ABCD Quality Risk Management 20 th March 2009

ABCD Quality Risk Management 20 th March 2009 - SAPRAA Helena van Schalkwyk Quality Assurance Manager Boehringer Ingelheim, South Africa QRM - SAPRAA 20 March 2009 PTS Dr. R. Beerbohm

Agenda q Risk Management - The Essentials q Background of QRM q Structured Risk Management Process: ICH Q 9 q Practical Implementation of QRM in your company q Summary and conclusions QRM - SAPRAA 20 March 2009 2

Agenda q Risk Management - The Essentials q Background of QRM q Structured Risk Management Process: ICH Q 9 q Practical Implementation of QRM in your company q Summary and conclusions QRM - SAPRAA 20 March 2009 3

Risk Management – The Essentials • Keep Risk Management SIMPLE - Beware of the following pitfalls …… v Recruiting “Risk Management Experts” v Writing a 30 page “Risk Management Policy/SOP”” v Seeing Risk Management as something NEW v The appointment of Risk Management CONSULTANTS QRM - SAPRAA 20 March 2009 4

Risk Management – The Essentials (cont) • We are already managing risks …… v. Because we live in a world full of risks! v“Zero Risk” does not exist anywhere! • Risk Management involves: v Identifying risks (what can go wrong/ what are the hazards) v Analysing risks (estimate risks associated with identified hazards) v Evaluating risks (are these risk really that bad? ) v Controlling and reducing these risks (mitigate severity, reduce probability of harm, increase likelihood of detection) v Accepting residual risk (accept level of risk) QRM - SAPRAA 20 March 2009 5

Risk Management – The Essentials (cont) • ICH Q 9 Definitions v “Harm” – damage to health, including the damage that can occur from loss of product, or availability quality v“Hazard” – potential source of harm v “ Risk” – combination of probability of occurrence of harm QRM - SAPRAA 20 March 2009 and severity 6

Risk Management – The Essentials (cont) • What is risk? v You only have risk where a hazard exists v Risk is the combination of probability of harm severity of that harm v Some risks we can’t do anything about v Other risks can be managed v DRIVING to work is a risky business QRM - SAPRAA 20 March 2009 and 7

Risk Management – The Essentials (cont) • What is your perception of risk? v High, medium or low based on v Probability of occurrence v Severity of potential harm Will you be satisfied with this level of risk or not? QRM - SAPRAA 20 March 2009 8

Risk Management – The Essentials (cont) QRM - SAPRAA 20 March 2009 9

Risk Management – The Essentials (cont) QRM - SAPRAA 20 March 2009 10

Risk Management – The Essentials (cont) v. Are you prepared to take your chances and accept the risk? or v Accept that the risk is unacceptable and in need of management and control? QRM - SAPRAA 20 March 2009 11
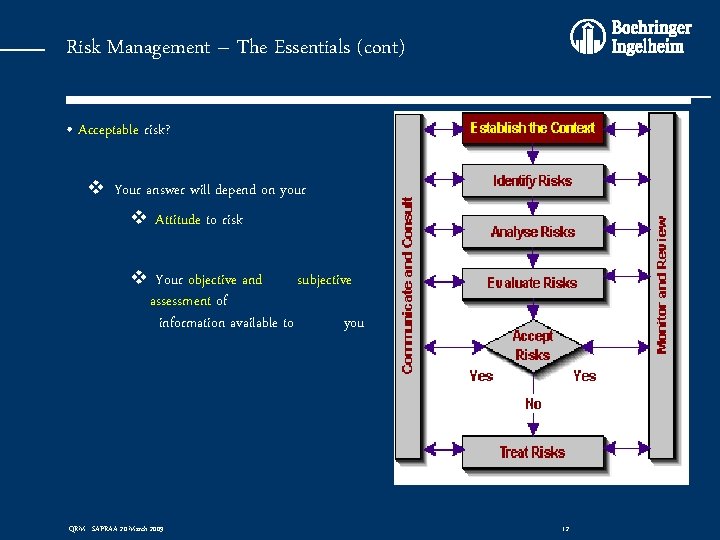
Risk Management – The Essentials (cont) • Acceptable risk? v Your answer will depend on your v Attitude to risk v Your objective and subjective assessment of information available to you QRM - SAPRAA 20 March 2009 12

