ABG 101 Question 1 A 44 year old

ABG 101

Question 1 • A 44 year old moderately dehydrated man was admitted with a 2/7 history of acute severe diarrhea. Electrolyte results: Na+ 134, K+ 2. 9, Cl- 108, HCO 3 - 16, Urea 25, Cr 150 , Alb 40 ABG: p. H 7. 31 p. CO 2 33 mm. Hg HCO 3 16 p. O 2 93 mm. Hg • What is the acid base disorder? 1. NAGMA with adequate compensation 2. HAGMA with adequate compensation 3. NAGMA with inadequate compensation 4. HAGMA with inadequate compensation 5. Respiratory Acidosis

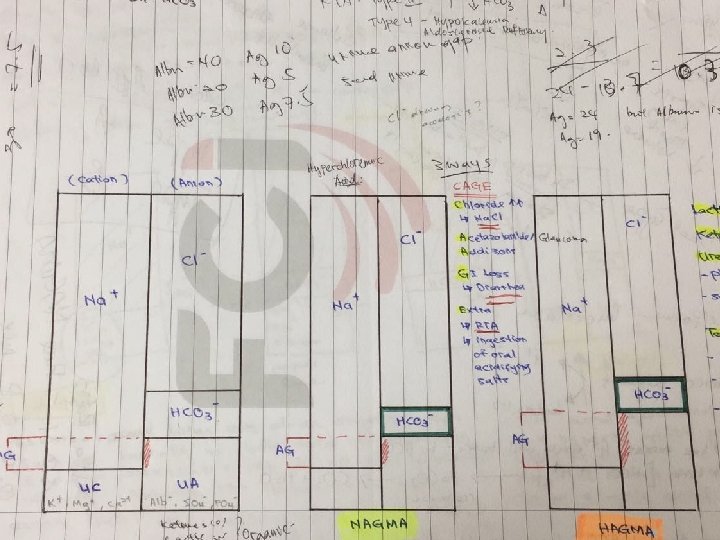
Approach 1. History / Physical examination 2. Look at the p. H 3. Look at the PCO 2 , HCO 3 - & determine whether primary process is : a) Metabolic (go to steps 4, 5, 6)or b) Respiratory ( go to step 7) 4. If Metabolic -> Determine Anion gap and compensation 5. If HAGMA , check delta ratio 6. If NAGMA , and cause is unclear, check urine anion gap 7. If Respiratory -> acute or chronic

History & Physical examination Demographic and social history PMHx History of presenting illness Drug history Examination : Fluid status , GCS , Systemic examination • Initial tests like ECG, CXR, UC 9 • • •

Look at the p. H • Is there an acid base disorder present? - If p. H < 7. 35, then acidemia - If p. H > 7. 45, then alkalemia - If p. H within normal range, then acid base disorder may not be present - p. H may be normal in the presence of a mixed acid base disorder (e. g Chronic type 2 respiratory failure with high PCO 2 and metabolic compensation with high HCO 3 -)

