Absolute age Mr Scichilone How old is old

Absolute age Mr. Scichilone

How old is old? Scientists use several strategies to determine age of rocks and fossils. 1. Absolute Age: actual age of an object or how long ago an event occurred by using radioactive dating. 2. Radioactive Dating: measures age by comparing the amount of radioactive element isotope present to how much is normally present in that material 3. Relative Age: age of an object compared to another- “I am older than you”

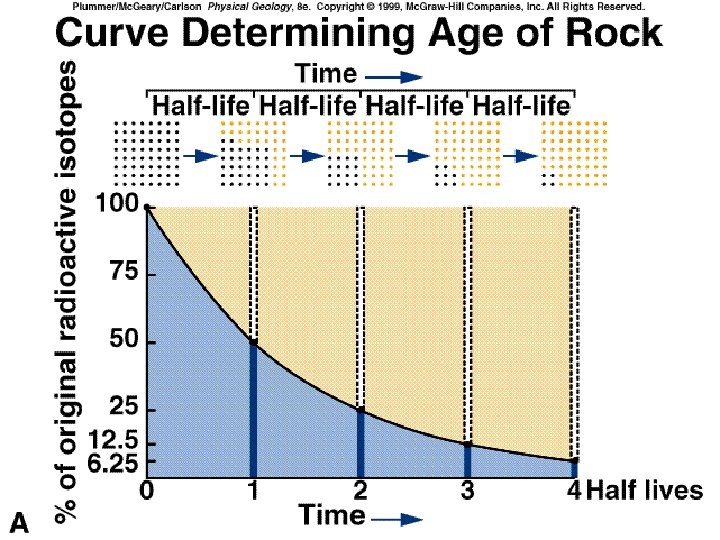
Unit 5 Lesson 2 Relative Dating and Absolute Dating How can the absolute age of rock be determined? • Radioactive isotopes are isotopes that are unstable and break down into other isotopes in a process called radioactive decay. • The radioactive isotope is called the parent isotope, and the stable isotope formed by its breakdown is called the daughter isotope. • Half-life is the time needed for half of a sample of a radioactive element to decay and form daughter isotopes. Copyright © Houghton Mifflin Harcourt Publishing Company

Unit 5 Lesson 2 Relative Dating and Absolute Dating How can the absolute age of rock be determined? • After one half-life has passed, one-half of the parent isotope has changed into daughter isotopes. Copyright © Houghton Mifflin Harcourt Publishing Company

Unit 5 Lesson 2 Relative Dating and Absolute Dating How can the absolute age of rock be determined? • Scientists study the amounts of parent and daughter isotopes to date samples. • Finding the absolute age of a sample by determining the relative percentage of a radioactive parent isotope and a daughter isotope is called radiometric dating. Copyright © Houghton Mifflin Harcourt Publishing Company

