ABSORB EXPERIENCE Learnings to Guide Future BVS Clinical

ABSORB® EXPERIENCE Learnings to Guide Future BVS Clinical Programs Chuck Simonton MD, FACC, FSCAI CMO, Abbott Vascular Santa Clara, CA, USA Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 1 of 25

DISCLOSURES: Chuck Simonton MD, FACC, FSCAI Chief Medical Officer Divisional Vice President, Global Medical Affairs Abbott Vascular Santa Clara, CA, USA Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 2 of 25

ABSORB® Trials Learning § In the ABSORB® portfolio of trials (ABSORB II, IV, China, Japan) Absorb® was implanted in over 3, 400 patients providing an opportunity to deconstruct and understand the reasons for the early and late term events § As detailed in several publications and presentations, implantation technique, specific to vessel sizing, was identified as one of the major contributors to early and very late scaffold thrombosis (Sc. T) § Errors in vessel sizing by visual estimation, resulting in undersizing and oversizing of the bioresorbable scaffold (BRS) can contribute to Sc. T § The use of imaging with OCT in vessel sizing can result in precise sizing of the vessel diameter to scaffold diameter, in addition to exacting full scaffold expansion and strut wall apposition Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 3 of 25
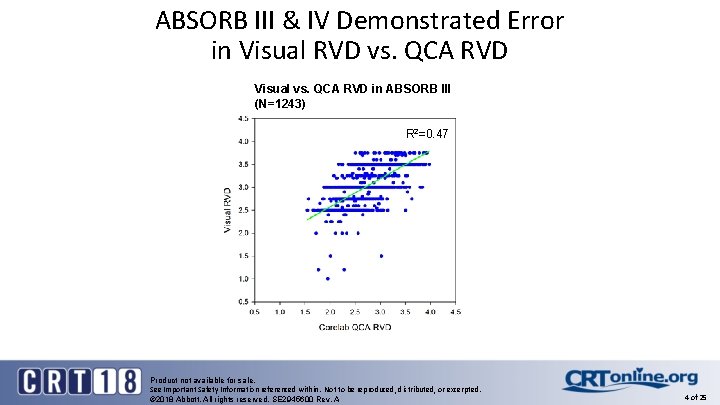
ABSORB III & IV Demonstrated Error in Visual RVD vs. QCA RVD Visual vs. QCA RVD in ABSORB III (N=1243) R 2=0. 47 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 4 of 25

