ABSORB Japan 3 year Clinical and Angiographic Results

ABSORB Japan: 3 -year Clinical and Angiographic Results of a Randomized trial Evaluating the Absorb Bioresorbable Vascular Scaffold vs. Metallic Drug - eluting Stent in de novo Native Coronary Artery Lesions Ken Kozuma, Kengo Tanabe, Takeshi Kimura on behalf of the ABSORB Japan Investigators.

Speaker's name: Ken Kozuma I do not have any potential conflict of interest I have the following potential conflicts of interest to report: • Honorarium: Abbott Vascular, Terumo • Institutional grant/research support: Abbott Vascular, Boston Scientific • Consultant: none • Employment in industry: none • Owner of a healthcare company: none • Stockholder of a healthcare company: none • Other(s):

Background • The 2 -year results of Absorb Japan presented in 2016 showed a relatively high incidence of very late scaffold thrombosis in Absorb as compared to XIENCE. • At 3 -years, the question of great interest among interventional cardiologists is whether the slope of the scaffold thrombosis curve will decrease after 2 years. • The 3 -year angiographic appearance would similarly be unique since this time-point is around the time of full scaffold resorption
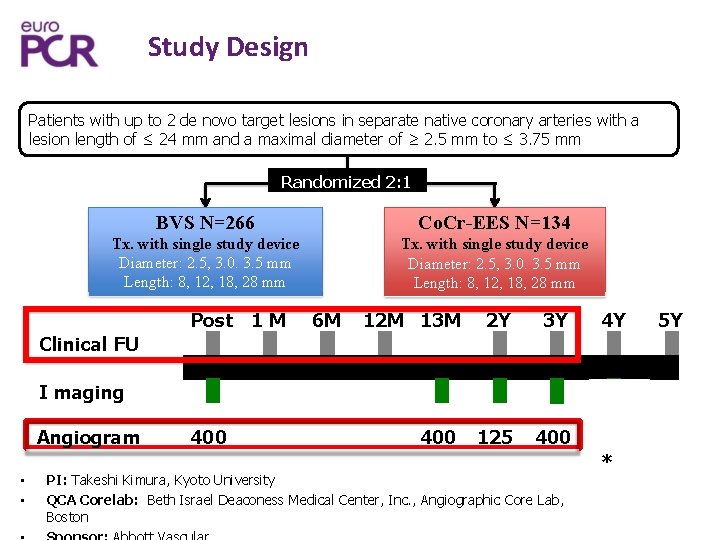