Risk Management – The Essentials (cont) • The risks associated with DRIVING in South Africa: v Approximately 14, 627 people are killed each year on roads South African (Road Traffic Management Corporation - Road Traffic Report March 2008 dated 17 April 2008) v That’s 40 people every day v Roads will become even more congested v Cars are becoming both faster and safer Is the risk acceptable to you or not? QRM - SAPRAA 20 March 2009 13

Risk Management – The Essentials (cont) • Risk IDENTIFICATION What can go wrong? v You may be involved in a fatal road accident – (killing yourself or someone else) v You may be seriously injured and unable to work for a period of time v You may be sent to prison if found guilty of dangerous driving v Incur costs for repairs to damaged vehicles and the inconvenience factor v You driver’s license may be suspended due to (multiple) speeding offences QRM - SAPRAA 20 March 2009 14

Risk Management – The Essentials (cont) • Risk IDENTIFICATION (cont) What can go wrong? v The inconvenience of relying on public transport limited availability in SA!) v You may lose your job if you have lost your driver’s license v Social hardships as a result of no income v Higher insurance premiums after the accident claim v High fuel costs resulting from uneconomical driving style QRM - SAPRAA 20 March 2009 (currently very 15

Risk Management – The Essentials (cont) • DRIVING: Risk ANALYSIS v What is the probability of occurrence? v What are the potential consequences (severity)? v Is this level of risk acceptable … or not? v How can I control (manage) the risks? QRM - SAPRAA 20 March 2009 16
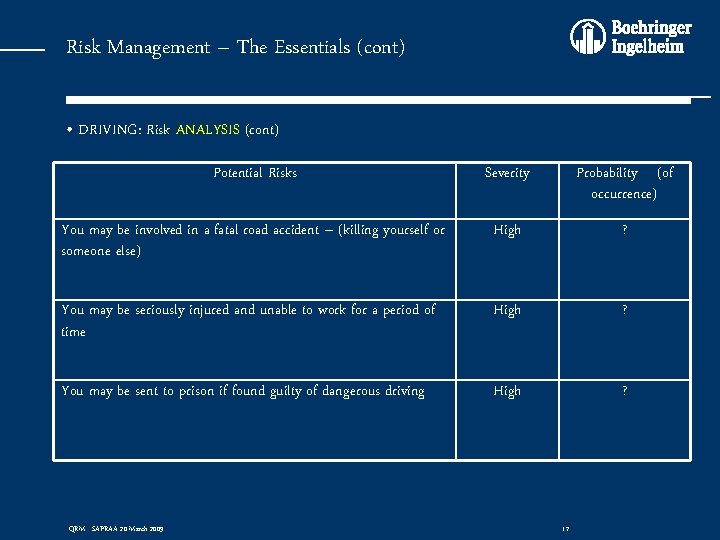
Risk Management – The Essentials (cont) • DRIVING: Risk ANALYSIS (cont) Potential Risks Severity Probability (of occurrence) You may be involved in a fatal road accident – (killing yourself or someone else) High ? You may be seriously injured and unable to work for a period of time High ? You may be sent to prison if found guilty of dangerous driving High ? QRM - SAPRAA 20 March 2009 17
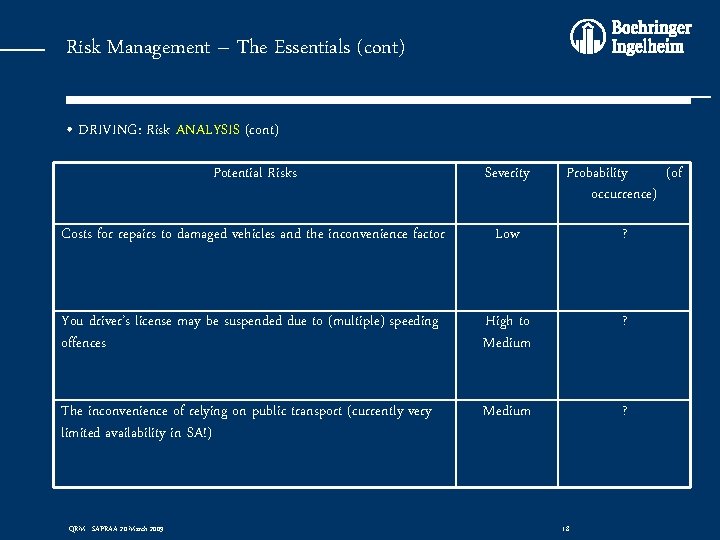
