Absolute Age of Rocks Notes Absolute Dating When

Absolute Age of Rocks Notes
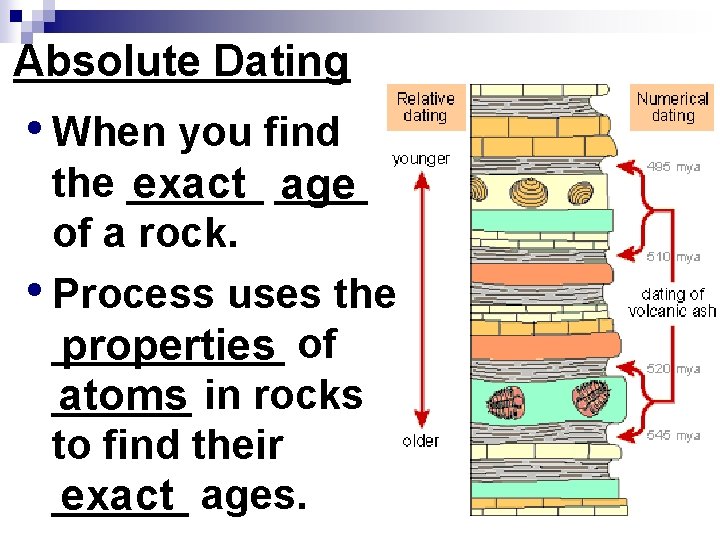
Absolute Dating • When you find the ______ exact ____ age of a rock. • Process uses the _____ properties of ______ atoms in rocks to find their ______ exact ages.

three kinds of • Atoms have _____ particles protons, ____: _____ neutrons and electrons • Atoms of the ________ same element have the ______ number of ____, same but can have a ____ number protonsof _____ different neutrons

• Isotope: atoms of an ____ element that have ____ neutrons different numbers of _____ • Ex: These are some isotopes of carbon • Carbon-12 has __ 6 neutrons 7 neutrons • Carbon-13 has __ • Carbon-14 has __ 8 neutrons Carbon-12 stable Carbon-13 stable Carbon-14 unstable
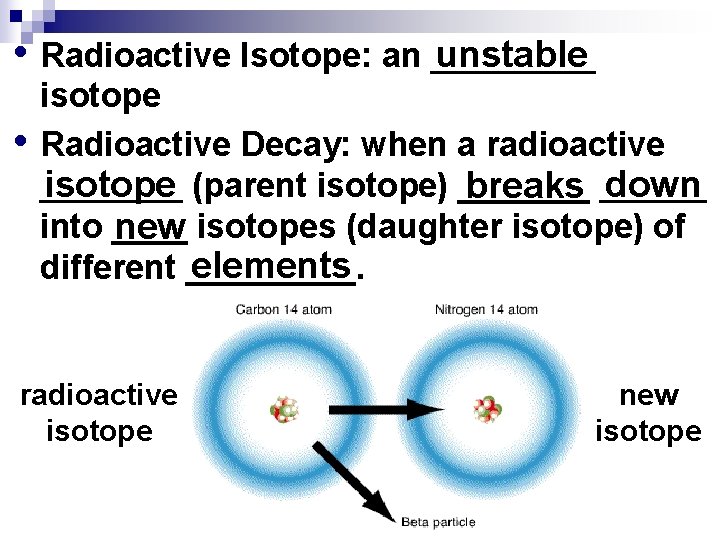
• Radioactive Isotope: an _____ unstable • isotope Radioactive Decay: when a radioactive down isotope (parent isotope) ________ breaks ______ into ____ new isotopes (daughter isotope) of elements different _____. radioactive isotope new isotope
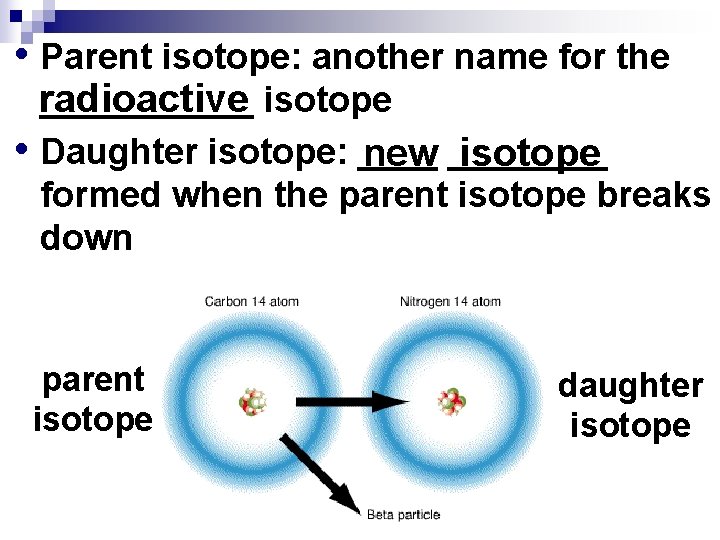
• Parent isotope: another name for the ______ isotope radioactive • Daughter isotope: ____ isotope new ____ formed when the parent isotope breaks down parent isotope daughter isotope
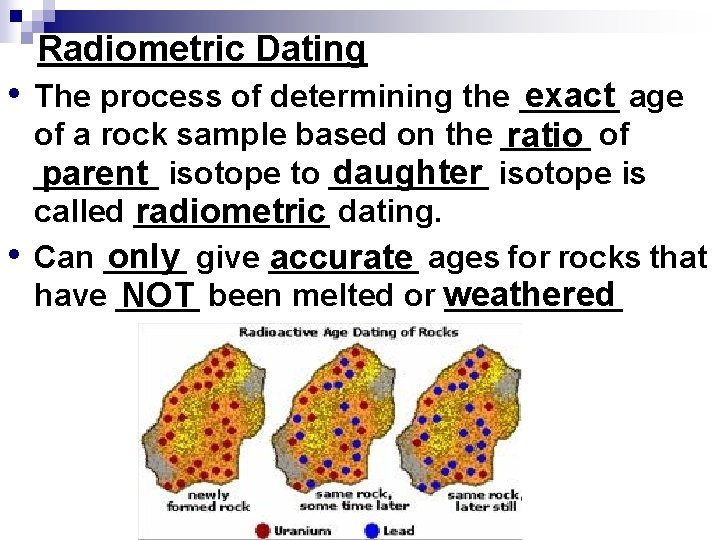
Radiometric Dating • • The process of determining the ______ exact age of a rock sample based on the _____ ratio of _______ daughter isotope is parent isotope to _____ called ______ radiometric dating. Can _____ only give _____ accurate ages for rocks that have __________ NOT been melted or weathered