ABSORB IV Physician Feedback Loop Resulted the Exclusion of Vessels < 2. 25 mm ABSORB IV: Visual vs. QCA RVD (N=747) Pre-Feedback loop R 2=0. 55 Under and oversizing of the vessel occurred despite feedback loop Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 5 of 25
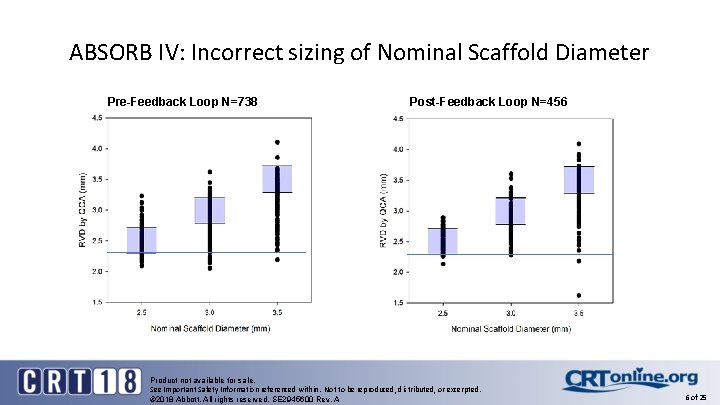
ABSORB IV: Incorrect sizing of Nominal Scaffold Diameter Pre-Feedback Loop N=738 Post-Feedback Loop N=456 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 6 of 25
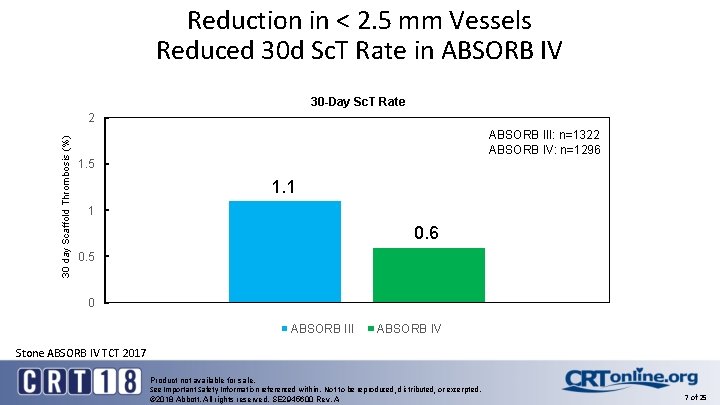
Reduction in < 2. 5 mm Vessels Reduced 30 d Sc. T Rate in ABSORB IV 30 -Day Sc. T Rate 30 day Scaffold Thrombosis (%) 2 ABSORB III: n=1322 ABSORB IV: n=1296 1. 5 1. 1 1 0. 6 0. 5 0 ABSORB III ABSORB IV Stone ABSORB IV TCT 2017 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 7 of 25
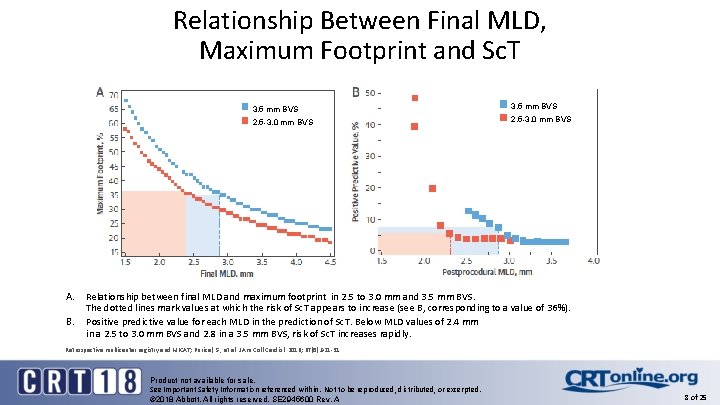
Relationship Between Final MLD, Maximum Footprint and Sc. T A. B. 3. 5 mm BVS 2. 5 -3. 0 mm BVS Relationship between final MLD and maximum footprint in 2. 5 to 3. 0 mm and 3. 5 mm BVS. The dotted lines mark values at which the risk of Sc. T appears to increase (see B, corresponding to a value of 36%). Positive predictive value for each MLD in the prediction of Sc. T. Below MLD values of 2. 4 mm in a 2. 5 to 3. 0 mm BVS and 2. 8 in a 3. 5 mm BVS, risk of Sc. T increases rapidly. Retrospective multicenter registry and MICAT; Puricel, S. , et al. J Am Coll Cardiol. 2016; 67(8): 921 -31. Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 8 of 25
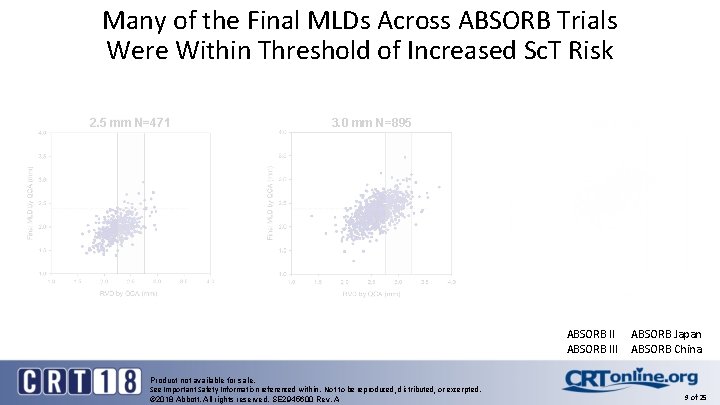
Many of the Final MLDs Across ABSORB Trials Were Within Threshold of Increased Sc. T Risk 2. 5 mm N=471 3. 0 mm N=895 3. 5 mm N=697 ABSORB III Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A ABSORB Japan ABSORB China 9 of 25
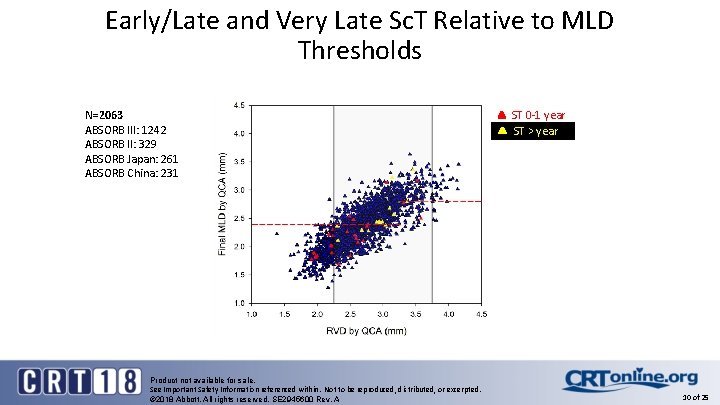
Early/Late and Very Late Sc. T Relative to MLD Thresholds N=2063 ABSORB III: 1242 ABSORB II: 329 ABSORB Japan: 261 ABSORB China: 231 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A ST 0 -1 year ST > year 10 of 25