Study Design Patients with up to 2 de novo target lesions in separate native coronary arteries with a lesion length of ≤ 24 mm and a maximal diameter of ≥ 2. 5 mm to ≤ 3. 75 mm Randomized 2: 1 BVS N=266 Co. Cr-EES N=134 Tx. with single study device Diameter: 2. 5, 3. 0. 3. 5 mm Length: 8, 12, 18, 28 mm Post 1 M 6 M 12 M 13 M 2 Y 3 Y 4 Y Clinical FU I maging Angiogram OCT 1 400 125 • ( PI: Takeshi Kimura, Kyoto University Subrandomized) • QCA Corelab: Beth Israel Deaconess Medical Center, Inc. , Angiographic Core Lab, Boston Y * 5 Y
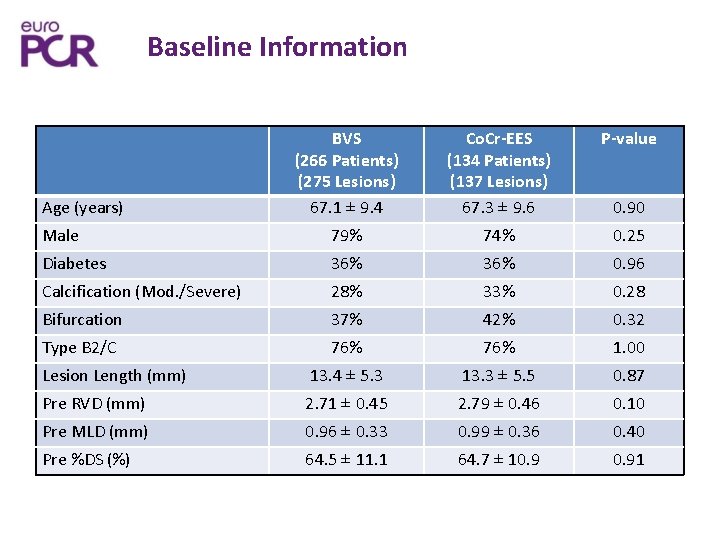
Baseline Information BVS (266 Patients) (275 Lesions) 67. 1 ± 9. 4 Co. Cr-EES (134 Patients) (137 Lesions) 67. 3 ± 9. 6 P-value Male 79% 74% 0. 25 Diabetes 36% 0. 96 Calcification (Mod. /Severe) 28% 33% 0. 28 Bifurcation 37% 42% 0. 32 Type B 2/C 76% 1. 00 Lesion Length (mm) 13. 4 ± 5. 3 13. 3 ± 5. 5 0. 87 Pre RVD (mm) 2. 71 ± 0. 45 2. 79 ± 0. 46 0. 10 Pre MLD (mm) 0. 96 ± 0. 33 0. 99 ± 0. 36 0. 40 Pre %DS (%) 64. 5 ± 11. 1 64. 7 ± 10. 91 Age (years) 0. 90
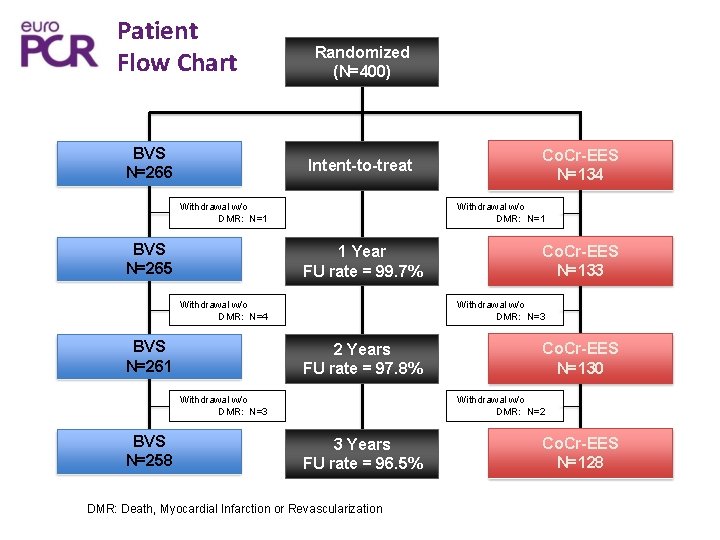
Patient Flow Chart BVS N=266 Randomized (N=400) Intent-to-treat Withdrawal w/o DMR: N=1 BVS N=265 Withdrawal w/o DMR: N=1 1 Year FU rate = 99. 7% Withdrawal w/o DMR: N=4 BVS N=261 Co. Cr-EES N=133 Withdrawal w/o DMR: N=3 2 Years FU rate = 97. 