Risk Management – The Essentials (cont) • DRIVING: Risk ANALYSIS (cont) Potential Risks Severity Probability (of occurrence) Costs for repairs to damaged vehicles and the inconvenience factor Low ? You driver’s license may be suspended due to (multiple) speeding offences High to Medium ? The inconvenience of relying on public transport (currently very limited availability in SA!) Medium ? QRM - SAPRAA 20 March 2009 18
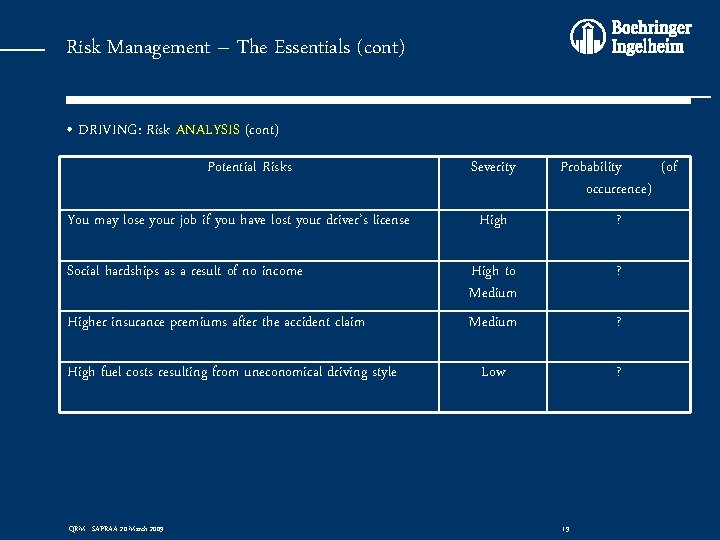
Risk Management – The Essentials (cont) • DRIVING: Risk ANALYSIS (cont) Potential Risks You may lose your job if you have lost your driver’s license Social hardships as a result of no income Higher insurance premiums after the accident claim High fuel costs resulting from uneconomical driving style QRM - SAPRAA 20 March 2009 Severity Probability (of occurrence) High ? High to Medium ? Low ? ? 19

Risk Management – The Essentials (cont) • DRIVING: Risk CONTROL What actions can you take to v Reduce severity? v Reduce probability of occurrence? v Increase ability to detection? You could v Drive slower v Increase following distance between you and the car in front of you v Buy a car with better safety specifications v Sell your high performance car for a slower/more economical one v Drive at a safer and slower speed v Leave earlier to reach your destination in time QRM - SAPRAA 20 March 2009 20

Risk Management – The Essentials (cont) • DRIVING: Risk CONTROL (cont) You could v Purchase a “Radar Detector” Please Note: Do NOT use RM to justify what you know is inherently wrong! purchasing a “Radar Detector” system!) (Like v Attend an advanced driving skills course v Talk to those who have witnessed accidents close by v Travel at alternative times (non-peak hours) of the day v Ensure that your car is always in a roadworthy condition QRM - SAPRAA 20 March 2009 21

Risk Management – The Essentials (cont) • DRIVING: Risk REVIEW Circumstances or situations could change which may then influence original decision your v You may be able to afford a safer car v You may decide to move to a job closer to home that would result in less stressful and dangerous travel v You may witness a serious accident that would influence your driving behaviour/style v New laws may be implemented to lower maximum speed limits, and much tougher enforcement of the traffic laws v You may have children which will motivate you to drive more careful QRM - SAPRAA 20 March 2009 22

