Absolute Dating Its Atomic Absolute Dating Age of

Absolute Dating It’s Atomic

Absolute Dating • Age of an earth material or event in years Relative age 3 rd 2 nd 1 st youngest Absolute 12 kya 1. 7 bya 8 kya

Early Earth’s Age Attempts • Bishop Ussher • Biblical interpretations • October 23 rd 4004 BC • Sedimentation rates • 3 my – 500 my • Halley/Joly • Ocean Salinity • 100 my • Lord Kelvin • Rate of planetary cooling • 30 my

Radioactive Revolution • Late 19 th century • Discovered by “accident” • Spontaneous transformation of an element into another isotope of the same element Henri Becquerel Marie Curie

Review of Atom • Protons – positively charged • Neutron – neutral • Electron – negatively charged • Isotope – element with different number of neutrons (same element different mass)

Two Main Types of Decay • Alpha Decay • Losses 2 protons and 2 neutrons • Mass decreases by 4 • Atomic # decreases by 2 • Essentially = loss of a positively charged He ion

• Beta-minus Decay • Neutron splits into proton and electron • Atomic number increases by one • Mass remains the same
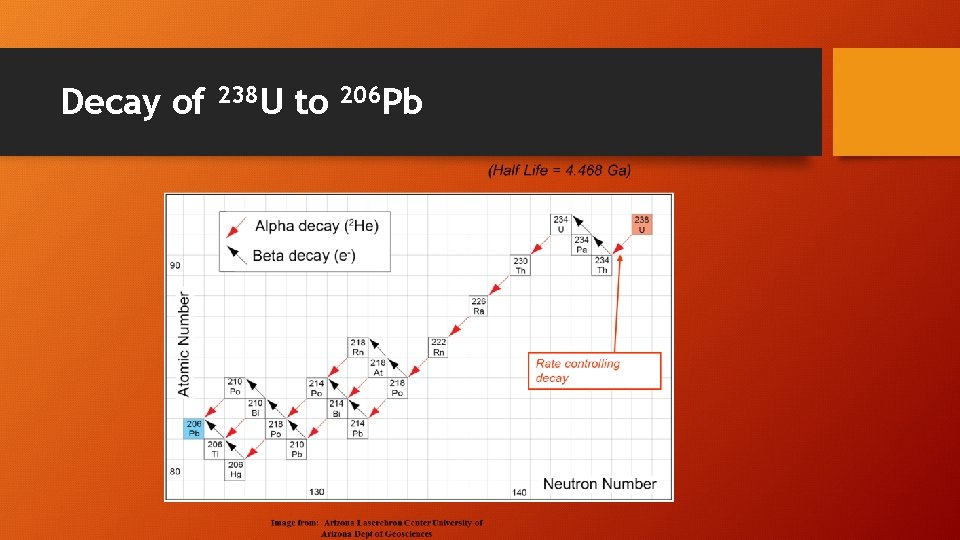
Decay of 238 U to 206 Pb
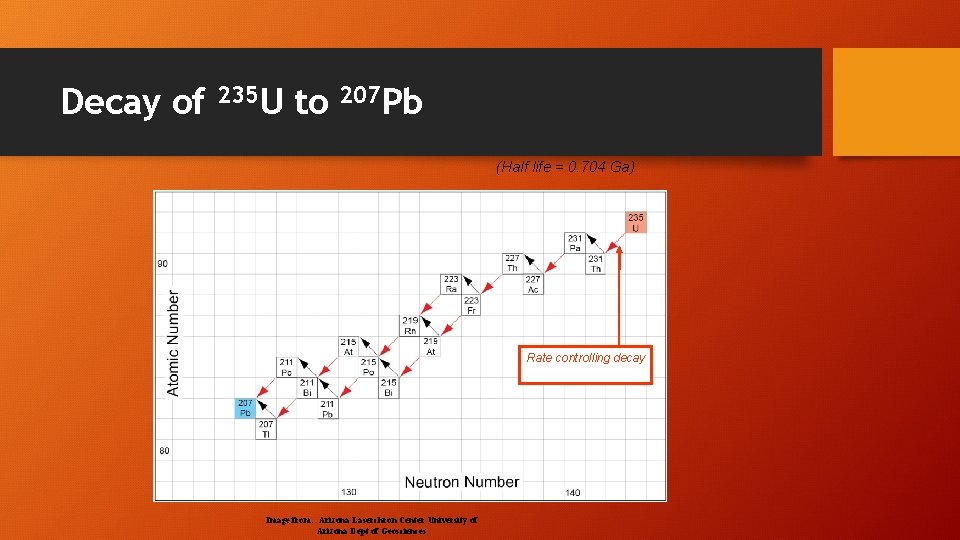
Decay of 235 U to 207 Pb (Half life = 0. 704 Ga) Rate controlling decay Image from: Arizona Laserchron Center University of Arizona Dept of Geosciences