Incorrect Vessel Sizing Creates Both Short and Long-term Risks Device thrombosis (%) Absorb XIENCE 1 – 3 Years p=0. 02 0 – 1 Year 5% HR [95% CI] = 2. 08 [0. 78, 5. 55] 4% p=0. 13 3% Sc. T Risk 0 -1 year Underexpansion Sc. T Risk 0 -1 year Malapposition 2% 1. 5% 1% 0. 7% 0% 0 Ellis ABSORB III TCT 2017 No. at Risk: Absorb® XIENCE 6 12 0. 8% 0. 3% 0. 0% 18 24 0. 0% 30 36 42 Time Post Procedure (Months) 1322 686 1273 668 1256 651 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 1211 634 11 of 25
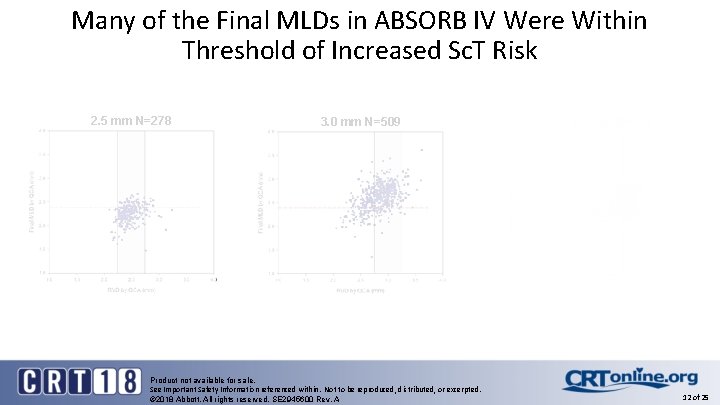
Many of the Final MLDs in ABSORB IV Were Within Threshold of Increased Sc. T Risk 2. 5 mm N=278 3. 0 mm N=509 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 3. 5 mm N=401 12 of 25
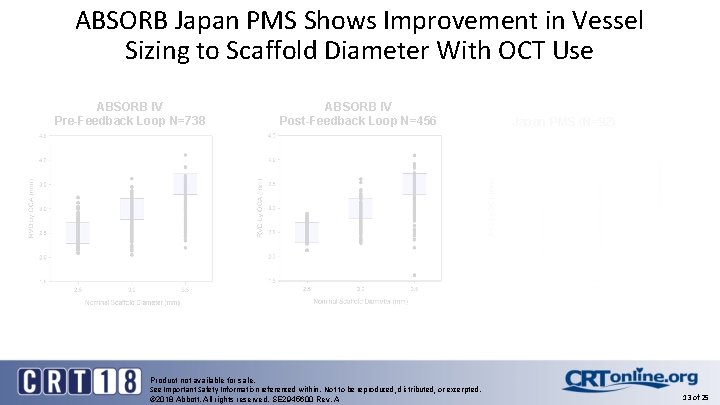
ABSORB Japan PMS Shows Improvement in Vessel Sizing to Scaffold Diameter With OCT Use ABSORB IV Pre-Feedback Loop N=738 ABSORB IV Post-Feedback Loop N=456 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A Japan PMS (N=92) 13 of 25

Key Steps to Success with Absorb 1. P: Prep the Lesion: NC balloon sized 1: 1 For calcified lesions: scoring balloons, cutting balloon (rotablator/CSI****) 2. S: Size the Scaffold: Guide catheter (eye), predil balloon, online QCA, or invasive imaging (IVUS, OCT) 3. P: Post-Dilate with NC Balloon sized 1: 1 or higher to high pressure (>16 A)