8% Withdrawal w/o DMR: N=3 BVS N=258 Co. Cr-EES N=134 Co. Cr-EES N=130 Withdrawal w/o DMR: N=2 3 Years FU rate = 96. 5% DMR: Death, Myocardial Infarction or Revascularization Co. Cr-EES N=128
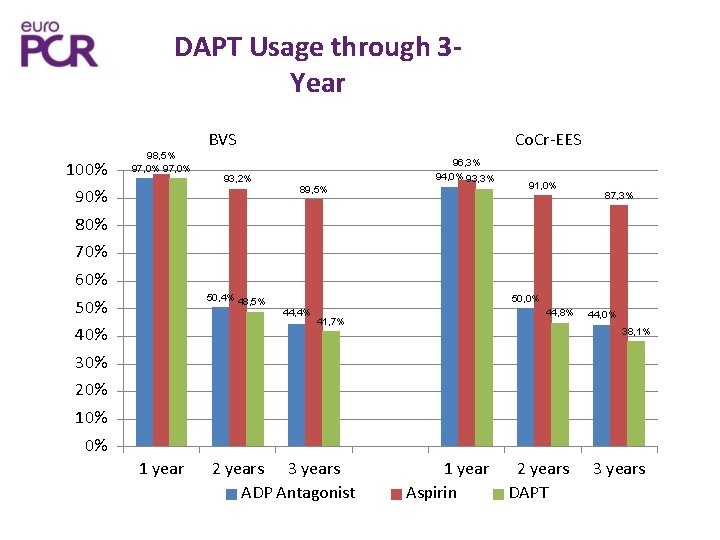
DAPT Usage through 3 Year 100% 98, 5% 97, 0% BVS 93, 2% 90% Co. Cr-EES 89, 5% 96, 3% 94, 0% 93, 3% 91, 0% 87, 3% 80% 70% 60% 50, 4% 48, 5% 50% 40% 50, 0% 44, 4% 44, 8% 41, 7% 44, 0% 38, 1% 30% 20% 10% 0% 1 year 2 years 3 years ADP Antagonist 1 year Aspirin 2 years DAPT 3 years
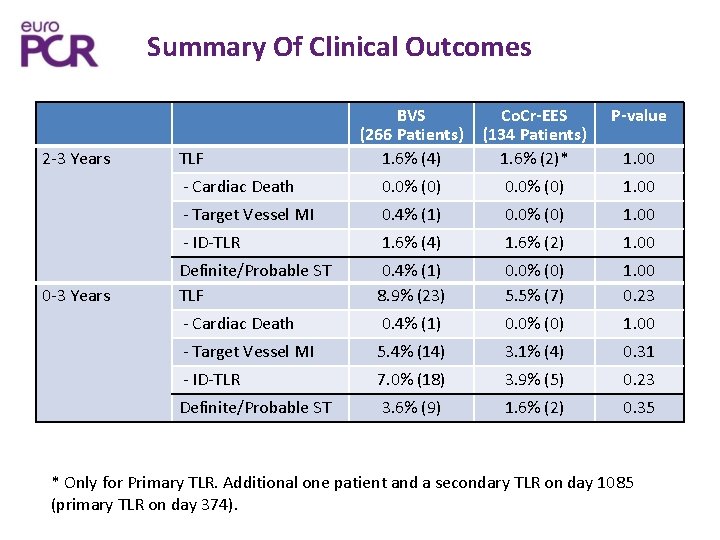
Summary Of Clinical Outcomes 2 -3 Years BVS (266 Patients) 1. 6% (4) Co. Cr-EES (134 Patients) 1. 6% (2)* P-value 0. 0% (0) 1. 00 0. 4% (1) BVS 1. 6% (4) (266 Patients) BVS(1) 0. 4% (266 Patients) 8. 9% (23) 0. 0% (0) Co. Cr-EES 1. 6% (2) (134 Patients) Co. Cr-EES 0. 0% (0) (134 Patients) 5. 5% (7) 1. 00 P-value 1. 00 - Cardiac Death 0. 4% (1) 0. 0% (0) 1. 00 - Target Vessel MI 5. 4% (14) 3. 1% (4) 0. 31 - ID-TLR 7. 0% (18) 3. 9% (5) 0. 23 Definite/Probable ST 3. 6% (9) 1. 6% (2) 0. 35 TLF - Cardiac Death - Target Vessel MI - ID-TLR 0 -3 Years Definite/Probable ST TLF 1. 00 0. 23 * Only for Primary TLR. Additional one patient and a secondary TLR on day 1085 (primary TLR on day 374).