Risk Management – The Essentials (cont) • FINALLY…. . remember the following: v Never use Risk Management to justify what you know is INHERENTLY wrong v Invest in an advance driving skills course instead of purchasing a “Radar Detector” QRM - SAPRAA 20 March 2009 23

Agenda q Risk Management – The Essentials q Background of QRM q Structured Risk Management Process: ICH Q 9 q Practical Implementation of QRM in your company q Summary and conclusions QRM - SAPRAA 20 March 2009 24

Background of QRM • FDA’s CONCERNS – 2001: Pharmaceutical Manufacturing v Innovation/continuous improvement difficult due to delays in approval at HA’s v Many product formulations and processes derived empirically v Too little knowledge on mechanisms impacting product quality v Very low efficiency and high cost – the opposite of what is necessary in a competitive global market v Industry hesitant/afraid to use new technologies QRM - SAPRAA 20 March 2009 25

Background of QRM (cont) • FDA’s CONCERNS – 2001 (cont): FDA unable to deliver on their statutory inspection obligations (low budget and high demand) • FDA’s CONCLUSION: v Current approach likely to be inadequate to meet future needs v Need to place emphasis on science and risk-based approaches • FDA’s INITIATIVES since 2002: v Process Analytical Technology (PAT) – not just testing, but a philosophy of Process Understanding v Control in-process rather than end-product testing v Minimises risks of poor quality QRM - SAPRAA 20 March 2009 26

Background of QRM (cont) • FDA’s INITIATIVES since 2002 (cont): v Quality by Design (Qb. D) approach – which parameters are critical to product quality? v Statistically designed experiments v ICH Q 8 QRM - SAPRAA 20 March 2009 27
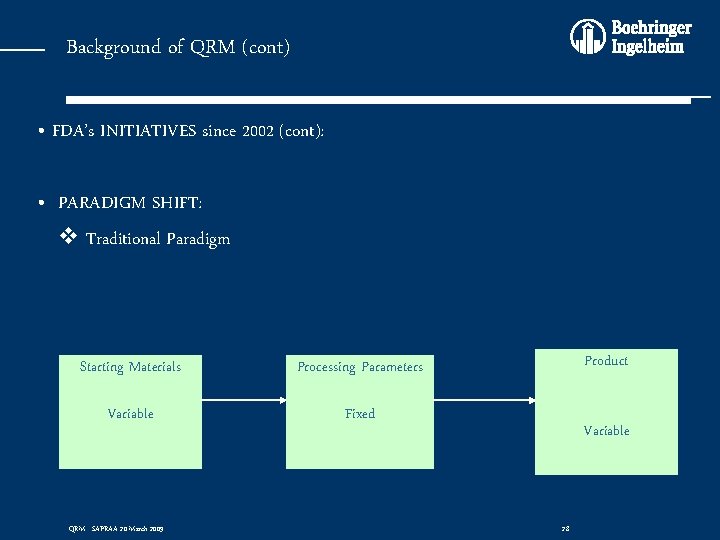
Background of QRM (cont) • FDA’s INITIATIVES since 2002 (cont): • PARADIGM SHIFT: v Traditional Paradigm Starting Materials Processing Parameters Variable Fixed QRM - SAPRAA 20 March 2009 Product Variable 28
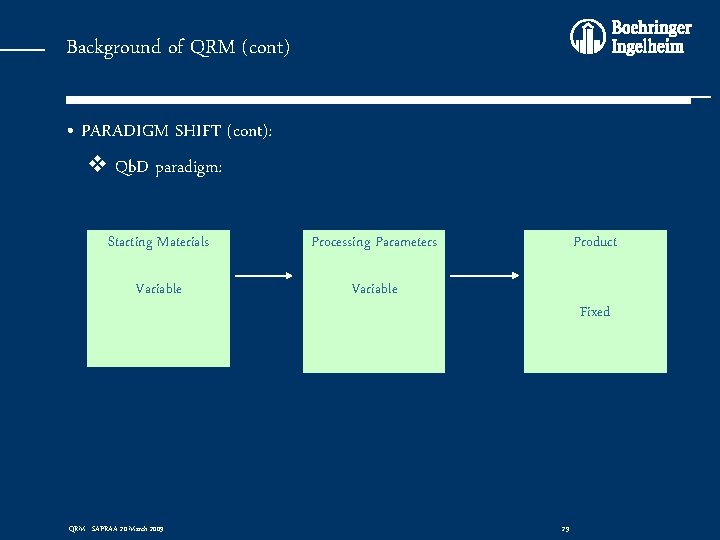
Background of QRM (cont) • PARADIGM SHIFT (cont): v Qb. D paradigm: Starting Materials Processing Parameters Variable QRM - SAPRAA 20 March 2009 Product Fixed 29