ABSORB PSP Impact of Technique on Early and Late Outcomes Following Coronary Bioresorbable Scaffold Implantation Gregg W. Stone, MD Columbia University Medical Center New. York-Presbyterian Hospital Cardiovascular Research Foundation
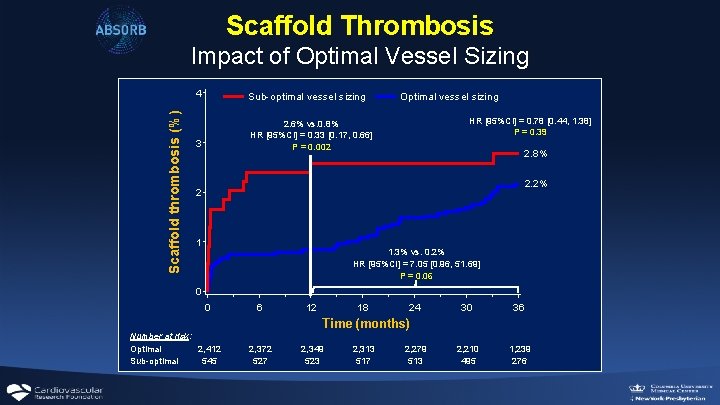
Scaffold Thrombosis Scaffold thrombosis (%) Impact of Optimal Vessel Sizing 4 Sub-optimal vessel sizing 3 2. 6% vs. 0. 8% HR [95%CI] = 0. 33 [0. 17, 0. 66] P = 0. 002 Optimal vessel sizing HR [95%CI] = 0. 78 [0. 44, 1. 38] P = 0. 39 2. 8% 2. 2% 2 1 1. 3% vs. 0. 2% HR [95%CI] = 7. 05 [0. 96, 51. 69] P = 0. 06 0 0 6 12 18 24 30 36 2, 210 495 1, 239 276 Time (months) Number at risk: Optimal 2, 412 Sub-optimal 545 2, 372 527 2, 349 523 2, 313 517 2, 279 513
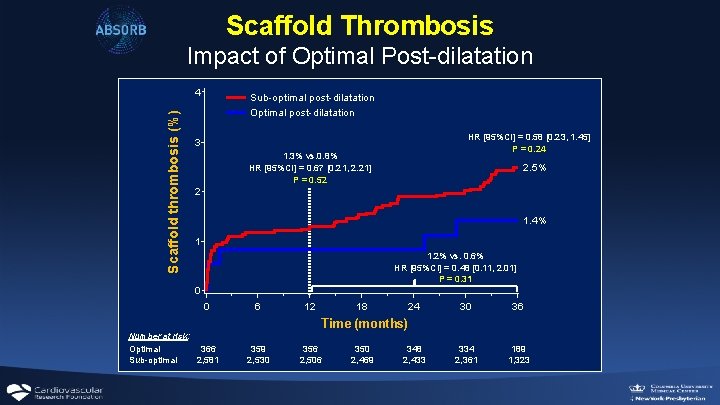
Scaffold Thrombosis Impact of Optimal Post-dilatation Scaffold thrombosis (%) 4 Sub-optimal post-dilatation Optimal post-dilatation HR [95%CI] = 0. 58 [0. 23, 1. 45] P = 0. 24 3 1. 3% vs. 0. 8% HR [95%CI] = 0. 67 [0. 21, 2. 21] P = 0. 52 2. 5% 2 1. 4% 1 1. 2% vs. 0. 6% HR [95%CI] = 0. 48 [0. 11, 2. 01] P = 0. 31 0 0 6 12 18 24 30 36 334 2, 361 189 1, 323 Time (months) Number at risk: Optimal 366 Sub-optimal 2, 581 359 2, 530 356 2, 506 350 2, 469 348 2, 433

Conclusion § Error in visual estimation is detrimental to BRS technology due to the risk of under-expansion or malapposition, resulting in increased risk of early/late or very late Sc. T § Thicker struts of 1 st Gen bioresorbable (BRS) technology requires a minimum value for MLD to reduce flow disturbances that can cause early Sc. T § Degrading and resorbing nature of BRS necessitates full apposition to vessel wall, to minimize very late events. Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 18 of 25

Conclusion (cont. ) § Absolute exclusion of very small vessels and malappositions can only be detected and eliminated with OCT § Operators in ABSORB Japan PMS used OCT with no pre-specified vessel sizing protocol, and showed improvements in vessel sizing to scaffold diameter § BRS requires precise PCI with the use of OCT to support the success of this technology Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 19 of 25