• To _____ age of a _____ calculate the ____ rock you need to know: 1. The ____ amount of _______ parent isotope in the rock daughter isotope amount of _____ 2. The ____ in the rock 3. The ____ half-life of the _______ parent isotope
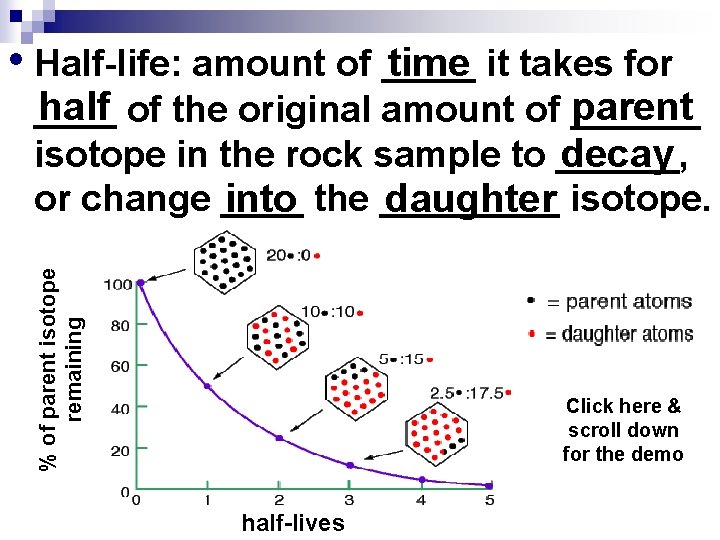
• Half-life: amount of _____ time it takes for % of parent isotope remaining half of the original amount of ______ parent ____ isotope in the rock sample to ______, decay or change ____ into the _____ daughter isotope. Click here & scroll down for the demo half-lives

When the rock formed it had 12 mg of parent isotope (100%). • Ex: There are 12 ___mg of parent _______ isotope in the years pass rock to start and the 10, 000 (1 half-life) half of the parent ____-life 10, 000 is _______ years. After Now there is 6 mg (50%) of the 10, 000 years there parent left and 6 mg (50%) of would be ___mg of the daughter isotope have formed. 6 parent _______ isotope ____ in Another 10, 000 years pass the rock left (half of the (2 half-lives) original amount). Now 3 mg (25%) of the parent left & 9 mg (75%) of the daughter formed.