Definitions and Normal Values • • Normal acid base values p. H PCO 2 HCO 3 Range: 7. 35 - 7. 45 36 -44 19 -29 Optimal value 7. 40 40 24 Acid base status is defined in terms of the plasma p. H. Acidemia - Decrease in the blood p. H below normal range of 7. 35 Alkalemia - Elevation in blood p. H above the normal range of 7. 45 • Acidosis – process that increases [H+] by increasing PCO 2 or by reducing [HCO 3 -] Alkalosis – process that reduces [H+] by reducing PCO 2 or by increasing [HCO 3 -] • Note* Although acidosis and alkalosis usually leads to acidemia and alkalemia respectively, the exception occurs when there is a mixed acid base disorder. In that situation, multiple acid base processes coexisting may lead to a normal p. H or a mixed picture.
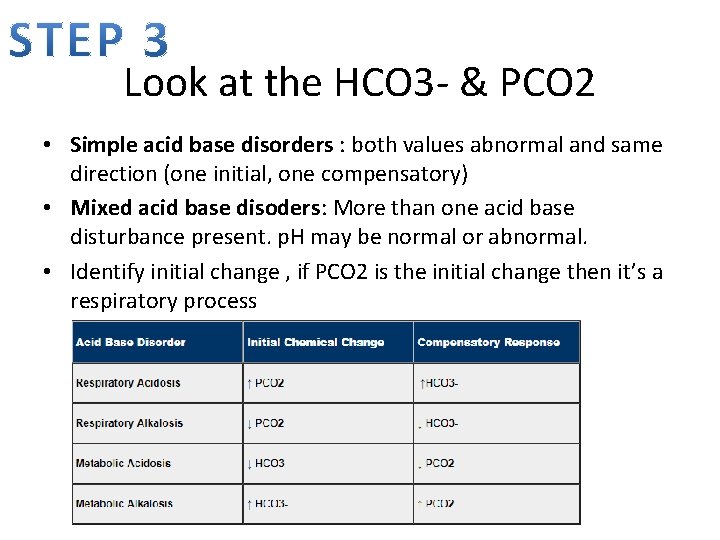
Look at the HCO 3 - & PCO 2 • Simple acid base disorders : both values abnormal and same direction (one initial, one compensatory) • Mixed acid base disoders: More than one acid base disturbance present. p. H may be normal or abnormal. • Identify initial change , if PCO 2 is the initial change then it’s a respiratory process

Why is acidosis so bad?

Alkalosis isn’t an angel either

Question 2 • 29 year old with PMHx of Cerebral Palsy p/w fever , URTI symptoms and a rash 2/7. Renal panel shows : Na 133 , k 4 , HCO 3 12 , Albumin 32 , cl 107. (Assume p. H is low and p. CO 2 is adequately compensating)What’s the corrected anion gap ? 1. 18 2. 10 3. 14 4. 16

AG and how to correct • AG = Na – (HCO 3 + Cl) – Unmeasured Anions (Albumin, Phosphates, Sulfates, Organic acids) > Cations (K+, Ca 2+, Mg 2+) – Normal 12 (8 -16) • Albumin decreases AG by 2. 5 -3 for every 10 g/L albumin. Corrected AG = AG + 0. 25(40 – serum alb) • This patient’s corrected AG is 16. So is it a HAGMA?

Delta ratio = � AG / �Bicarb

Delta gap • • ∆ gap = ∆ AG - ∆ HCO 3 ∆ gap = (AG -12) minus (24 – HCO 3) ∆ gap = AG + HCO 3 -36 (normal 0 +/-6) *In a pure HAGMA, ∆ gap should remain within -6 to +6 because the AG and HCO 3 move by equal amounts in opposite directions – If > +6, there is an additional metabolic alkalosis – If < -6, there is an additional hyperchloremic acidosis/NAGMA
If HAGMA , check Delta Ratio

Calculation • Anion Gap = Na – (cl+HCO 3) • Corrected AG • Delta (� ) AG • 133 – (107+12) = 14 • 14 + 0. 25(40 -32) = 16 • 16 -12 = 4 • �Bicarb • 24 - 12 = 12 Corrected AG = AG + 0. 25(40 – serum alb) � AG = Corrected AG – 12 �Bicarb = 24 – serum HCO 3 This patient’s delta ratio is 0. 33

Question 3 • Which of the following drugs can cause NAGMA? 1. Furosemide 2. Spironolactone 3. Acetazolamide 4. 1 & 2 5. 2 & 3 6. 1 & 3

This patient • This patient recently had an eye op and was started on acetazolamide by SNEC. After running it by them, we stopped it and her bicarb normalised after 5 days

Causes of NAGMA • Renal loss of HCO 3 – – Renal Tubular Acidosis – proximal/distal Tubular Injury (ATN) Hypo-aldosterone states: inadequate NH 3 production Drugs • GI loss of HCO 3 – Diarrhea, NG aspirates , Laxative abuse, ileostomy etc • Too much cl- (cations) – TPN, high chloride content solutions (normal saline!)