Important Safety Information The XIENCE V®, XIENCE nano®, XIENCE PRIME® LL, XIENCE Xpedition® SV and XIENCE Xpedition® LL , XIENCE Alpine. TM (XIENCE Family) of Everolimus Eluting Coronary Stents on the MULTI-LINK VISION® or MULTI-LINK MINI VISION® Delivery Systems INDICATIONS The XIENCE Family of Everolimus Eluting Coronary Stent Systems are indicated for improving coronary luminal diameter in patients, including those with diabetes mellitus, with symptomatic heart disease due to de novo native coronary artery lesions for XIENCE V (length ≤ 28 mm), XIENCE PRIME, XIENCE Xpedition and XIENCE Alpine (lengths ≤ 32 mm) with reference vessel diameters of ≥ 2. 25 mm to ≤ 4. 25 mm. Additionally, the entire XIENCE Family is indicated for treating de novo chronic total coronary occlusions. CONTRAINDICATIONS The XIENCE Family of stents is contraindicated for use in patients: • Who cannot receive antiplatelet and/or anti-coagulant therapy • With lesions that prevent complete angioplasty balloon inflation or proper placement of the stent or stent delivery system • With hypersensitivity or contraindication to everolimus or structurally-related compounds, cobalt, chromium, nickel, tungsten, acrylic, and/or fluoropolymers. §WARNINGS • Ensure that the inner package sterile barrier has not been opened or damaged prior to use. • Judicious patient selection is necessary because the use of this device carries the associated risk of stent thrombosis, vascular complications, and/or bleeding events. • This product should not be used in patients who are not likely to comply with the recommended antiplatelet therapy. PRECAUTIONS • Stent implantation should only be performed by physicians who have received appropriate training. Stent placement should be performed at hospitals where emergency coronary artery bypass graft surgery is accessible. • Subsequent restenosis may require repeat dilatation of the arterial segment containing the stent. Long-term outcomes following repeat dilatation of the stent are presently unknown. • Risks and benefits should be considered in patients with severe contrast agent allergies. • Care should be taken to control the guiding catheter tip during stent delivery, deployment and balloon withdrawal. Before withdrawing the stent delivery system, visually confirm complete balloon deflation by fluoroscopy to avoid guiding catheter movement into the vessel and subsequent arterial damage. • Stent thrombosis is a low-frequency event that is frequently associated with myocardial infarction (MI) or death. • When DES are used outside the specified Indications for Use, patient outcomes may differ from the results observed in the SPIRIT family of trials • Compared to use within the specified Indications for Use, the use of DES in patients and lesions outside of the labeled indications may have an increased risk of adverse events, including stent thrombosis, stent embolization, MI, or death. • Orally administered everolimus combined with cyclosporine is associated with increased serum cholesterol and triglycerides levels. • A patient’s exposure to drug and polymer is proportional to the number and total length of implanted stents. See Instructions for Use for current data on multiple stent implantation. Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A • Safety and effectiveness of the XIENCE Family of stents have not been established for subject populations with the following clinical settings: • Patients with prior target lesion or in-stent restenosis related brachytherapy, patients in whom mechanical atherectomy devices or laser angioplasty catheters are used in conjunction with XIENCE Family stents, women who are pregnant or lactating, men intending to father children, pediatric patients, unresolved vessel thrombus at the lesion site, coronary artery reference vessel diameters < 2. 25 mm or > 4. 25 mm or lesion length > 32 mm, lesions located in saphenous vein grafts, unprotected left main coronary artery, ostial lesions, lesions located at a bifurcation or previously stented lesions, diffuse disease or poor flow (TIMI < 1) distal to the identified lesions, excessive tortuosity proximal to or within the lesion, recent acute myocardial infarction (AMI) or evidence of thrombus in target vessel multivessel disease, and in-stent restenosis • Everolimus has been shown to reduce the clearance of some • • prescription medications when administered orally along with cyclosporine (Cs. A). Formal drug interaction studies have not been performed with the XIENCE Family of stents because of limited systemic exposure to everolimus eluted from the stent. Everolimus is an immunosuppressive agent. Consideration should be given to patients taking other immunosuppressive agents or who are at risk for immune suppression. Oral everolimus use in renal transplant patients and advanced renal cell carcinoma patients was associated with increased serum cholesterol and triglycerides, which in some cases required treatment. Nonclinical testing has demonstrated that the XIENCE Family of stents, in single and in overlapped configurations are MR conditional up to 68 mm in length for XIENCE V and XIENCE nano only and up to 71 mm in length for all other XIENCE Family stents. It can be scanned safely under the conditions in the Instructions for Use. The XIENCE Family of stents should be handled, placed, implanted, and removed according to the Instructions for Use. 20 of 25