Background of QRM (cont) • ICH v EU and Japan joined USA to define a new paradigm at an ICH workshop in Brussels in July 2003 v This workshop agreed a 5 year vision: v Create a single, harmonised global standard and interpretation based on good principles QRM - SAPRAA 20 March 2009 quality science and risk management 30

Background of QRM (cont) • ICH (cont) v Achieve 5 year vision by incremental steps Old: New: QRM - SAPRAA 20 March 2009 Pharmaceutical Development (Q 8) Data transfer / variable output Knowledge transfer / consistent output Quality Risk Management (Q 9) Unstructured approach Structured process Quality Systems (Q 10) Large variability on Q-systems Consistency on Q-systems 31

Background of QRM (cont) • ICH (cont) Flexible Regulatory Approach v Regulators evaluate risk, based on: v Product and Process Design (Q 8) v Measures to evaluate and manage risks (Q 9) v Q-system implementation (Q 10) v Regulators determine risk and modify level of oversight accordingly for: v Submissions v Post-approval review v GMP Inspections v Result v Removal of barriers to continuous improvement v Efficient use of resources by industry and regulator QRM - SAPRAA 20 March 2009 32

Background of QRM (cont) • Design Space Design space is the multi-dimensional combination and interaction of input variables (e. g. material attributes) and process parameters that have been demonstrated to provide assurance of quality Knowledge Space Design Space Batch process/control parameters The batch process/control parameters are NOT registered and, hence, moving them within the Design Space QRM - SAPRAA 20 March 2009 is NOT a change 33

Background of QRM (cont) • ICH Q 10 – Pharmaceutical Quality System v Objective of Q 10 - to establish a new guideline describing model for an effective QMS for the Pharmaceutical Industry, referred to as the Pharmaceutical QMS QRM - SAPRAA 20 March 2009 34

Background of QRM (cont) • Q 10 and Management Responsibility v Management commitment, management review and communication are important v Senior Management v Establish a PQS appropriate for the organisation and compliant with regulations v Ensure that PQS responsibilities and authorities are defined and communicated v Has ultimate responsibility to foster a company-wide commitment to quality and for the successful functioning of the PQS QRM - SAPRAA 20 March 2009 35

Background of QRM (cont) • ICH Q 11 – API Development v Q 11 will aim to define the development process for API’s similar to those contained products (Detail for the 3. 2. S. 2 Section of the CTD) in Q 8 for medicinal • The New Regulatory Paradigm – Conclusion v It makes sense v FDA is driving it forward v If we can make it to work, it will benefit patients, industry and regulators v We should focus on what adds value for patients QRM - SAPRAA 20 March 2009 36

Background of QRM (cont) • QRM – Summary v Realise that v current situation is not sustainable v v By Industry By Regulators v current situation is not in the best interest for patients QRM is a win: win situation for: v v v QRM - SAPRAA 20 March 2009 Industry Regulators and Patients 37

Agenda q Risk Management – The Essentials q Background of QRM q Structured Risk Management Process: ICH Q 9 q Practical Implementation of QRM in your company q Summary and conclusions QRM - SAPRAA 20 March 2009 38