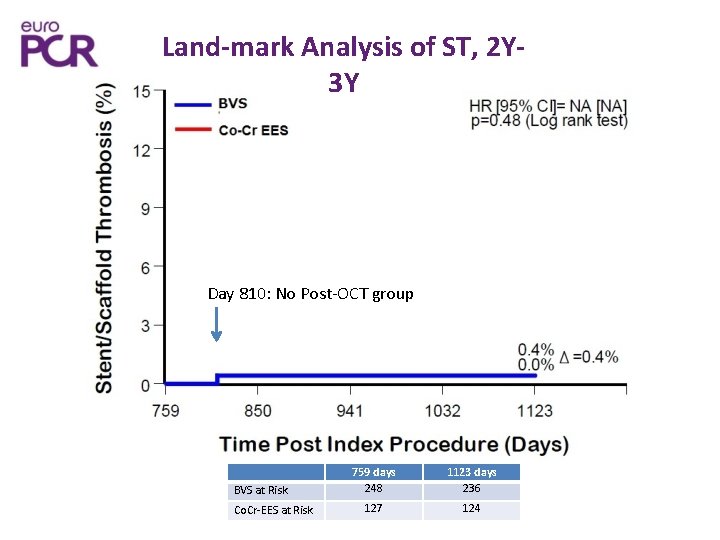
Land-mark Analysis of ST, 2 Y 3 Y Day 810: No Post-OCT group BVS at Risk Co. Cr-EES at Risk 759 days 248 1123 days 236 127 124
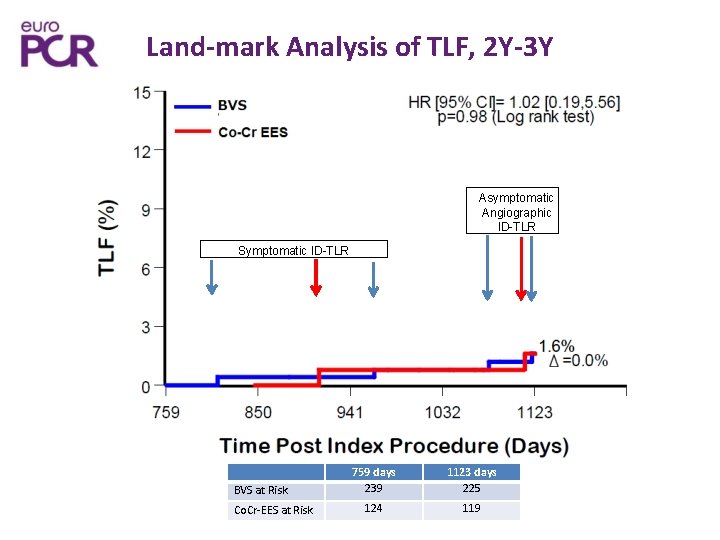
Land-mark Analysis of TLF, 2 Y-3 Y Asymptomatic Angiographic ID-TLR Symptomatic ID-TLR BVS at Risk Co. Cr-EES at Risk 759 days 239 1123 days 225 124 119
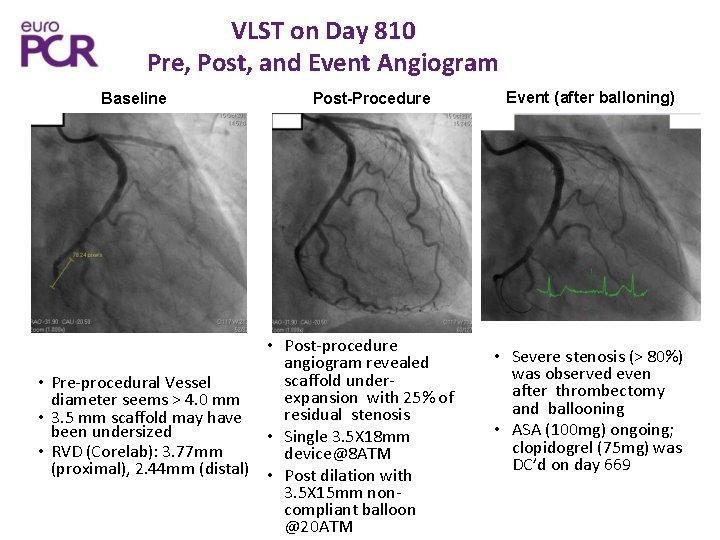
VLST on Day 810 Pre, Post, and Event Angiogram Baseline Post-Procedure • Post-procedure angiogram revealed scaffold under • Pre-procedural Vessel expansion with 25% of diameter seems > 4. 0 mm residual stenosis • 3. 5 mm scaffold may have been undersized • Single 3. 5 X 18 mm • RVD (Corelab): 3. 77 mm device@8 ATM (proximal), 2. 44 mm (distal) • Post dilation with 3. 5 X 15 mm noncompliant balloon @20 ATM Event (after balloning) • Severe stenosis (> 80%) was observed even after thrombectomy and ballooning • ASA (100 mg) ongoing; clopidogrel (75 mg) was DC’d on day 669
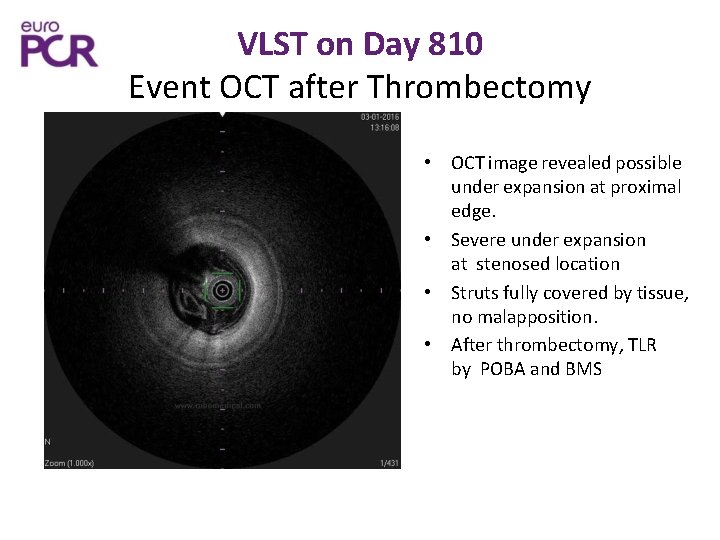
VLST on Day 810 Event OCT after Thrombectomy • OCT image revealed possible under expansion at proximal edge. • Severe under expansion at stenosed location • Struts fully covered by tissue, no malapposition. • After thrombectomy, TLR by POBA and BMS
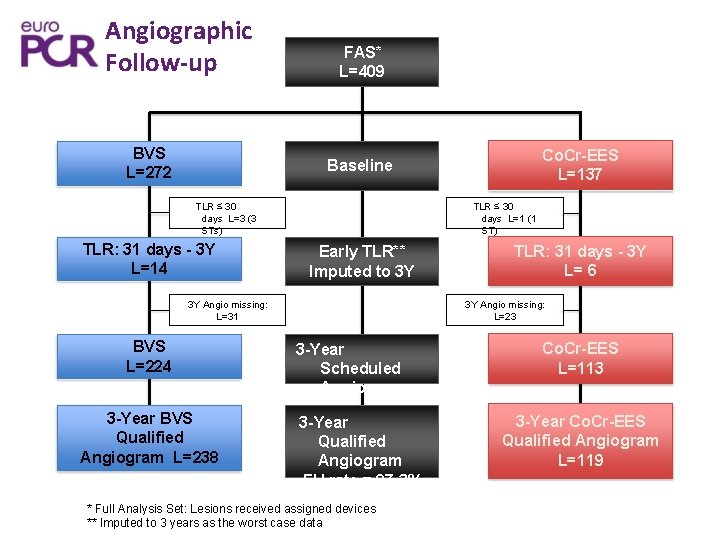
