ABSORB III Pivotal Trial Main Results Gregg W

ABSORB III: Pivotal Trial Main Results Gregg W. Stone, MD Columbia University Medical Center New. York-Presbyterian Hospital Cardiovascular Research Foundation

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship Company • Consultant • Study chairman • Reva Corp. • Abbott (uncompensated) Vascular

ABSORB III + IV Clinical Trial Program ABSORB III ~2, 250 pts with up to 2 de novo lesions in different epicardial vessels enrolled, with follow-up for at least 5 years, at up to 122 US and non-US sites 2, 008 pts were randomized 2: 1 ABSORB v XIENCE (+50 lead-in pts and 200 pt non-randomized angio/IVUS/OCT/VM FU cohort) RVD: 2. 50 - 3. 75 mm; Lesion length: ≤ 24 mm Scaffold diameters: 2. 5, 3. 0 and 3. 5 mm Scaffold lengths: 12, 18, and 28 mm Primary endpoint (n=2, 000): TLF at 1 year (powered for noninferiority) – US approval PIs: SG Ellis, DJ Kereiakes Study chairman: GW Stone
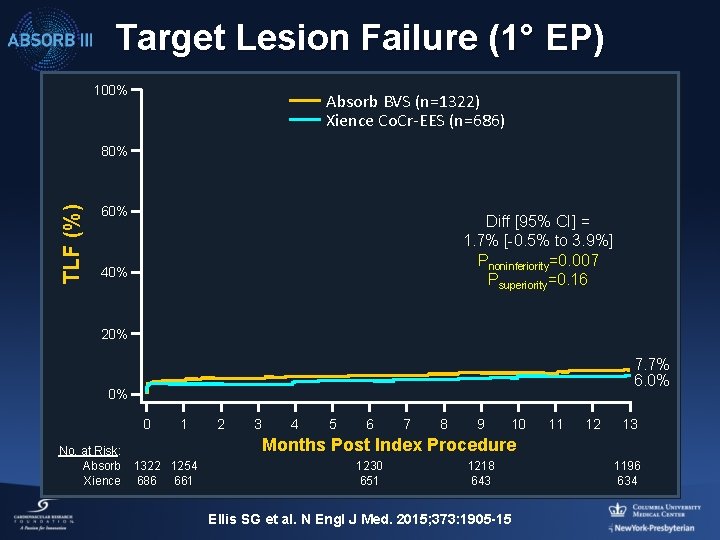
Target Lesion Failure (1° EP) 100% Absorb BVS (n=1322) Xience Co. Cr-EES (n=686) TLF (%) 80% 60% Diff [95% CI] = 1. 7% [-0. 5% to 3. 9%] Pnoninferiority=0. 007 Psuperiority=0. 16 40% 20% 7. 7% 6. 0% 0% 0 1 No. at Risk: Absorb 1322 1254 Xience 686 661 2 3 4 5 6 7 8 9 10 11 12 13 Months Post Index Procedure 1230 651 1218 643 Ellis SG et al. N Engl J Med. 2015; 373: 1905 -15 1196 634