Unit 5 Lesson 2 Relative Dating and Absolute Dating What is the best rock for radiometric dating? • Igneous rocks are the best types of rock samples to use for radiometric dating. • When they form, minerals in igneous rocks often contain only a parent isotope and none of the daughter isotope. • This makes the isotope percentage more accurate and easier to interpret. Copyright © Houghton Mifflin Harcourt Publishing Company
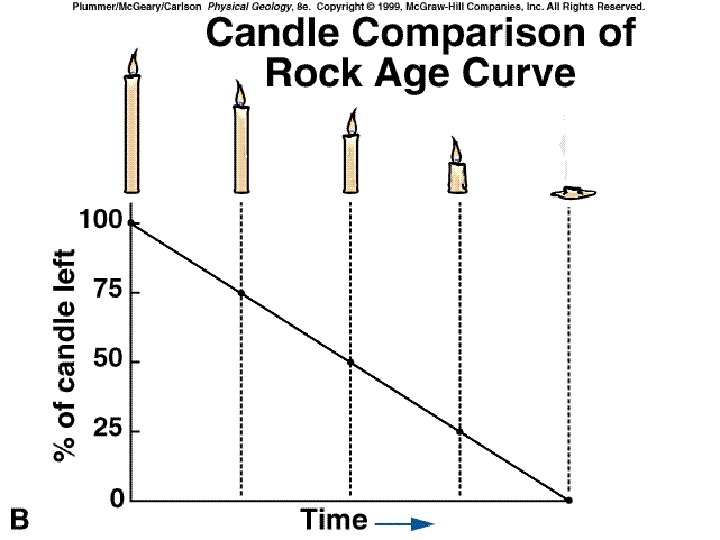
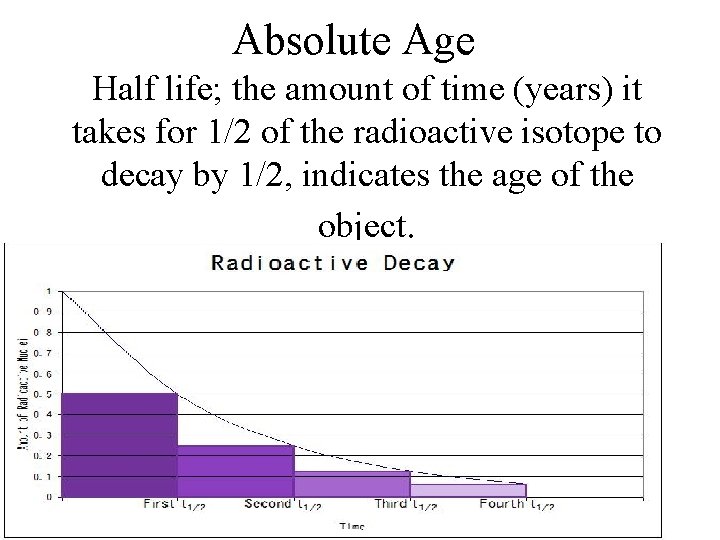
Absolute Age Half life; the amount of time (years) it takes for 1/2 of the radioactive isotope to decay by 1/2, indicates the age of the object. 8
Try This!! If Carbon has a half life of 5730 years; how much will be left after 3 half lives? How many half lives will Carbon have gone through if there is only th 1/16 left? 10
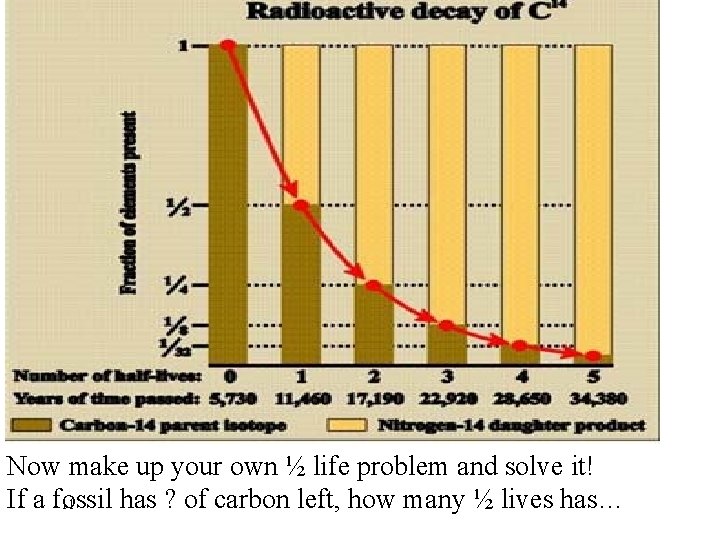
Now make up your own ½ life problem and solve it! If a fossil has ? of carbon left, how many ½ lives has… 11