If NAGMA & unclear cause ->Urine AG • Urine Anion Gap • Distinguishing between 3 groups of causes of NAGMA is usually clinically obvious Urine AG is useful when its not Urine anion gap = (Normally 0 +/- 20) [Na+] + [K+] - [Cl-] • If the acidosis is due to loss of base via the bowel then the kidneys can response appropriately by increasing ammonium (which is excreted along with cl-) excretion to cause a net loss of H+ from the body • In a patient with a hyperchloremic metabolic acidosis: A negative UAG suggests GI loss of bicarbonate (eg diarrhea) A positive UAG suggests impaired renal acidification (ie renal tubular acidosis).

Causes of HAGMA – organic acidosis • • - Lactic Acidosis Ketoacidosis Diabetes Alcohol-induced Starvation Renal Failure Toxin Ingestion Ethylene Glycol Methanol Salicylates M-Methanol U-Uremia (chronic kidney failure) D-Diabetic ketoacidosis P-Propylene glycol I-Infection, Iron, Isoniazid, Inborn errors of metabolism L-Lactic acidosis E-Ethylene glycol /Ethanol S-Salicylates Corrected AG >12

Metabolic acidosis causes summary

Lactic Acidosis • Lactate is a byproduct of anaerobic glycolysis, cleared by liver & kidney; normal value 0. 5 – 1 m. Eq/L • Type A: Tissue Ischemia/Decreased perfusion or oxygenation – Shock, cardiac arrest, low CO, severe anemia, seizures • Type B: Altered Cell Metabolism (not related to hypoperfusion) – Mitochondrial dysfunction, increased pyruvate production, inhibition of cytochrome oxidase - B 1 - Impaired hepatic metabolism/ renal clearance - B 2 - Drugs (Metformin, Linezolid, NRTIs, Lorazepam, Propofol, HAART) - B 3 - Inborn errors of metabolism **Interesting read – D-lactic acidosis

Causes of metabolic alkalosis

Metabolic Alkalosis • Cause usually obvious from history; Reduced ECF vs Not reduced ; 3 common causes: 1. Excess mineralocorticoid 2. Vomiting 3. Diuretics • Urinary cl-

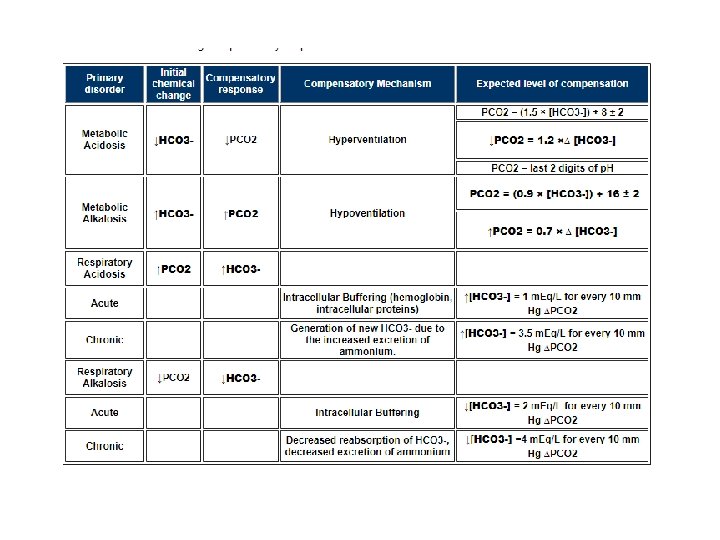
If Metabolic , check compensation • • Compensatory Responses: Metabolic Acidosis The body responds to metabolic acidosis by trying to restore the PCO 2 / [HCO 3 -] ratio. This is done by reducing the PCO 2. • The reduction in PCO 2 is accomplished by increasing alveolar ventilation. The drop in arterial p. H stimulates both the central and peripheral chemoreceptors controlling respiration, resulting in an increase in alveolar ventilation. -> Kussmaul respiration • Winter's Formula: To estimate the expected PCO 2 range based on respiratory compensation PCO 2 = (1. 5 × [HCO 3 -]) + 8 ± 2 • In general, respiratory compensation results in a 1. 2 mm. Hg reduction in PCO 2 for every 1. 0 meq/L reduction in the plasma HCO 3 - concentration down to a minimum PCO 2 of 10 to 15 mm. Hg.