• There are ____ commonly two parent isotopes _____ used in radiometric dating. 1. Potassium-40: Has a half-life of 1. 3 ___ billion years. Decays into the _____ daughter isotope argon-40 _____. Used to date rocks older ______ than about 1____ million years.
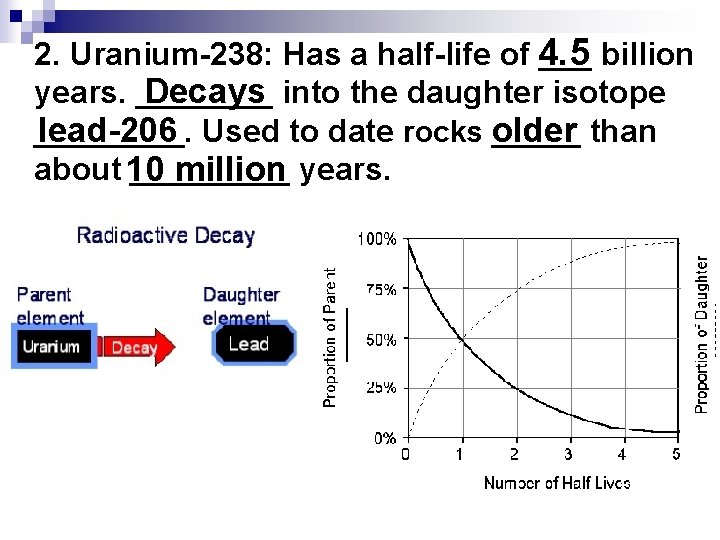
2. Uranium-238: Has a half-life of 4. 5 ___ billion Decays into the daughter isotope years. _________ lead-206. Used to date rocks _____ older than about 10 _____ million years.

Some of the Radioactive Isotopes Used in Radiometric Dating

Radiocarbon Dating • Carbon-14 is used to date fossils, ______ 75, 000 years bones , and wood up to _______ old.

used by • Carbon from the environment is _____ organisms to build tissue in their ______ bodies. _______ • Throughout their lives, the amount carbon of _______ remains constant. _____
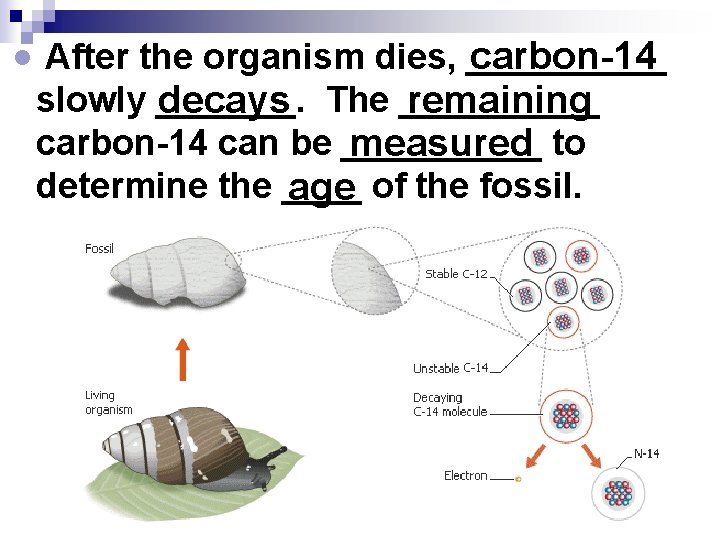
After the organism dies, _____ carbon-14 slowly _______. remaining decays The _____ carbon-14 can be _____ measured to determine the ____ age of the fossil.

How Old is the Earth? age of the earth is _____ estimated by • The ____ meteorites finding the absolute ages of ______ rocks from our solar system and ______ moon. the ______

• Based on this information, the ____ age of the ______ earth is estimated to be around 4. 6 ______ billion years old
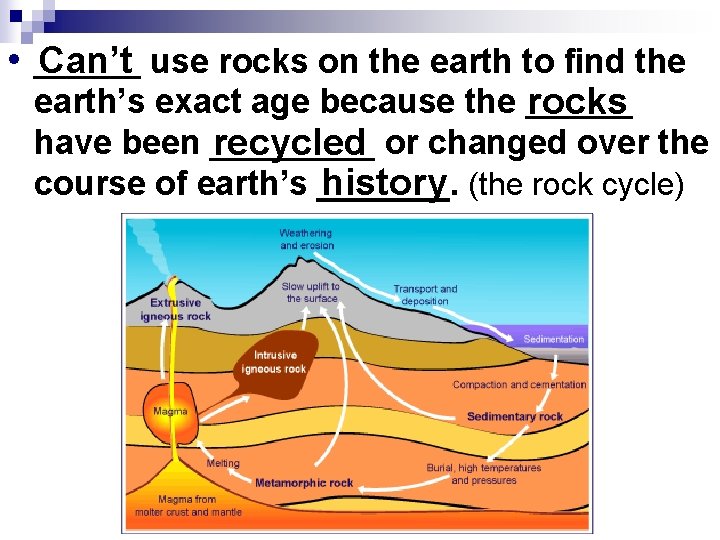
• ______ Can’t use rocks on the earth to find the earth’s exact age because the ______ rocks have been _____ recycled or changed over the history (the rock cycle) course of earth’s _______.

- Slides: 21