The grid below represents a quantity of C 14. Each time you click, one half-life goes by. Try it! C 14 – blue N 14 - red Half lives 0 C-14% N-14% Age in Years 100% 0% 0 yrs
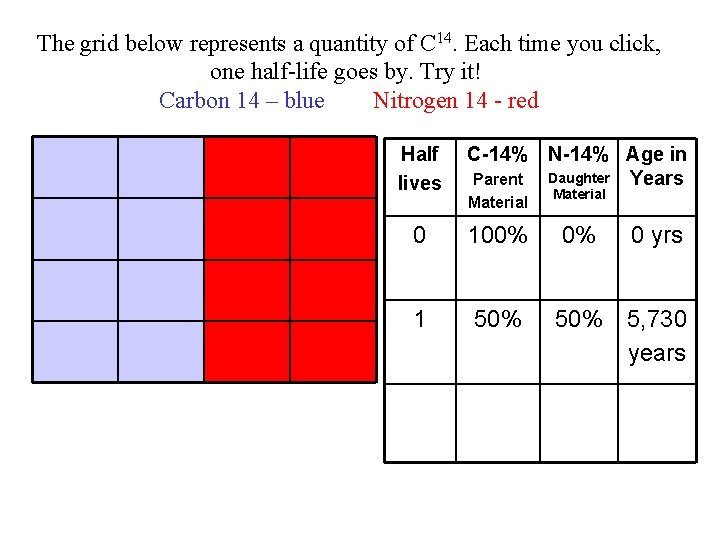
The grid below represents a quantity of C 14. Each time you click, one half-life goes by. Try it! Carbon 14 – blue Nitrogen 14 - red Half lives C-14% N-14% Age in Daughter Years Parent Material 0 100% 0% 1 50% 5, 730 years 0 yrs
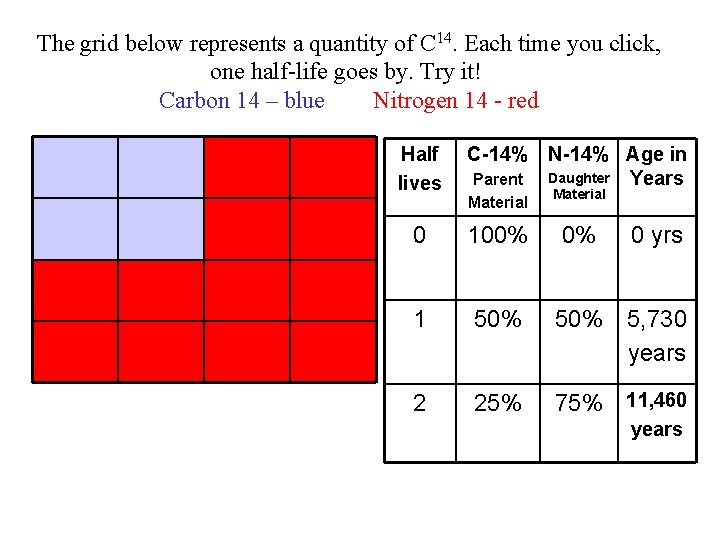
The grid below represents a quantity of C 14. Each time you click, one half-life goes by. Try it! Carbon 14 – blue Nitrogen 14 - red Half lives C-14% N-14% Age in Daughter Years Parent Material 0 100% 0% 1 50% 5, 730 years 2 25% 75% 11, 460 0 yrs years
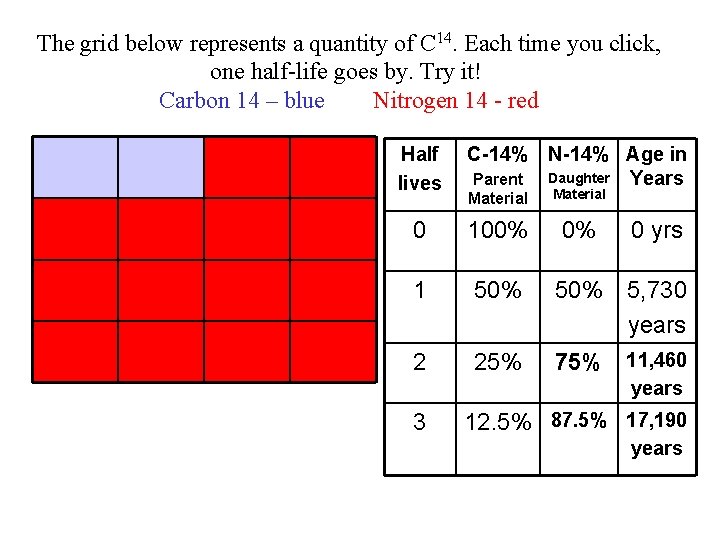
The grid below represents a quantity of C 14. Each time you click, one half-life goes by. Try it! Carbon 14 – blue Nitrogen 14 - red Half lives C-14% N-14% Age in Daughter Years Parent Material 0 100% 0% 1 50% 5, 730 years 2 25% 75% 11, 460 0 yrs years 3 12. 5% 87. 5% 17, 190 years

Half Life Lab 1. Place 100 atoms (pennies) into the plastic box heads up. 2. Shake for 10 seconds 3. Spill pennies on tray and remove the decayed atoms (represented by tails) 4. Count the number of un-decayed atoms (represented by any appearance heads) left on the tray 5. Enter the number of decayed and un-decayed atoms into the table. 6. Put the un-decayed pennies back in the box. 7. Repeat steps 2 -5 until all the atoms have decayed 8. Create a line graph to represent your data in Table (# of undecayed atoms vs. time)