Compensation • Compensatory Responses: Metabolic Alkalosis • Increase in bicarb due to addition of alkali which kidneys cannot excrete. Usually associated with renal impairment because the kidney has good alkali clearing • Body responds to increase in HCO 3 - by increasing PCO 2 • Reduction of ventilation • On average the p. CO 2 rises 0. 7 mm. Hg for every 1. 0 meq/L increment in the plasma [HCO 3 -]. • Formula : PCO 2 = (0. 9 × [HCO 3 -]) + 16 ± 2

Question 4 • A 22 year old female with type I DM, presents to the emergency department with a 1/7 history of nausea, vomiting, polyuria, polydypsia and vague abdominal pain. O/e Deep sighing breathing, orthostatic hypotension, and dry mucous membranes. • Labs: Na 132 , K 6. 0, Cl 93, HCO 3 - 11 glucose 720, BUN 38, Cr 2. 6. UA: p. H 5, SG 1. 010, ketones negative, glucose positive. Plasma ketones trace. ABG: p. H 7. 27 HCO 3 - 10 PCO 2 23 • What is the acid base disorder? 1. NAGMA with adequate compensation 2. NAGMA with inadequate compensation 3. HAGMA with adequate compensation 4. HAGMA with inadequate compensation 5. Respiratory alkalosis

Breathe….

Question 5 • A 70 year old man with history of CHF presents with increased shortness of breath and leg swelling. ABG: p. H 7. 24, PCO 2 60 mm. Hg, PO 2 52 HCO 3 - 27 • What is the acid base disorder? 1. Metabolic acidosis 2. Acute Respiratory acidosis with metabolic alkalosis 3. Acute Respiratory acidosis 4. Chronic Respiratory acidosis 5. Not sure


Causes of respiratory alkalosis

Acute or Chronic Respiratory Acidosis? • An acute respiratory process will produce a compensatory response that is due primarily to rapid intracellular buffering. • A chronic respiratory process will produce a more significant compensatory response that is due primarily to renal adaptation, which takes a longer time to develop. - To assess if acute or chronic, determine the extent of compensation.

Compensation • Respiratory acidosis • In Respiratory Acidosis, the elevation in PCO 2 result from a reduction in alveolar ventilation. Elevation in PCO 2 is never due to an increase in CO 2 production. • CO 2 goes up , body responds by increasing HCO 32 mechanisms a) rapid cell buffering (acute, within minutes) b) increase in net acid excretion (chronic) • Because these mechanisms occur at different moments in time, acute respiratory acidosis can be distinguished from chronic respiratory acidosis.

Compensation • Acute respiratory acidosis: Increase CO 2 -> increased Carbonic acid (H 2 CO 3) , not bufferable by HCO 3 Carbonic acid (H 2 CO 3 ) can only be buffered by the limited intracellular buffers (primarily hemoglobin and proteins). H 2 CO 3 + Hb- → HHb + HCO 3 Resultant increase in the plasma [HCO 3 -], averaging 1 meq/L for every 10 mm. Hg rise in the PCO 2.