Structured Risk Management Process: ICH Q 9 • Q 9 – Table of Contents 1. Introduction 2. Scope 3. Principles of QRM 4. General QRM Process 5. RM Methodology 6. Integration of QRM process into industry and regulatory operations 7. Glossary 8. References Annex I – RM Methods and Tools Annex II – Potential Appliances of QRM - SAPRAA 20 March 2009 39

Structured Risk Management Process: ICH Q 9 (cont) • Q 9 – Principles of QRM Two primary principles: 1. The evaluation of the quality risk should be based on scientific knowledge and always links back to the protection of the patient. 2. The level of effort, formality and documentation of the QRM process should be corresponding with the level of risk. QRM - SAPRAA 20 March 2009 40

Structured Risk Management Process: ICH Q 9 (cont) • QRM is: A systematic process for the assessment, control, communication and review of risks to the quality of the drug (medicinal) product across the product lifecycle • QRM Process: v Consists of three stages: v Risk Assessment v Risk Control v Risk Review QRM - SAPRAA 20 March 2009 41
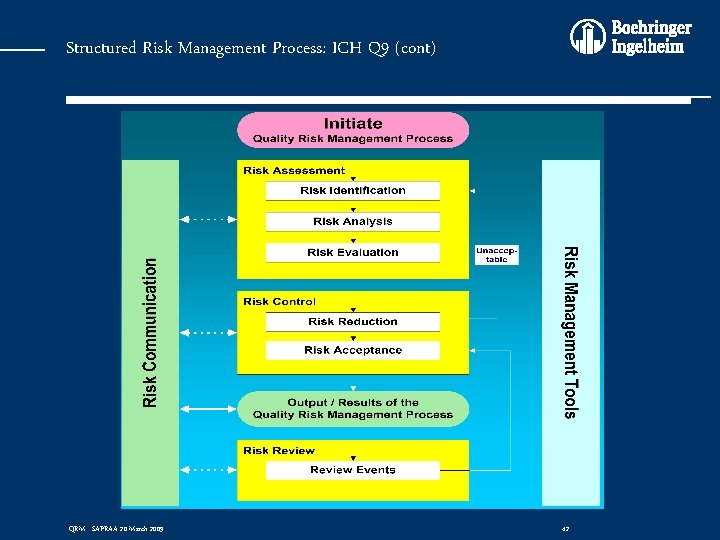
Structured Risk Management Process: ICH Q 9 (cont) QRM - SAPRAA 20 March 2009 42

