ABC 107442 SHAPE Study of Hypersensitivity to Abacavir

ABC 107442 (SHAPE) Study of Hypersensitivity to Abacavir and Pharmacogenetic Evaluation Michael Saag, Rukmini Balu, Elizabeth Phillips, * Philip Brachman, Claudia Martorell, William Burman, Britt Stancil, Michael Mosteller, Cindy Brothers, Paul Wannamaker, Arlene Hughes, Denise Sutherland-Phillips, Simon Mallal and Mark Shaefer for the SHAPE team *Presenting author
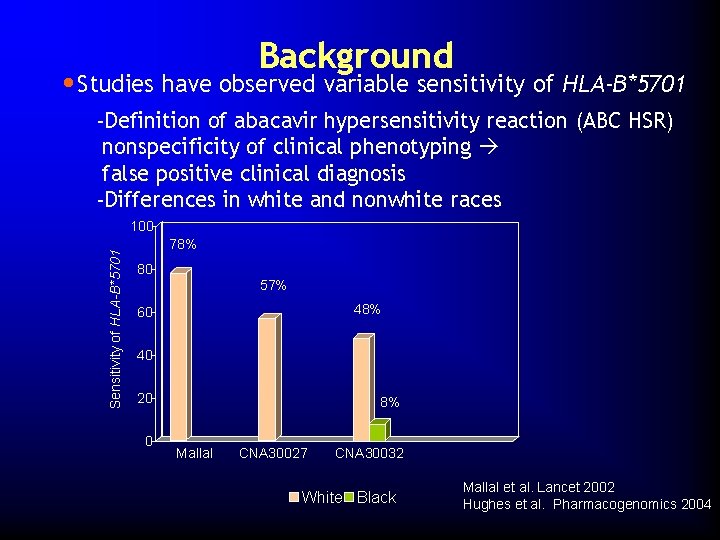
Background • Studies have observed variable sensitivity of HLA-B*5701 -Definition of abacavir hypersensitivity reaction (ABC HSR) nonspecificity of clinical phenotyping false positive clinical diagnosis -Differences in white and nonwhite races Sensitivity of HLA-B*5701 100 78% 80 57% 48% 60 40 20 0 8% Mallal CNA 30027 CNA 30032 White Black Mallal et al. Lancet 2002 Hughes et al. Pharmacogenomics 2004