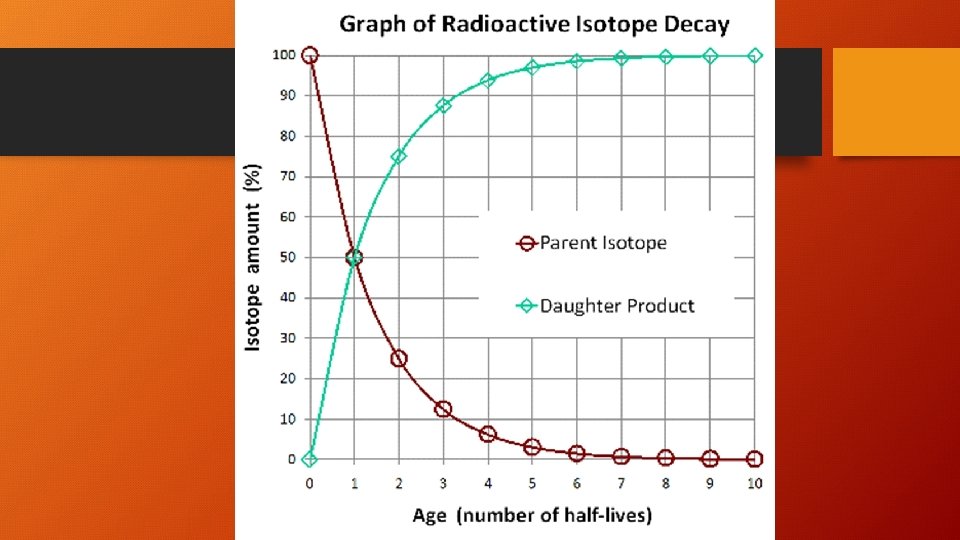
How do we know age? • Half-life • Period of time during which half the parent atoms in a closed system decay to form daughter atoms • Spontaneous • Unique for each element
Example

Sample Problem Sample: 1024 grams Isotope A (10 k) half-life Isotope B Daughter Parent 960 grams 64 grams 10, 000 20, 000 30, 000 40, 000 1 st 2 nd 3 rd 4 th 512 / 512 256 / 768 128 / 896 64 / 960
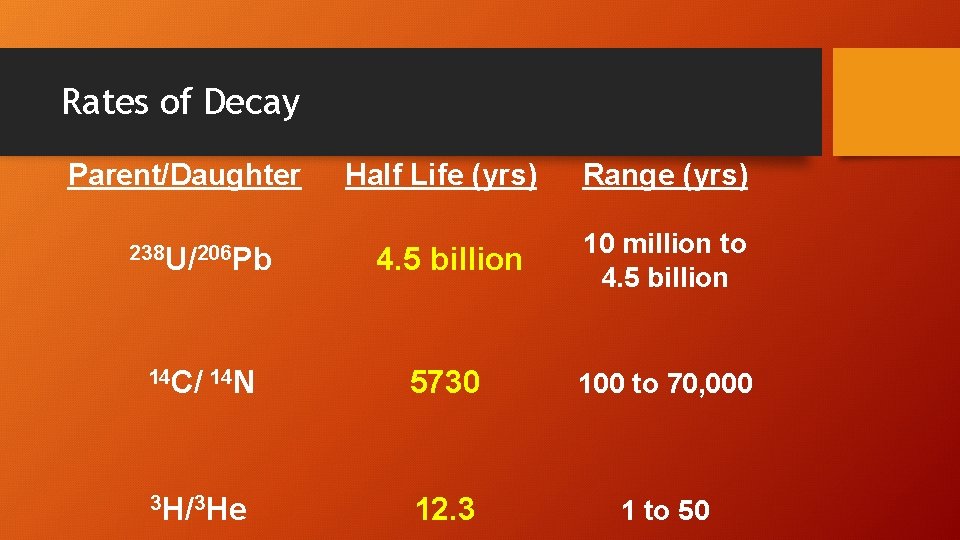
Rates of Decay Parent/Daughter Half Life (yrs) Range (yrs) 238 U/206 Pb 4. 5 billion 10 million to 4. 5 billion 14 C/ 14 N 5730 100 to 70, 000 3 H/3 He 12. 3 1 to 50