Compensation • Chronic Respiratory Acidosis Persistent elevation of PCO 2 -> Stimulates excretion of titratable acid and ammonium -> causing increased HCO 3 - to ECF Process takes a few days 3. 5 meq/L increase in the plasma HCO 3 - concentration for every 10 mm. Hg increase in the PCO 2. Renal compensation offers more significant p. H protection in the setting of chronic respiratory acidosis in contrast to intracellular buffering in the acute situation. Chronic respiratory acidosis is commonly caused by COPD. These patients can tolerate a PCO 2 of up to 90 -110 mm. Hg and not have a severe reduction in p. H due to renal compensation. • Mechanisms are similar in acute and chronic respiratory alkalosis
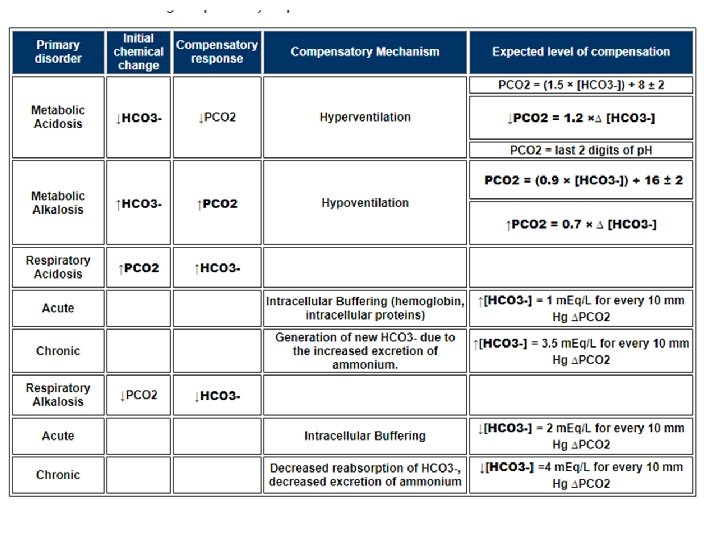

Summary of Compensatory mechanisms 1. 2. 3. 4. 5. 6. Compensatory responses never return the ph to normal or overshoot. The basis of compensatory responses is to maintain the PCO 2/[HCO 3 -] ratio. Therefore, the direction of the compensatory response is always the same as that of the initial change. Compensatory response to respiratory disorders is two-fold; a fast response due to cell buffering and a significantly slower response due to renal adaptation. Compensatory response to metabolic disorders involves only an alteration in alveolar ventilation. Metabolic responses cannot be defined as acute or chronic in terms of respiratory compensation because the extent of compensation is the same in each case.

Question 6 • A 50 year old insulin dependent diabetic woman was brought to the ED by ambulance. She was semi-comatose and had been ill for several days. Current medication was digoxin and a thiazide diuretic for CHF. Lab results Serum chemistry: Na 132, K 2. 7, Cl 79, HCO 3 - 19 Glu 815, Lactate 0. 9 urine ketones 3+ ABG: p. H 7. 41 PCO 2 32 HCO 3 - 19 p. O 2 82 • What is the acid base disorder? 1. HAGMA + Metabolic Alkalosis 2. HAGMA 3. NAGMA 4. Acute Respiratory alkalosis 5. NAGMA + Metabolic Alkalosis 6. No acid base disturbance

Graduating Question • 70 year old man admitted with severe pneumonia. Unwell for a week. Vomiting x 5/7. • No PMHx , Not on any medications. • O/e Hyperventilating and in distress. On Oxygen via facemask. BP 85/55

• p. H: p. H is greater than 7. 44 so an alkalaemia is present. The cause is an alkalosis • Pattern: p. CO 2 & bicarbonate are both low suggesting either a metabolic acidosis or a respiratory alkalosis. As we already know an alkalosis is present then the primary disorder is a respiratory alkalosis. • Compensation: Asessing the compensation for respiratory alkalosis. The expected HCO 3 is (24 - 8) = 16. The actual HCO 3 is higher (19) which indicates the presence of a metabolic alkalosis. • Anion Gap: Very high so there must be a high-anion gap metabolic acidosis present as well. Delta ratio 3. 2 • Impression: Respiratory alkalosis + HAGMA + metabolic alkalosis!

Thank you
- Slides: 43