What is SHAPE? · Retrospective case-control study – To evaluate the sensitivity and specificity of HLA-B*5701 – Study design necessary because of low carriage frequency of HLA-B*5701 and low rates of ABC HSR in Blacks · Addresses two important aspects of pharmacogenetic research – Generalizability to White and Black races – Supplement clinical definition of ABC HSR with patch test (phenotype)
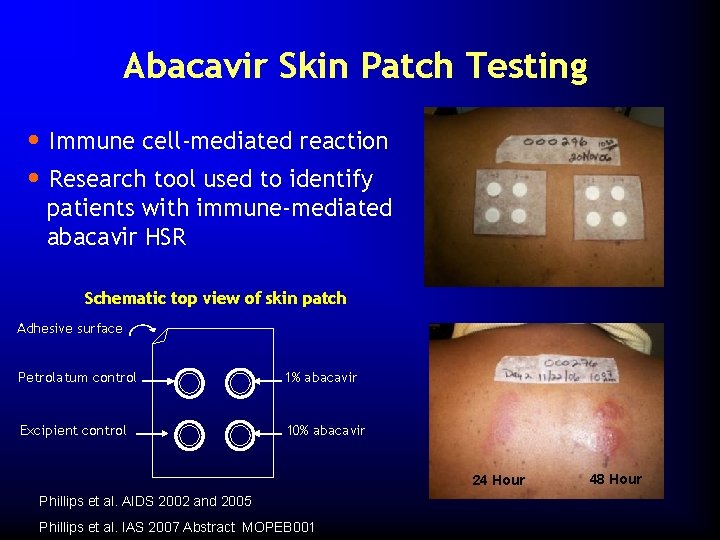
Abacavir Skin Patch Testing • Immune cell-mediated reaction • Research tool used to identify patients with immune-mediated abacavir HSR Schematic top view of skin patch Adhesive surface Petrolatum control 1% abacavir Excipient control 10% abacavir 24 Hour Phillips et al. AIDS 2002 and 2005 Phillips et al. IAS 2007 Abstract MOPEB 001 48 Hour
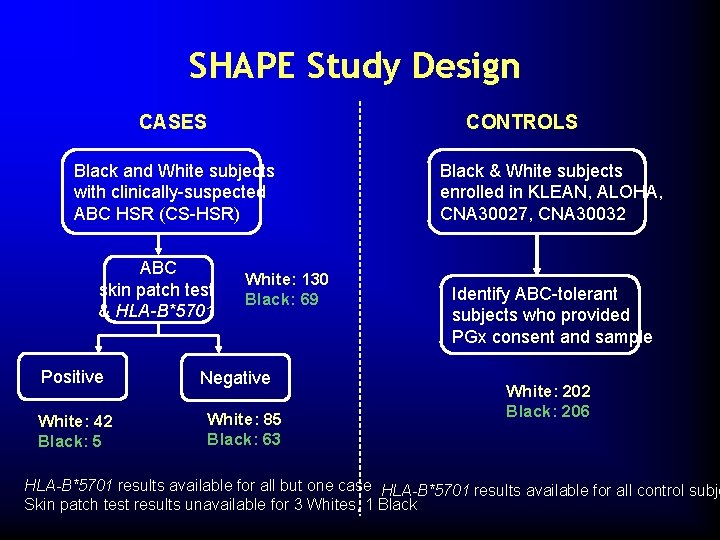
SHAPE Study Design CONTROLS CASES Black and White subjects with clinically-suspected ABC HSR (CS-HSR) ABC skin patch test & HLA-B*5701 Positive White: 42 Black: 5 White: 130 Black: 69 Negative White: 85 Black: 63 