Angiographic Follow-up BVS L=272 FAS* L=409 Co. Cr-EES L=137 Baseline TLR ≤ 30 days L=3 (3 STs) TLR: 31 days - 3 Y L=14 TLR ≤ 30 days L=1 (1 ST) Early TLR** Imputed to 3 Y 3 Y Angio missing: L=31 BVS L=224 3 -Year BVS Qualified Angiogram L=238 TLR: 31 days - 3 Y L= 6 3 Y Angio missing: L=23 3 -Year Scheduled Angiogram 3 -Year Qualified Angiogram FU rate = 87. 3% * Full Analysis Set: Lesions received assigned devices ** Imputed to 3 years as the worst case data Co. Cr-EES L=113 3 -Year Co. Cr-EES Qualified Angiogram L=119
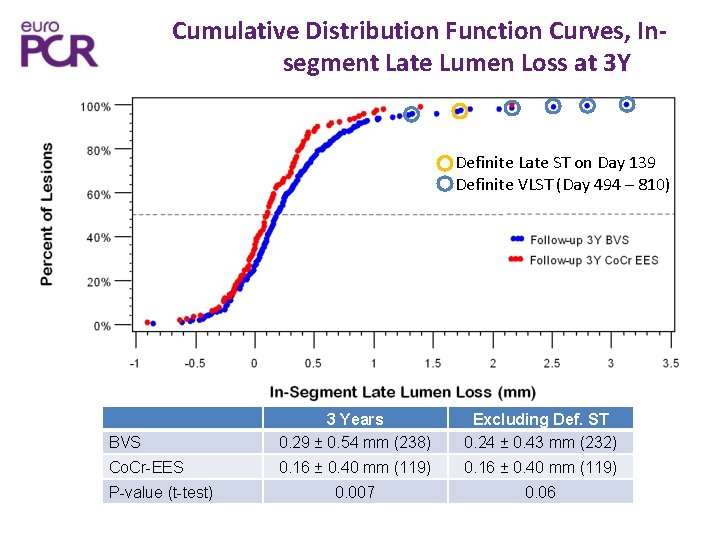
Cumulative Distribution Function Curves, Insegment Late Lumen Loss at 3 Y Definite Late ST on Day 139 Definite VLST (Day 494 – 810) BVS 3 Years 0. 29 ± 0. 54 mm (238) Excluding Def. ST 0. 24 ± 0. 43 mm (232) Co. Cr-EES 0. 16 ± 0. 40 mm (119) 0. 007 0. 06 P-value (t-test)
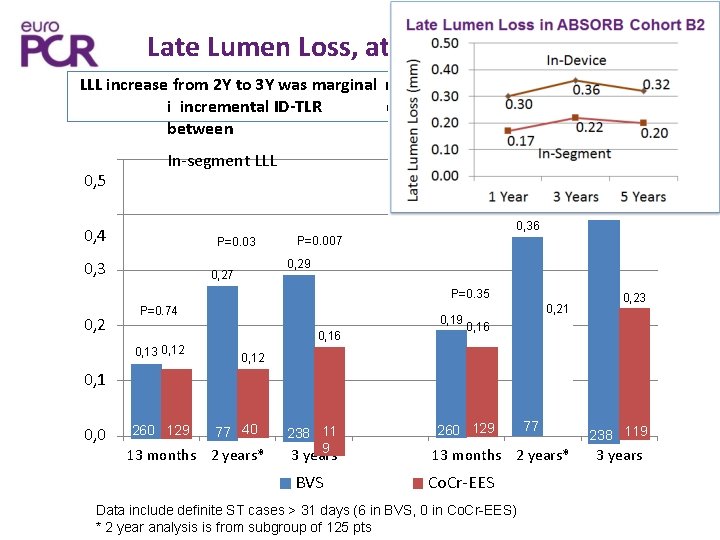
Late Lumen Loss, at 13 M, 2 Y and 3 Y LLL increase from 2 Y to 3 Y was marginal n both group Associated with low rates of 2 -3 years (1. 6% in both arms) i incremental ID-TLR between In-device LLL In-segment LLL 0, 5 P=0. 003 P=0. 04 0, 36 0, 4 P=0. 03 0, 3 P=0. 007 0, 29 0, 27 P=0. 35 0, 2 0, 39 P=0. 74 0, 13 0, 12 0, 19 0, 16 0, 21 0, 23 0, 16 0, 12 0, 1 0, 0 260 129 77 40 13 months 2 years* 238 11 9 260 129 77 40 3 years 13 months 2 years* BVS Co. Cr-EES Data include definite ST cases > 31 days (6 in BVS, 0 in Co. Cr-EES) * 2 year analysis is from subgroup of 125 pts 238 119 3 years