1. What does it mean when we say an atom has “decayed”? 2. At the end of each half-life, at approximately what percentage are the atoms “decaying”? 3. Does the amount of time you shake the box affect the outcome? 4. Do the number of atoms you start with affect the outcome? Explain. 5. How do scientists use radioactive decay to date fossils and artifacts? 17
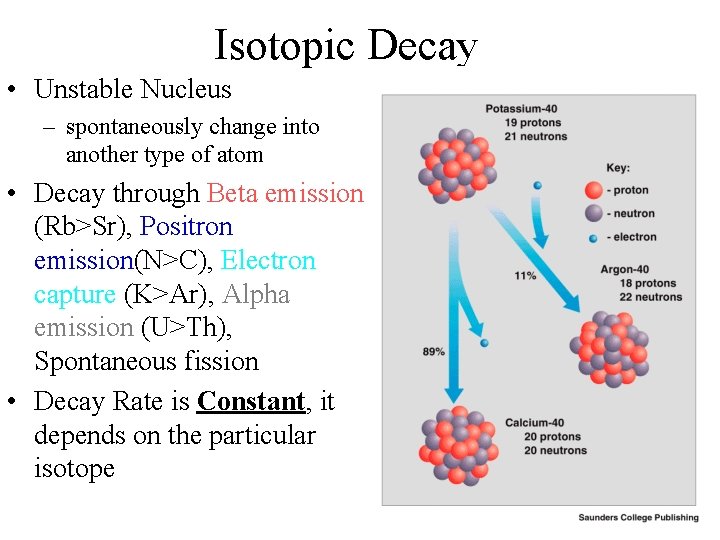
Isotopic Decay • Unstable Nucleus – spontaneously change into another type of atom • Decay through Beta emission (Rb>Sr), Positron emission(N>C), Electron capture (K>Ar), Alpha emission (U>Th), Spontaneous fission • Decay Rate is Constant, it depends on the particular isotope

Radioactive Decay Series

Isotopic Dating Methods • Rubidium-Strontium – Whole Rock, Feldspar, Mica, Clays >100 my – Half-Life 48. 8 by • Potassium-Argon – Volcanics, Feldspar, Mica, Clays >100, 000 my – Half-Life 1. 25 by • Uranium-Thorium-Lead [238 U>>206 Pb: 235 U>>207 Pb] – Whole Rock, Zircon – Half-Life 4. 47 by & 0. 704 by • Samarium-Neodymium – Whole Rock, Silicate Minerals – Half-Life 106 by • Fission Track-
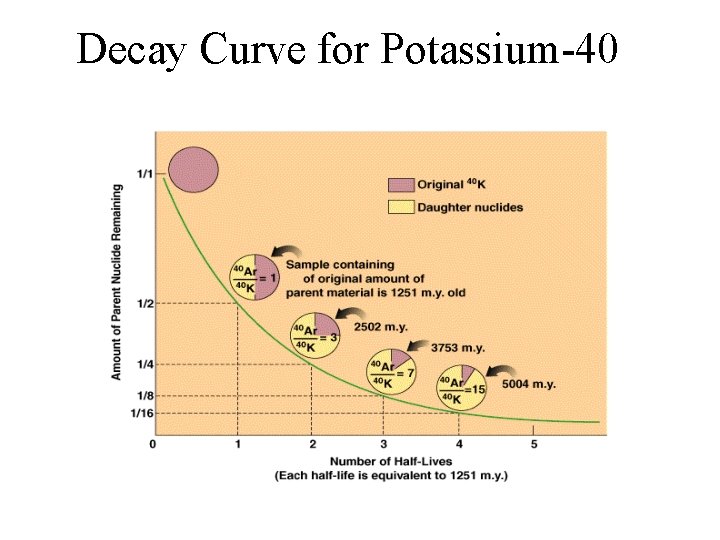
Decay Curve for Potassium-40

Isotopic Dating Methods • Fission Track– Discovered in 1960’s – Small tunnels seen with a Scanning Electron Microscope (SEM) – Due to spontaneous fission of Uranium isotopes – Used to determine the number of years elapsed since uranium bearing mineral formed – Counts of the tracks produced, the original number of Uranium atoms and the decay rate of Uranium – Used to date rocks a few 100 years to billions of years old – Used preferentially for time periods between 50, 000 and 1, 000 years

Fission Tracks

Other Isotopic Dating Methods • Carbon-14 – Ocean Mixing, Dating Organic Remains – Half-life 5730 years • Tritium – Ground and Surface Water Mixing, Glacial Growth – Half-Life 12. 3 years • Silicon-32 – Ocean Mixing – Half-Life 280 years • Beryllium-10 – Sedimentation Rates, Subduction of Sediments – Half-Life 1. 5 x 106 years • Chlorine-36 – Young Volcanics, glaciation, erosion surfaces – Half-Life 3. 1 x 105 years