Black & White subjects enrolled in KLEAN, ALOHA, CNA 30027, CNA 30032 Identify ABC-tolerant subjects who provided PGx consent and sample White: 202 Black: 206 HLA-B*5701 results available for all but one case HLA-B*5701 results available for all control subje Skin patch test results unavailable for 3 Whites, 1 Black

Results
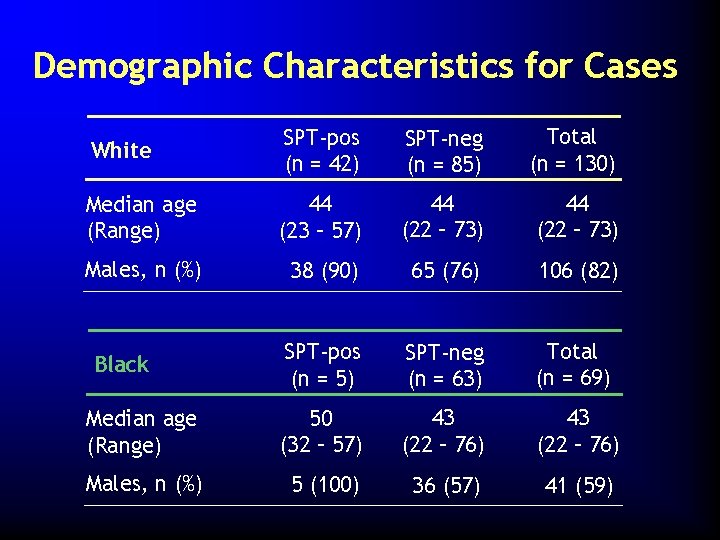
Demographic Characteristics for Cases White SPT-pos (n = 42) SPT-neg (n = 85) Total (n = 130) Median age (Range) 44 (23 – 57) 44 (22 – 73) Males, n (%) 38 (90) 65 (76) 106 (82) SPT-pos (n = 5) SPT-neg (n = 63) Total (n = 69) Median age (Range) 50 (32 – 57) 43 (22 – 76) Males, n (%) 5 (100) 36 (57) 41 (59) Black
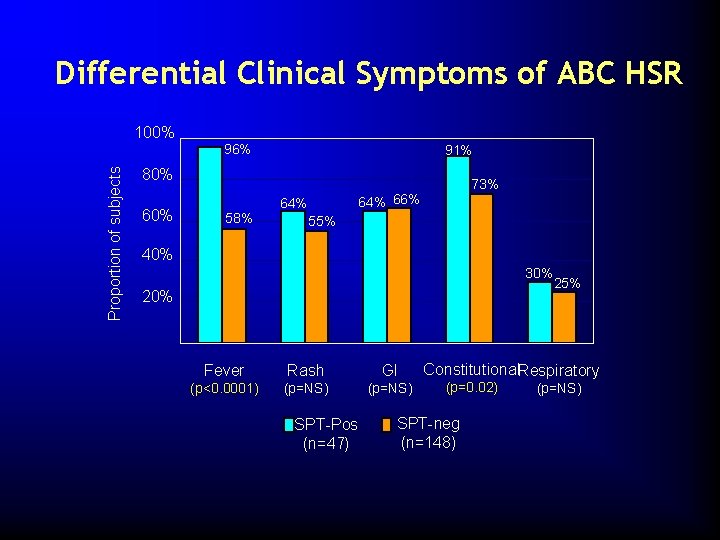
Differential Clinical Symptoms of ABC HSR 100% Proportion of subjects 96% 91% 80% 60% 58% 73% 64% 66% 64% 55% 40% 30% 20% Fever Rash GI (p<0. 0001) (p=NS) SPT-Pos (n=47) 25% Constitutional. Respiratory (p=0. 02) SPT-neg (n=148) (p=NS)