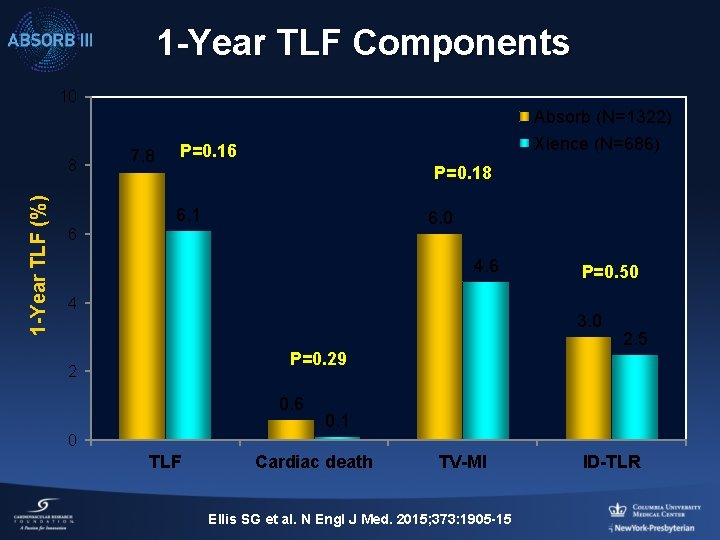
1 -Year TLF Components 10 1 -Year TLF (%) 8 7. 8 Absorb (N=1322) Xience (N=686) P=0. 16 P=0. 18 6. 1 6. 0 6 4 P=0. 50 3. 0 2. 5 P=0. 29 2 0. 6 0. 1 0 TLF Cardiac death TV-MI Ellis SG et al. N Engl J Med. 2015; 373: 1905 -15 ID-TLR

Peri-Procedural MI by Definition Absorb (N=1322) Xience (N=686) Difference p-value >3 x ULN 6. 8% 6. 6% 0. 2 0. 89 >5 x ULN (protocol) 3. 0% 2. 8% 0. 2 0. 75 >8 x ULN 1. 3% 0. 0 0. 96 >10 x ULN 0. 9% 1. 2% -0. 3 0. 58 SCAI definition* 0. 9% 1. 2% -0. 3 0. 58 CK-MB threshold *>10 x ULN or >5 x ULN with new Q waves or new persistent LBBB J Am Coll Cardiol 2013; 62: 1563 -70 Ellis SG et al. N Engl J Med. 2015; 373: 1905 -15

Device Thrombosis to 1 Year Absorb (N=1322) Xience (N=686) p-value 1. 54% 0. 74% 0. 13 - Early (0 to 30 days) 1. 06% 0. 73% 0. 46 - Late (> 30 to 1 year) 0. 46% 0. 00% 0. 10 - Definite* (1 year) 1. 38% 0. 74% 0. 21 - Probable (1 year ) 0. 15% 0. 00% 0. 55 Device Thrombosis (def*/prob) *One “definite ST” in the Absorb arm by ITT was in a pt that was treated with Xience Ellis SG et al. N Engl J Med. 2015; 373: 1905 -15
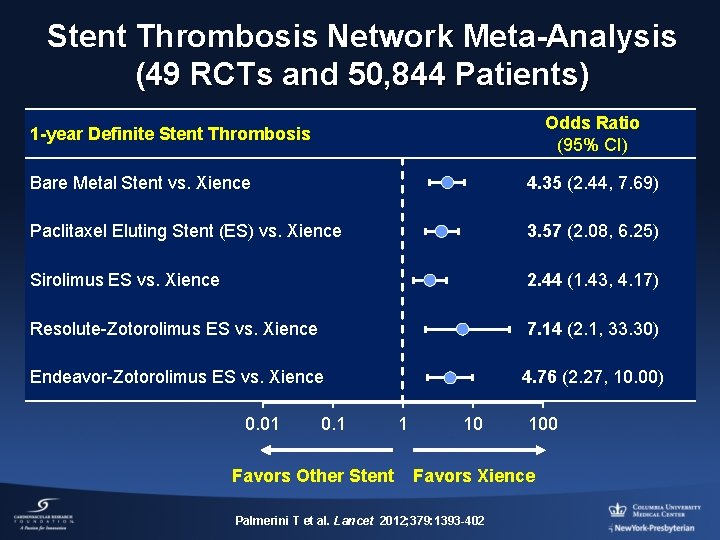