Important Safety Information The XIENCE V®, XIENCE nano®, XIENCE PRIME® LL, XIENCE Xpedition® SV and XIENCE Xpedition® LL , XIENCE Alpine. TM (XIENCE Family) of Everolimus Eluting Coronary Stents on the MULTI-LINK VISION® or MULTI-LINK MINI VISION® Delivery Systems (continued) POTENTIAL ADVERSE EVENTS § Adverse events (in alphabetical order) which may be associated with percutaneous coronary and treatment procedure including coronary stent use in native coronary arteries include, but are not limited to: • Abrupt closure, Access site pain, hematoma, or hemorrhage, Acute myocardial infarction, Allergic reaction or hypersensitivity to contrast agent or cobalt, chromium, nickel, tungsten, acrylic and fluoropolymers; and drug reactions to antiplatelet drugs or contrast agent, Aneurysm, Arterial perforation and injury to the coronary artery, Arterial rupture, Arteriovenous fistula, Arrhythmias, atrial and ventricular, Bleeding complications, which may require transfusion, Cardiac tamponade, Coronary artery spasm, Coronary or stent embolism, Coronary or stent thrombosis, Death, Dissection of the coronary artery, Distal emboli (air, tissue or thrombotic), Emergent or non-emergent coronary surgery, Fever, Hypotension and / or hypertension, Infection and pain at insertion site, Injury to the coronary artery, Ischemia (myocardial), Myocardial infarction (MI), Nausea and vomiting, Palpitations, Peripheral ischemia (due to vascular injury), Pseudoaneurysm, Renal Failure, Restenosis of the stented segment of the artery, Shock/pulmonary edema, Stroke / cerebrovascular accident (CVA), Total occlusion of coronary artery, Unstable or stable angina pectoris, Vascular complications including at the entry site which may require vessel repair, Vessel dissection § Adverse events associated with daily oral administration of everolimus to organ transplant patients include but are not limited to: • Abdominal pain (including upper abdominal pain); Anemia; Angioedema; Anorexia; Asthenia; Constipation; Cough; Delayed wound healing/fluid accumulation; Diarrhea; Dyslipidemia (including hyperlipidemia and hypercholesterolemia); Dyspnea; Dysgeusia; Dyspepsia; Dysuria; Dry skin; Edema (peripheral); Epistaxis; Fatigue; Headache; Hematuria; Hyperglycemia (may include new onset of diabetes); Hyperlipidemia; Hyperkalemia; Hypertension; Hypokalemia; Hypomagnesemia; Hypophosphatemia; Increased serum creatinine; Infections and serious infections: bacterial, viral, fungal, and protozoal infections (may include herpes virus infection, polyoma virus infection which may be associated with BK virus associated nephropathy, and/or other opportunistic infections); Insomnia; Interaction with strong inhibitors and inducers of CY 3 PA 4 or Pg. P; Leukopenia; Lymphoma and other malignancies (including skin cancer); Male infertility (azospermia and/or oligospermia); Mucosal inflammation (including oral ulceration and oral mucositis); Nausea; Neutropenia; Non-infectious pneumonitis; Pain: extremity, incision site and procedural, back, chest, and musculoskeletal; Proteinuria; Pruritus; Pyrexia; Rash; Stomatitis; Thrombocytopenia; Thrombotic microangiopathy (TMA)/Thrombotic thrombocytopenic purpura (TTP)/ Hemolytic uremic syndrome (HUS); Tremor; Urinary tract infection; Upper respiratory tract infection; Vomiting • Live vaccines should be avoided and close contact with those that have had live vaccines should be avoided. Fetal harm can occur when administered to a pregnant woman. There may be other potential adverse events that are unforeseen at this time. Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 21 of 25

