEMI Yes vs No -1. 02 0. 3254 0. 36 [0. 05, 2. 74] Diabetes Yes vs No 0. 88 0. 0822 2. 41 [0. 89, 6. 52] 2. 5 vs No 2. 5 1. 18 0. 0137 3. 26 [1. 27, 8. 35] 0. 01 0. 1369 1. 01 [1. 00, 1. 03] Scaffold Size ✓ Total Scaffold Length (mm) Medication Clopidogrel vs No Clopidogrel 0. 87 0. 1277 2. 38 [0. 78, 7. 30] Bifurcation Yes vs No -1. 29 0. 2109 0. 27 [0. 04, 2. 08] Moderate/Severe vs No 1. 14 0. 0497 3. 11 [1. 00, 9. 67] B 2 or C vs A or B 1 -0. 22 0. 6502 0. 80 [0. 32, 2. 06] Stents Implanted >1 per lesion Yes vs no 0. 49 0. 3112 1. 62 [0. 64, 4. 16] Post Dilatation Yes vs No PSP Yes vs No -0. 21 0. 6706 0. 81 [0. 31, 2. 12] PSPR Yes vs No -0. 26 0. 5899 0. 77 [0. 30, 1. 97] DAPT at 1 Y Yes vs No 11. 03 0. 9921 61635 [0. 00, >999] 1 -20 vs >50 1. 50 0. 0166 4. 47 [1. 31, 15. 2] 21 -50 vs >50 0. 57 0. 2933 1. 77 [0. 61, 5. 16] Yes vs No 0. 46 0. 3433 1. 59 [0. 61, 4. 13] Calcification ✓ ACC/AHA Lesion Class Number of Procedures per Center Imaging used ✓ CS

Risk for Scaffold Thrombosis Multivariate Analysis Variable Coding for Binary Variables INTERCEPT Scaffold Size Coefficient (SE)1 P-value 2 Odds Ratio 1 [95% CI] Maximum Rescaled RSquare 3 -4. 49 2. 5 vs. >2. 5 mm 1. 18 (0. 48) 0. 0136 3. 27 [ 1. 28, 8. 37] Model Checking Statistics , P-value of Hosmer and Lemeshow Goodness-of-Fit Test C-Statistic (the Area Under the ROC Curve): 0. 64 Independent QCA analysis: 5/14 in vessels <2. 5 mm [1. 8, 2. 3 mm] 2/14 undersized 0. 04

Summary • UK ABSORB Registry represents the spectrum of operators and procedures at rollout of BVS in the UK – Large scale (>1000 patients) – Complete follow up (99%) – High quality data with independent CEC and Corelab adjudication • Contemporary practice for BVS – Careful lesion selection – High rate of postdilatation (PSP) – High rate of invasive imaging • BVS results demonstrate – High efficacy: Low TLF of 3. 2% at 12 m (similar to best in class DES) – Safety: Elevated scaffold thrombosis 1. 4% (attributed to small vessels, undersizing, operator experience) – The 3 year follow up for this cohort is ongoing • These results continue to inform best practices and point towards strategies to mitigate scaffold thrombosis
- Slides: 14