Stent Thrombosis Network Meta-Analysis (49 RCTs and 50, 844 Patients) Odds Ratio (95% CI) 1 -year Definite Stent Thrombosis Bare Metal Stent vs. Xience 4. 35 (2. 44, 7. 69) Paclitaxel Eluting Stent (ES) vs. Xience 3. 57 (2. 08, 6. 25) Sirolimus ES vs. Xience 2. 44 (1. 43, 4. 17) Resolute-Zotorolimus ES vs. Xience 7. 14 (2. 1, 33. 30) Endeavor-Zotorolimus ES vs. Xience 4. 76 (2. 27, 10. 00) 0. 01 0. 1 Favors Other Stent 1 10 100 Favors Xience Palmerini T et al. Lancet 2012; 379: 1393 -402

Stent Thrombosis Network Meta-Analysis (49 RCTs and 50, 844 Patients) + ABSORB III 1 -year Definite Stent Thrombosis Odds Ratio (95% CI) Bare Metal Stent vs. Xience 4. 35 (2. 44, 7. 69) Paclitaxel Eluting Stent (ES) vs. Xience 3. 57 (2. 08, 6. 25) Sirolimus ES vs. Xience 2. 44 (1. 43, 4. 17) Resolute-Zotorolimus ES vs. Xience 7. 14 (2. 1, 33. 30) Endeavor-Zotorolimus ES vs. Xience 4. 76 (2. 27, 10. 00) Absorb vs. Xience (ABSORB III trial) 2. 11 (0. 92, 4. 84) 0. 01 0. 1 1 10 100 Favors Other Stent Favors Xience Palmerini T et al. Lancet 2012; 379: 1393 -402
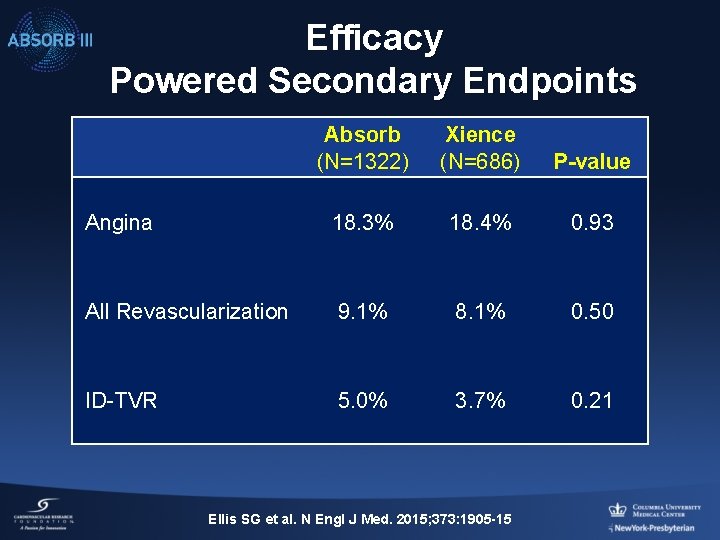
Efficacy Powered Secondary Endpoints Absorb (N=1322) Xience (N=686) P-value Angina 18. 3% 18. 4% 0. 93 All Revascularization 9. 1% 8. 1% 0. 50 ID-TVR 5. 0% 3. 7% 0. 21 Ellis SG et al. N Engl J Med. 2015; 373: 1905 -15
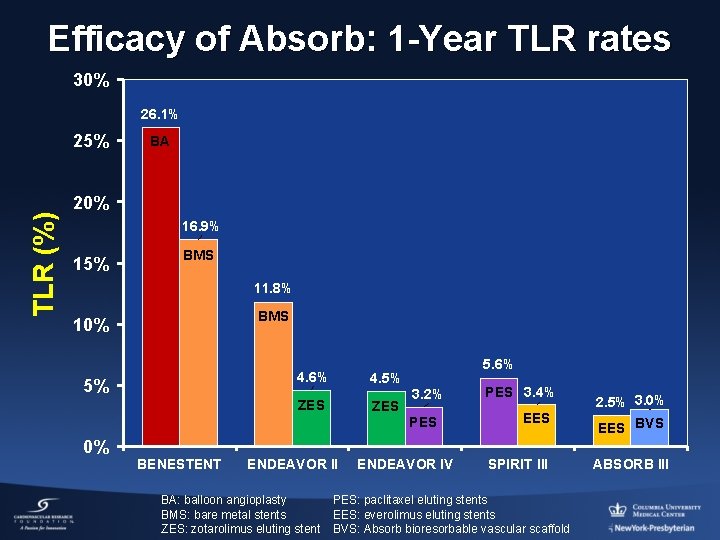