Important Safety Information Absorb GT 1 TM Bioresorbable Vascular Scaffold System INDICATIONS The Absorb GT 1 Bioresorbable Vascular Scaffold (BVS) is a temporary scaffold that will fully resorb over time and is indicated for improving coronary luminal diameter in patients with ischemic heart disease due to de novo native coronary artery lesions (length ≤ 24 mm) with a reference vessel diameter of ≥ 2. 5 mm and ≤ 3. 75 mm. CONTRAINDICATIONS The Absorb GT 1 BVS System is contraindicated for use in: • Patients who cannot tolerate, including allergy or hypersensitivity to, procedural anticoagulation or post-procedural antiplatelet regimen. • Patients with a known allergy or hypersensitivity to everolimus or structurally related compounds, device materials (poly [L-lactide], poly [D, L-lactide], platinum), or contrast medium who cannot be adequately premedicated. WARNINGS • Careful selection of the target lesion reference vessel diameter to the scaffold diameter is required to minimize potential damage to the scaffold during post-dilatation and ensure adequate scaffold apposition and appropriate minimum lumen diameter. • Adequate lesion preparation prior to scaffold implantation is required to ensure safe delivery of the scaffold across the target lesion. It is not recommended to treat patients having a lesion that prevents complete inflation of an angioplasty balloon. It is strongly recommended to achieve a residual stenosis between 20% and 40% after pre-dilatation to enable successful delivery and full expansion of the scaffold. • Ensure the scaffold is not post-dilated beyond the allowable expansion limits ( see Absorb GT 1 IFU Section 12. 7 - Clinician Use Information, Further Expansion of the Deployed Scaffold). • Antiplatelet therapy should be administered post-procedure (see Absorb GT 1 IFU Section 9. 1 - Patient Selection and Treatment, Individualization of Treatment). • This product should not be used in patients who are not likely to comply with the recommended antiplatelet therapy. • Judicious selection of patients is necessary, since the use of this device carries the associated risk of scaffold thrombosis, vascular complications, and/or bleeding events. • If quantitative imaging determines a vessel size < 2. 5 mm, do not implant the Absorb GT 1 BVS. Implantation of the device in vessels < 2. 5 mm may lead to an increased risk of adverse events such as myocardial infarction and scaffold thrombosis. PRECAUTIONS • Implantation of the scaffold should be performed only by physicians who have received appropriate training. • Do not exceed the Rated Burst Pressure (RBP) as indicated on the product label. • Post-dilatation is strongly recommended for optimal scaffold apposition. When performed, post-dilatation should be at high pressure (> 16 atm) with a noncompliant balloon. • Care must be taken to properly size the scaffold to ensure that the scaffold is in full contact with the arterial wall upon deflation of the balloon. All efforts should be made to assure that the scaffold is not under dilated. Refer to Absorb GT 1 IFU Section 12. 7 Clinical Use Information – Further Expansion of the Deployed Scaffold • In small vessels (visually assessed as ≤ 2. 75 mm), on-line QCA or intravascular imaging is strongly recommended to accurately measure and confirm appropriate vessel sizing (≥ 2. 5 mm). • Balloon dilatation of any cells of a deployed Absorb GT 1 BVS may cause scaffold damage. Avoid scaffolding across any side branches ≥ 2. 0 mm in diameter. Placement of a scaffold has the potential to compromise side branch patency. • It is not recommended to treat patients having a lesion with excessive tortuosity proximal to or within the lesion. • Non-clinical testing has demonstrated the Absorb GT 1 BVS is MR Conditional. A patient with this device can be safely scanned in all MR environments 3 T or less. • Safety and effectiveness of the Absorb GT 1 BVS have not been established for subject populations with the following characteristics: • Coronary artery reference vessel diameters < 2. 5 mm or > 3. 75 mm • Lesion lengths >24 mm • Lesions located in arterial or saphenous vein grafts • Lesions located in unprotected left main artery • Ostial lesions • Lesions located at a bifurcation • Previously stented lesions • Moderate to severe calcification • Chronic total occlusion or poor flow (< TIMI 1) distal to the identified lesions • Three-vessel disease • Unresolved thrombus at the lesion site or anywhere in the vessel to be treated • Excessive tortuosity proximal to or within the lesion • Recent acute myocardial infarction (AMI) POTENTIAL ADVERSE EVENTS Adverse events that may be associated with PCI, treatment procedures and the use of a coronary scaffold in native coronary arteries include the following, but are not limited to: Allergic reaction or hypersensitivity to latex, contrast agent, anesthesia, device materials (platinum, or polymer [poly (L-lactide) (PLLA), polymer poly (D, L-lactide) (PDLLA)]), and drug reactions to everolimus, anticoagulation, or antiplatelet drugs, Vascular access complications which may require transfusion or vessel repair, including: , Catheter site reactions, Bleeding (ecchymosis, oozing, hematoma, hemorrhage, retroperitoneal hemorrhage), Arteriovenous fistula, pseudoaneurysm, dissection, perforation / rupture, Embolism (air, tissue, plaque, thrombotic material or device), Peripheral nerve injury, Peripheral ischemia, Coronary artery complications which may require additional intervention, including: , Total occlusion or abrupt closure, Arteriovenous fistula, pseudoaneurysm, dissection, perforation / rupture, Tissue prolapse / plaque shift, Embolism (air, tissue, plaque, thrombotic material or device), Coronary or scaffold thrombosis (acute, subacute, late, very late), Stenosis or restenosis, Pericardial complications which may require additional intervention, including: , Cardiac tamponade, Pericardial effusion, Pericarditis, Cardiac arrhythmias (including conduction disorders, atrial and ventricular arrhythmias), Cardiac ischemic conditions (including myocardial ischemia, myocardial infarction [including acute], coronary artery spasm and unstable or stable angina pectoris), Stroke / Cerebrovascular accident (CVA) and Transient Ischemic Attack (TIA), System organ failures: , Cardio-respiratory arrest, Cardiac failure, Cardiopulmonary failure (including pulmonary edema), Renal insufficiency / failure, Shock, Blood cell disorders (including Heparin Induced Thrombocytopenia [HIT]), Hypotension / hypertension, Infection, Nausea and vomiting, Palpitations, dizziness, and syncope, Chest pain, Fever, Pain, Death. Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 22 of 25