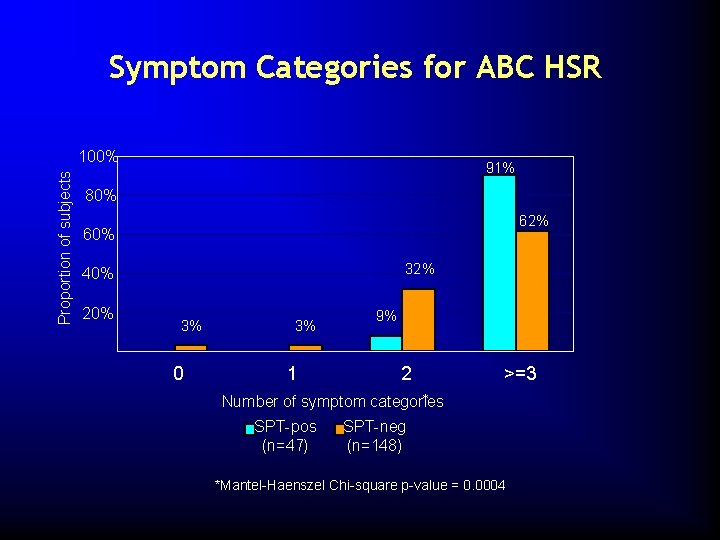
Symptom Categories for ABC HSR Proportion of subjects 100% 91% 80% 62% 60% 32% 40% 20% 3% 3% 0 3% 1 9% 2 >=3 * Number of symptom categories SPT-pos (n=47) SPT-neg (n=148) *Mantel-Haenszel Chi-square p-value = 0. 0004
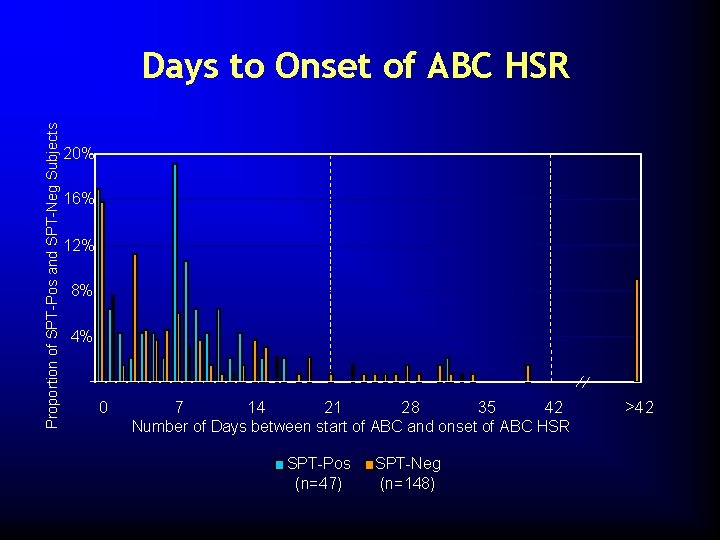
Proportion of SPT-Pos and SPT-Neg Subjects Days to Onset of ABC HSR 20% 16% 12% 8% 4% 0 7 14 21 28 35 42 Number of Days between start of ABC and onset of ABC HSR SPT-Pos (n=47) SPT-Neg (n=148) >42
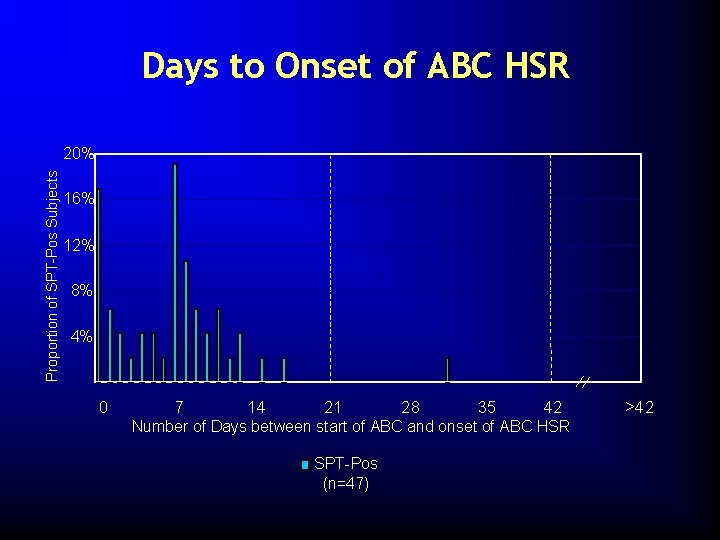
Days to Onset of ABC HSR Proportion of SPT-Pos Subjects 20% 16% 12% 8% 4% 0 7 14 21 28 35 42 Number of Days between start of ABC and onset of ABC HSR SPT-Pos (n=47) >42
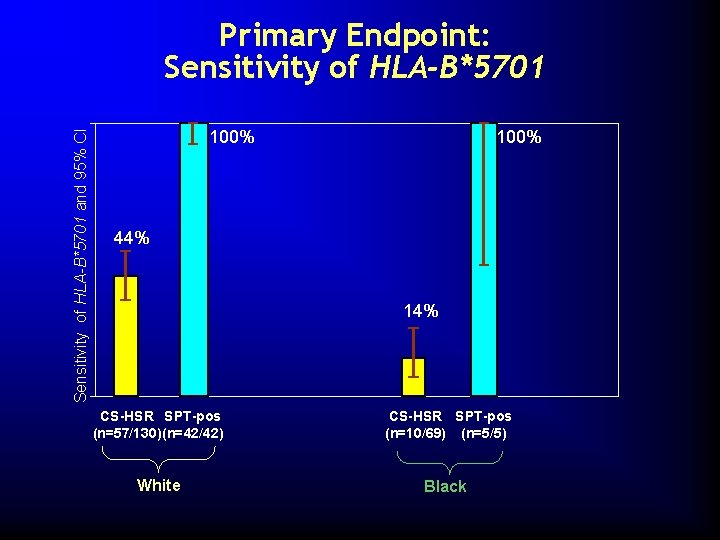
Sensitivity of HLA-B*5701 and 95% CI Primary Endpoint: Sensitivity of HLA-B*5701 100% 44% 14% CS-HSR SPT-pos (n=57/130) (n=42/42) CS-HSR SPT-pos (n=10/69) (n=5/5) White Black
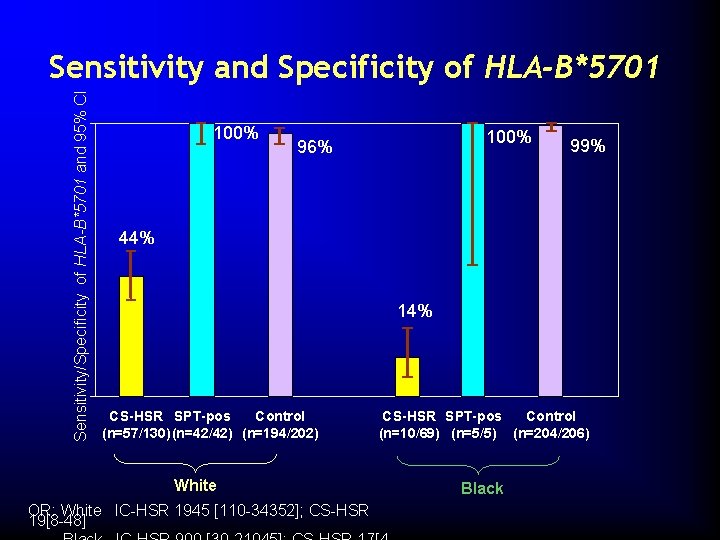