Structured Risk Management Process: ICH Q 9 (cont) • When a “situation/deviation” occurs v Appoint a “situation/deviation” owner and start to investigate v Seniority dependent on initial severity assessment v Decide what approach to Risk Assessment is applicable v Use a multi-disciplinary team approach v v People with the right knowledge and experience for the situation/deviation Team Leader At least one person who understands Risk Management tools/methods FACILITATOR v Determine deadline for completion QRM - SAPRAA 20 March 2009 43
Example of process mapping QRM - SAPRAA 20 March 2009 ABCD 44
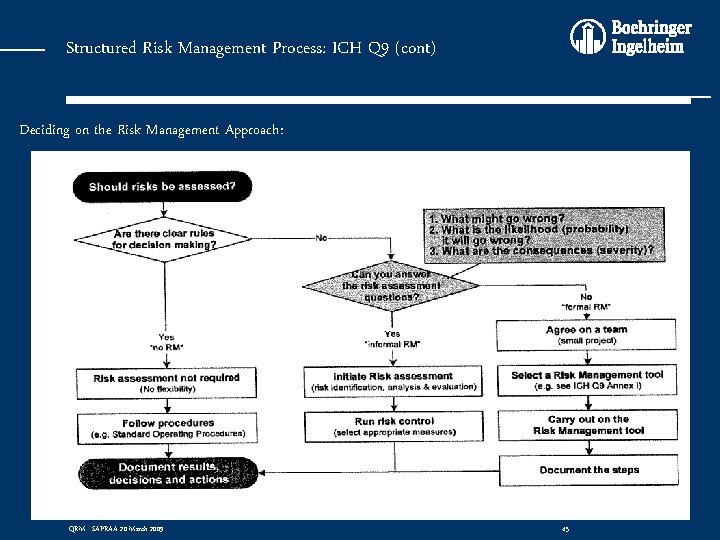
Structured Risk Management Process: ICH Q 9 (cont) Deciding on the Risk Management Approach: QRM - SAPRAA 20 March 2009 45
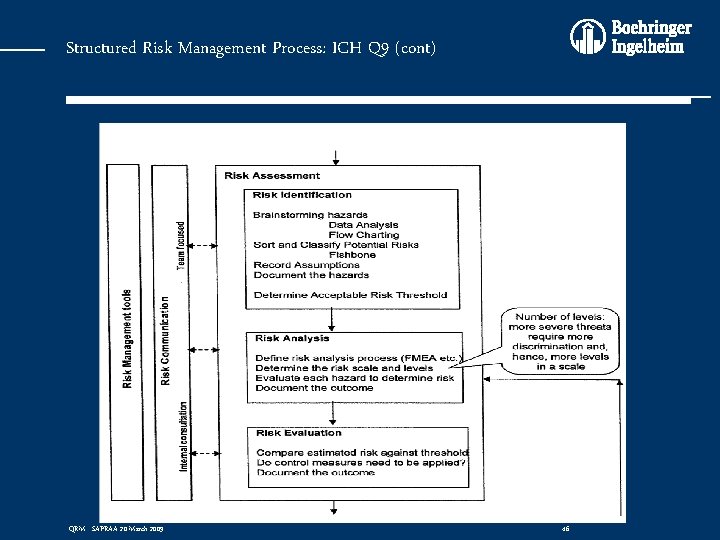
Structured Risk Management Process: ICH Q 9 (cont) QRM - SAPRAA 20 March 2009 46
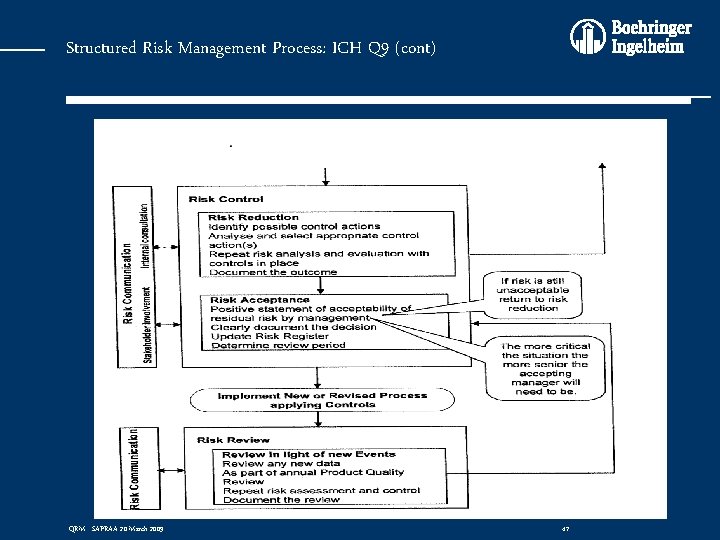
Structured Risk Management Process: ICH Q 9 (cont) QRM - SAPRAA 20 March 2009 47

Structured Risk Management Process: ICH Q 9 (cont) Formal tools of Risk Assessment according to ICH Q 9, Annex I: QRM - SAPRAA 20 March 2009 48

Structured Risk Management Process: ICH Q 9 (cont) • Conclusion: v Using a structured, systematic process for QRM more complex situations v ICH Q 9 provides a good framework for this structured v Your company should develop its own implementation adds value in approach v Focus to be pro-active QRM - SAPRAA 20 March 2009 49

Agenda q Risk Management – The Essentials q Background to QRM q Structured Risk Management Process: ICH Q 9 q Practical Implementation of QRM in your company q Summary and conclusions QRM - SAPRAA 20 March 2009 50
Practical Implementation of QRM in your company • Implementation – Making it happen in your company “If you want to make enemies, try and change something” - Woodrow Wilson To effectively plan the process to move to a more effective way of using QRM you have to look at the following: Context Organisation Culture QRM - SAPRAA 20 March 2009 51