Caution: This product is intended for use by or under the direction of a physician. Prior to use, reference the Instructions for Use provided inside the product carton (when available), at eifu. abbottvascular. com or at Manuals. sjm. com for more detailed information on Indications, Contraindications, Warnings, Precautions and Adverse Events Tests performed by and data on file at Abbott. Illustrations are artist’s representations only and should not be considered as engineering drawings or photographs. Photos on file at Abbott 3200 Lakeside Dr. , Santa Clara, CA. 95054 USA, Tel: 1. 800. 227. 9902 Absorb is a trademark of the Abbott Group of Companies. www. Vascular. Abbott © 2018 Abbott. All rights reserved. SE 2945600 Rev. A Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 23 of 25

BACK-UP SLIDES Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 24 of 25

ABSORB Japan RCT OCT Cohorts Experienced Error in Vessel Sizing OCT 1 (N=72) OCT 2 (N=82) Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 25 of 25
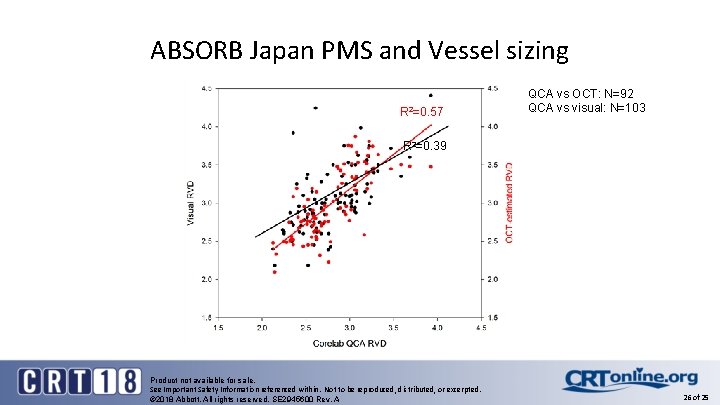
ABSORB Japan PMS and Vessel sizing R 2=0. 57 QCA vs OCT: N=92 QCA vs visual: N=103 R 2=0. 39 Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 26 of 25

ABSORB RCT Japan and ABSORB Japan PMS Japan RCT (N=261) Japan PMS (N=123) Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 27 of 25
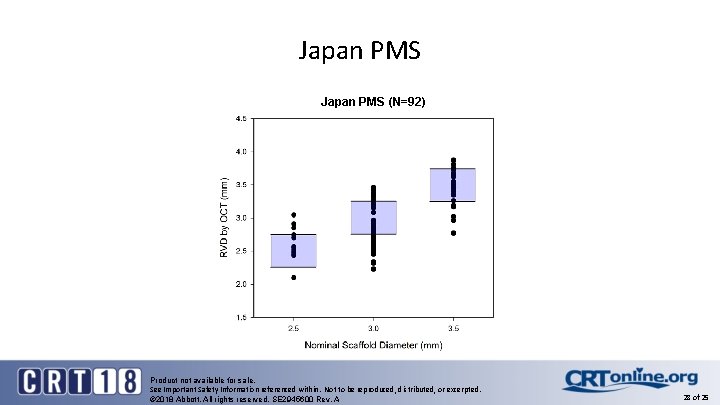
Japan PMS (N=92) Product not available for sale. See Important Safety Information referenced within. Not to be reproduced, distributed, or excerpted. © 2018 Abbott. All rights reserved. SE 2945600 Rev. A 28 of 25
- Slides: 28