Efficacy of Absorb: 1 -Year TLR rates 30% 26. 1% TLR (%) 25% BA 20% 16. 9% 15% BMS 11. 8% BMS 10% 5% 4. 6% 4. 5% ZES 5. 6% 3. 2% PES 0% BENESTENT ENDEAVOR II BA: balloon angioplasty BMS: bare metal stents ZES: zotarolimus eluting stent ENDEAVOR IV PES 3. 4% EES 2. 5% 3. 0% EES BVS SPIRIT III ABSORB III PES: paclitaxel eluting stents EES: everolimus eluting stents BVS: Absorb bioresorbable vascular scaffold

ABSORB III Analysis In Very Small Vessels • Subgroup analyses were conducted to explore the nonsignificant differences in event rates between Absorb and Xience • Given the thicker struts of Absorb, a biologically relevant analysis was to examine outcomes in smaller vessels • Absorb BVS is intended for use in vessels with RVD 2. 5 – 3. 75 mm (by visual assessment) • We analyzed Absorb outcomes in very small vessels by QCA (Note: 2. 5 mm by visual assessment is ~2. 25 by QCA (QCA under-estimates vessel diameter)
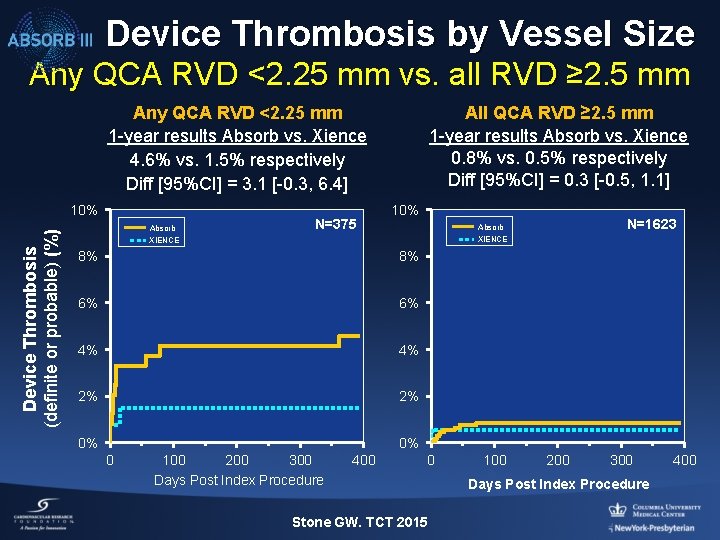
Device Thrombosis by Vessel Size Any QCA RVD <2. 25 mm vs. all RVD ≥ 2. 5 mm Any QCA RVD <2. 25 mm 1 -year results Absorb vs. Xience 4. 6% vs. 1. 5% respectively Diff [95%CI] = 3. 1 [-0. 3, 6. 4] Device Thrombosis (definite or probable) (%) 10% Absorb N=375 All QCA RVD ≥ 2. 5 mm 1 -year results Absorb vs. Xience 0. 8% vs. 0. 5% respectively Diff [95%CI] = 0. 3 [-0. 5, 1. 1] 10% 8% 8% 6% 6% 4% 4% 2% 2% 0% 0% 0 N=1623 Absorb XIENCE 100 200 300 Days Post Index Procedure 400 0 Stone GW. TCT 2015 100 200 300 Days Post Index Procedure 400
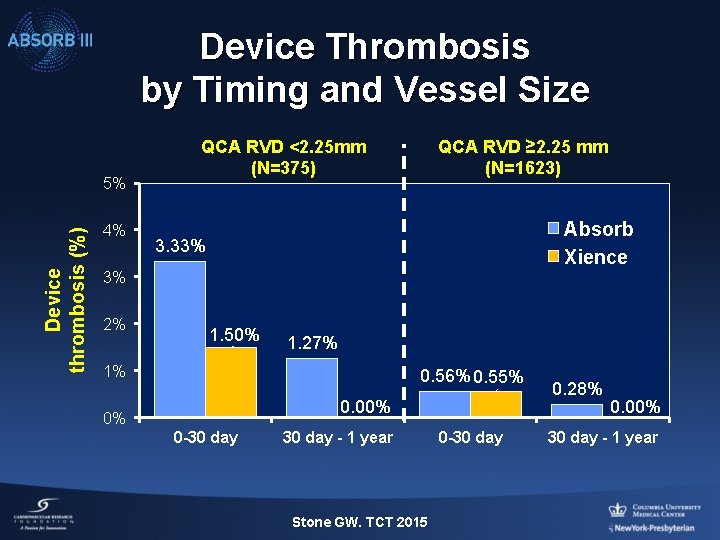