Practical Implementation of QRM in your company (cont) • Planning for SUCCESSFUL Implementation v Involve people early on in the process v See the change from the perspective of others v Create dissatisfaction with the current situation (status quo) “ let them know there must be a better way of doing things” • Your company MUST v Accept an open “risk aware” culture v Acknowledge that ZERO RISK doesn’t exist v Accept that you have “been doing” RM from day one v RM must be kept SIMPLE QRM - SAPRAA 20 March 2009 52

Practical Implementation of QRM in your company (cont) • Planning for SUCCESSFUL Implementation (cont) v There must be a clear objective – give DIRECTION v Start small and go for early wins v Communicate extensively v Publicise your successes and give credit to people v Make sure that your company v Is aware of ICH Q 9 and the business benefits v Is aware that your competitors are already using RM as well v Does not re-invent the wheel … learn from others (HSE, etc) QRM - SAPRAA 20 March 2009 53

Practical Implementation of QRM in your company (cont) • Planning for SUCCESSFUL Implementation (cont) Important Rules to remember: v RULE 1 Ensure that you have expert knowledge of your PRODUCTS, PROCESSES, PROCEDURES at operational level, on each site and in each department v RULE 2 Ensure your organisation is aware of ICH Q 9 and the opportunity it affords Provide education and training and start with SENIOR MANAGEMENT QRM - SAPRAA 20 March 2009 54

Practical Implementation of QRM in your company (cont) • Important Rules (cont): v RULE 3 Encourage an open, risk awareness culture v Understand that risks always exist v Understand current QRM approach v Take ICH Q 9 into account during your QRM process v Identify opportunities for improvement v Where will a more structured approach add value? v Complex issues, recurring deviations QRM - SAPRAA 20 March 2009 55

Practical Implementation of QRM in your company (cont) • Important Rules (cont): v RULE 4 Keep RM SIMPLE v Focus on the outcome, not the tools v Learn from others (HSE, Medical Devices, Food Industry, etc. ) v RULE 5 Integrate QRM with existing QMS v Remember ICH Q 8, 9 and 10 work together v Q 9 needs to be applied across the whole product lifecycle v Do NOT set up separate risk departments v Coordinate implementation and allocate resources v Prioritise projects QRM - SAPRAA 20 March 2009 56

Practical Implementation of QRM in your company (cont) v RULE 6 v Be open to new ways of thinking v Expect resistance – this is normal. Work with resistance, NOT against it v Remember that you can’t force anyone to do anything … unless they really want to v Proper PLANNING = successful IMPLEMENTATION QRM is a better way of doing what you are already doing WHEN it is necessary. – Peter Gough, DBA QRM - SAPRAA 20 March 2009 57

Agenda q Risk Management – The Essentials q Background of QRM q Structured Risk Management Process: ICH Q 9 q Practical Implementation of QRM in your company q Summary and conclusions QRM - SAPRAA 20 March 2009 58

Summary and conclusions v We must do RM properly v Poor RM will not impress the regulators v The regulators will think we do not know what’s really important v If we don’t know what’s insignificant, how can we know what’s important? v If EVERYTHING is critical, NOTHING is critical Ultimately, it is all about CREDIBILITY! ****************************** QRM - SAPRAA 20 March 2009 59

Acknowledgements and References • Contents of QRM Presentation prepared for the SAPRAA Meeting of 20 March 2009 based on contents of presentation slides and notes from “Practical Application of QRM” course presented by David Begg & Associates and with the permission of David Begg & Associates to use material from DBA in this presentation. Please visit: www. david-begg-associates. com for more information about the company and services offered. • ICH Q 8, 9 and 10 (4. 01 SA • Annex 20 to the updated proposed South African GMP Guide to GMP Sept 08 v 3. doc) QRM - SAPRAA 20 March 2009 60
- Slides: 60