Limitations • Scheduled angiographic follow-up (13 M, 2 Y, 3 Y) may have impacted on the ID-TLR rate (oculo-stenotic reflex) • ABSORB Japan was initiated in 2013, i. e. at a time before adopting optimal techniques based on peer-to-peer experiences were known. Although post-dilatations were performed in ~80% of cases, the mean pressure applied was relatively low at 15. 5 atm. • ABSORB Japan is a modestly-sized trial and not powered for longer-term clinical endpoints. Results from a larger study (ABSORB III and IV) are needed to understand long-term safety

Conclusions • Between 2 and 3 years, the safety and effectiveness in ABSORB Japan was comparable for Absorb BVS and Co. Cr-EES, with incrementally low TLF rates of 1. 6% in both arms. – In both arms the increase in TLF was similarly driven by TLR Compared to the prior year, only 1 VLST occurred between 2 and 3 years, in a BVS-treated lesion with visibly high restenosis. • Angiographic late lumen loss (LLL) of BVS at 3 years was within a clinically acceptable range (< 0. 4 mm), and consistent with that observed in Cohort B. – LLL is stable with similar rates 2 and 3 years and consistent with the low ID-TLR rates reported from 2 -3 years – In ABSORB Cohort B LLL has been shown to peak at 3 -years. • While encouraging, longer term data from larger randomized studies (ABSORB III and IV) are needed to confirm the decline in incremental event rates at longer follow up.
- Slides: 17