Device Thrombosis by Timing and Vessel Size Device thrombosis (%) 5% 4% QCA RVD <2. 25 mm (N=375) QCA RVD ≥ 2. 25 mm (N=1623) Absorb Xience 3. 33% 3% 2% 1. 50% 1. 27% 1% 0. 56% 0. 55% 0. 00% 0% 0 -30 day - 1 year Stone GW. TCT 2015 0 -30 day 0. 28% 0. 00% 30 day - 1 year
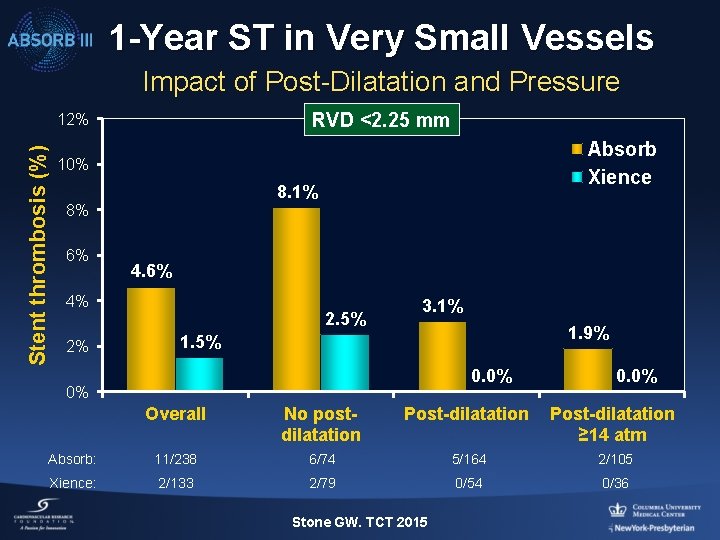
1 -Year ST in Very Small Vessels Impact of Post-Dilatation and Pressure RVD <2. 25 mm Stent thrombosis (%) 12% Absorb Xience 10% 8. 1% 8% 6% 4% 2% 2. 5% 3. 1% 1. 9% 1. 5% 0. 0% 0% 0. 0% Overall No postdilatation Post-dilatation ≥ 14 atm Absorb: 11/238 6/74 5/164 2/105 Xience: 2/133 2/79 0/54 0/36 Stone GW. TCT 2015
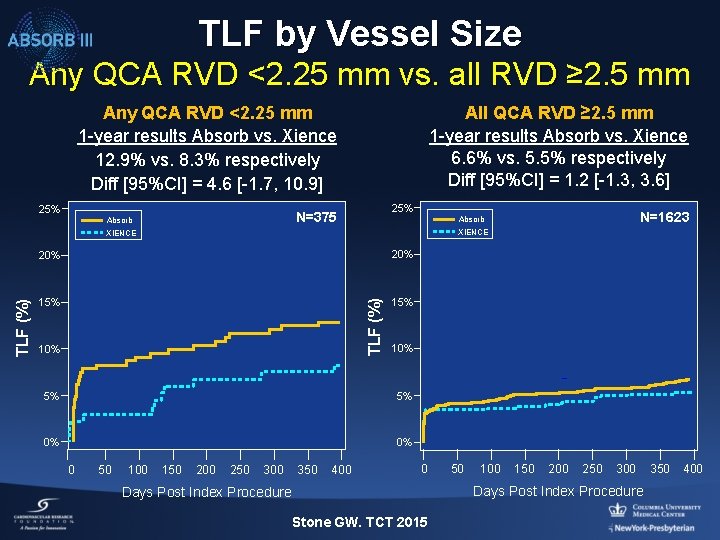
TLF by Vessel Size Any QCA RVD <2. 25 mm vs. all RVD ≥ 2. 5 mm Any QCA RVD <2. 25 mm 1 -year results Absorb vs. Xience 12. 9% vs. 8. 3% respectively Diff [95%CI] = 4. 6 [-1. 7, 10. 9] 25% All QCA RVD ≥ 2. 5 mm 1 -year results Absorb vs. Xience 6. 6% vs. 5. 5% respectively Diff [95%CI] = 1. 2 [-1. 3, 3. 6] 25% N=375 Absorb 20% 15% TLF (%) 20% 10% 5% 5% 0% 0% 0 50 100 N=1623 Absorb XIENCE 150 200 250 300 350 400 0 Days Post Index Procedure Stone GW. TCT 2015 50 100 150 200 250 300 Days Post Index Procedure 350 400
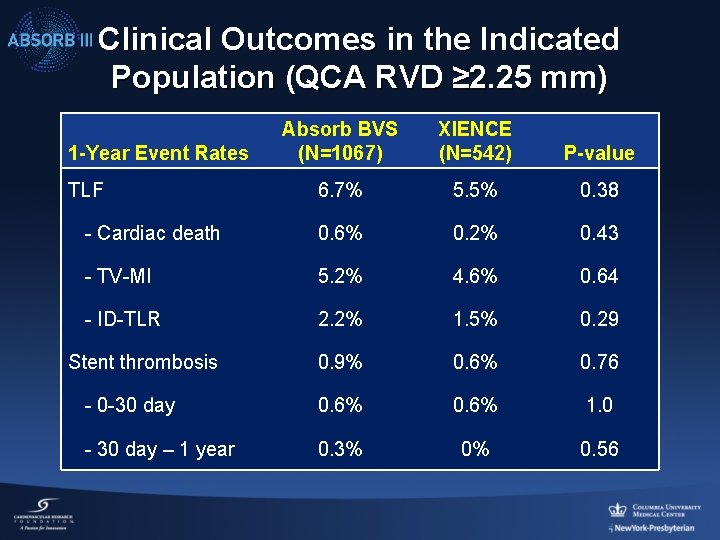