Non Isotopic Dating Techniques • Thermoluminescence- emission of light when a substance is heated – Time a sample was buried Archeological – Ceramics, Glass: time of last heating – <100 years • Racemization- change in amino acids from living to dead organisms – Bones, Teeth: burial of organic matter – <106 years • Hydration Rinds- 50 um thick layers produced when volcanic glass is exposed to the atmosphere – Need constant climate – Obsidian: artifact manufacture – <105 years

Absolute Age • Uses of Isotopic Dating – Igneous Rocks • Eruption or Intrusion of Magma – Metamorphic Events • reset clock gives time of metamorphism – Organic remains- fossils (70, 000 years) • Reliability of Isotopic Dating – Accuracy (true age) and precision (reproducibility of measurements) of analytical instrumentation

Combining Relative and Absolute Ages • Using various ‘relative age’ principles, as well as • Isotopic dating – – – Usually of igneous rocks Metamorphic rocks give age of metamorphism (reset the ‘clock’) (younger age) Sedimentary rocks give the age of the formation of minerals within the rock (older age)

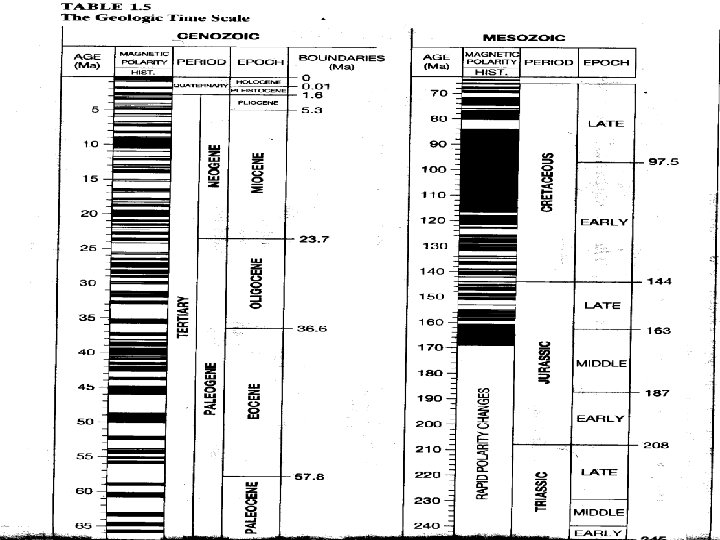
The Geomagnetic Time Scale • Times of normal (North magnetic pole) vs. reverse polarity (South magnetic pole) • Last time polarity flipped was 20, 000 y. b. p. • Polarity Intervals- segments of time in which the magnetic field is reversed or normal • Magnetic data from volcanic rocks and deep sea sediments

Rhythmic Bands • Deposited in response to changes in environmental factors – seasons, daily fluctuations in sunlight, tides • Types – Varves • glacial lakes, winter and summer, clay and silt • tidal couplets • dated back to 20, 000 years in the Baltic; Recognized in older rocks of 3. 5 by, tidal effects recognized in rocks 2. 5 by – Tree Rings- dendrochronology • summer and spring; small cells vs. large cells • extend 8, 000 years in some localities – Corals and Bivalve Shells • Seasonal changes, daily changes in light – Ice Layers • 3000 m long cores date back 65, 000 years, atmospheric and climate studies
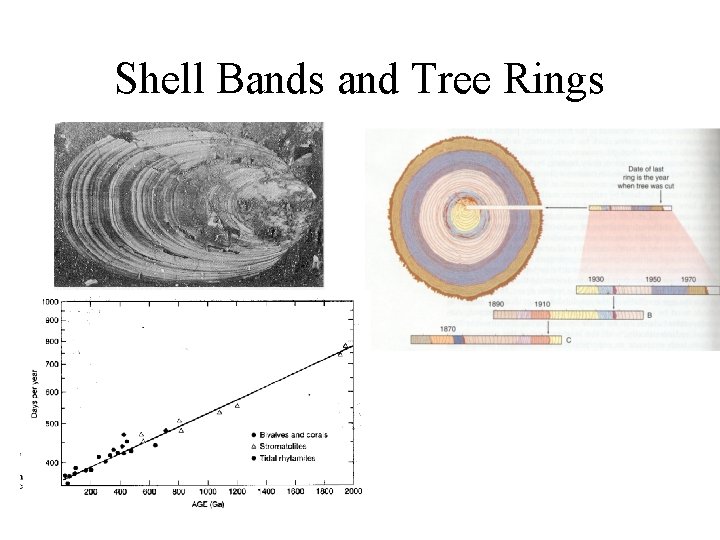
Shell Bands and Tree Rings
- Slides: 32