Uses Parent/Daughter Material Dated 238 U/206 Pb Igneous and Metamorphic rocks 14 C/ 14 N Organic material 3 H/3 He Water
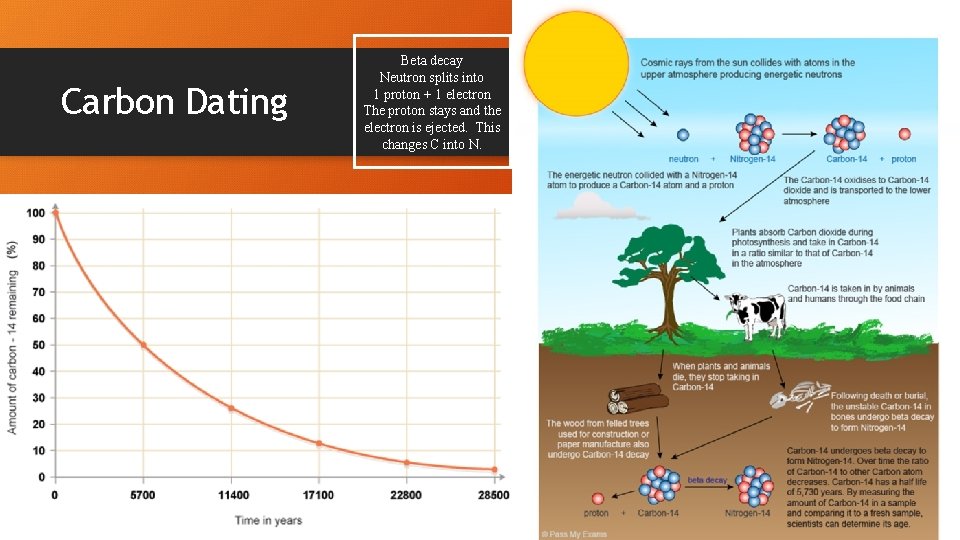
Carbon Dating Beta decay Neutron splits into 1 proton + 1 electron The proton stays and the electron is ejected. This changes C into N.

Uranium (U) – Thorium (Th) – Lead (Pb) • U-238 decays to Pb-206 • Half life = 4. 5 billion years • U-235 decays to Pb-207 • Half life = 713 million years • Th-232 decays to Pb-208 • Half life = 14. 1 billion years


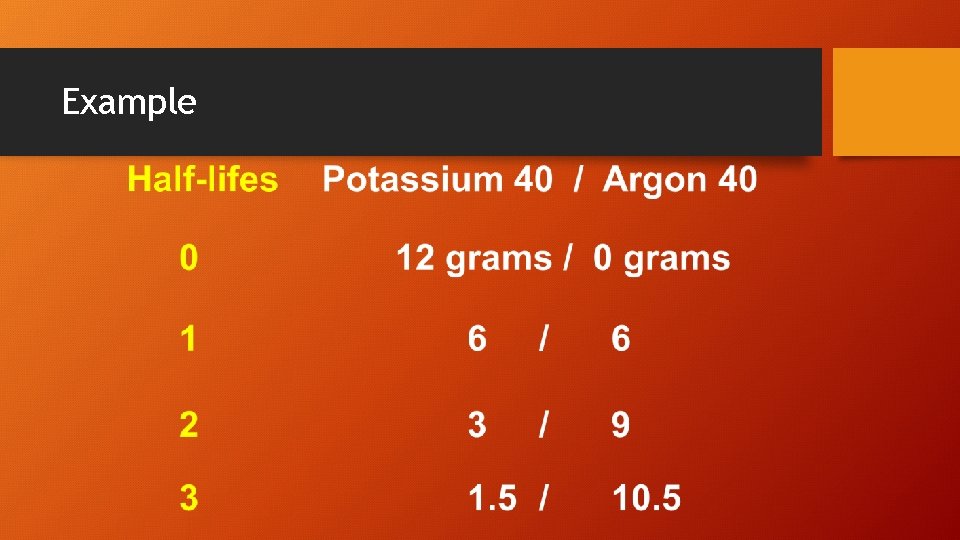
Potassium (K) – Argon (Ar) • • Potassium is extremely common Half-life is 1. 3 billion years Dating range from 100, 000 to 4. 6 billion years Can be useful for young or old rocks

Practice What is the Parent / Daughter ratio of a sample of radiogenic material if the original sample was 200 g. and 3 half-lifes have passed. If the half-life is 20, 000 years, how old is the sample? What is the half-life of an isotopic pair if the sample is 2 million years old and contains 625 g of parent and 9375 g of daughter isotope?
- Slides: 20