Clinical Outcomes in the Indicated Population (QCA RVD ≥ 2. 25 mm) Absorb BVS (N=1067) XIENCE (N=542) P-value 6. 7% 5. 5% 0. 38 - Cardiac death 0. 6% 0. 2% 0. 43 - TV-MI 5. 2% 4. 6% 0. 64 - ID-TLR 2. 2% 1. 5% 0. 29 0. 9% 0. 6% 0. 76 - 0 -30 day 0. 6% 1. 0 - 30 day – 1 year 0. 3% 0% 0. 56 1 -Year Event Rates TLF Stent thrombosis

ABSORB III 1 -Year Conclusions • Primary endpoint was met: Absorb BVS was noninferior to Xience for the composite safety and effectiveness endpoint of TLF at 1 year in the entire study population • Absorb BVS was highly effective, with ID-TLR rates comparable to Xience • When implanted in appropriately sized vessels, the safety profile of ABSORB was similar to Xience. In contrast, in very small vessels (those smaller than intended for this device), a signal for increased thrombosis was observed

ABSORB III 1 -Year Perspectives • The 1 -year results with this first-in-class device may be enhanced by better operator technique (appropriate lesion preparation, device sizing, more frequent post-dilatation, use of intravascular imaging, etc. ), the importance of which only became evident after trial enrollment • The comparable overall outcomes between Absorb and Xience at 1 year sets the stage for the benefits of Absorb in restoring normal coronary physiology and adaptive vascular responses to translate into improved long-term clinical outcomes, a hypothesis being tested in the ABSORB IV trial (results expected in 2020)
- Slides: 19