Sensitivity/Specificity of HLA-B*5701 and 95% CI Sensitivity and Specificity of HLA-B*5701 100% 96% 99% 44% 14% CS-HSR SPT-pos Control (n=57/130) (n=42/42) (n=194/202) White OR: White IC-HSR 1945 [110 -34352]; CS-HSR 19[8 -48] CS-HSR SPT-pos Control (n=10/69) (n=5/5) (n=204/206) Black
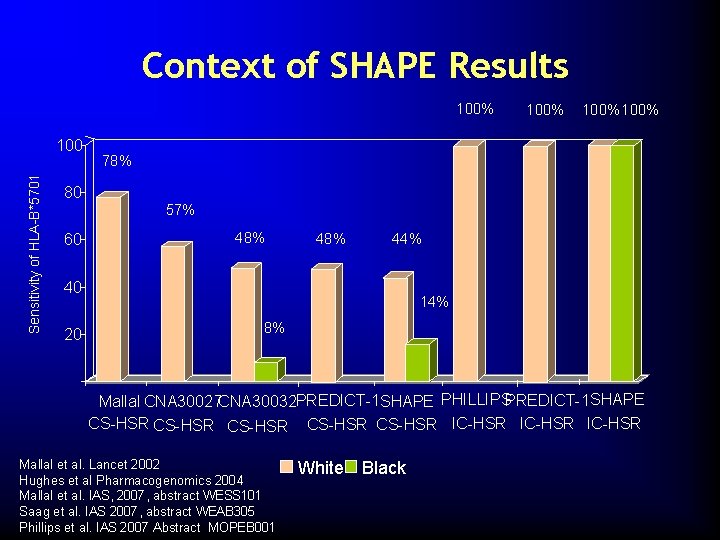
Context of SHAPE Results 100% Sensitivity of HLA-B*5701 100% 78% 80 57% 60 48% 44% 40 20 14% 8% Mallal CNA 30027 CNA 30032 PREDICT-1 SHAPE PHILLIPSPREDICT-1 SHAPE CS-HSR CS-HSR IC-HSR Mallal et al. Lancet 2002 Hughes et al Pharmacogenomics 2004 Mallal et al. IAS, 2007, abstract WESS 101 Saag et al. IAS 2007, abstract WEAB 305 Phillips et al. IAS 2007 Abstract MOPEB 001 White Black
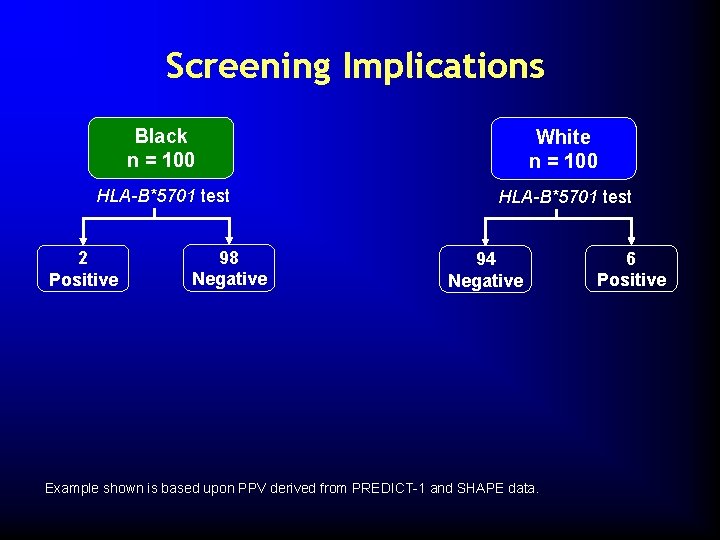
Screening Implications Black n = 100 White n = 100 HLA-B*5701 test 2 Positive 98 Negative 94 Negative Example shown is based upon PPV derived from PREDICT-1 and SHAPE data. 6 Positive
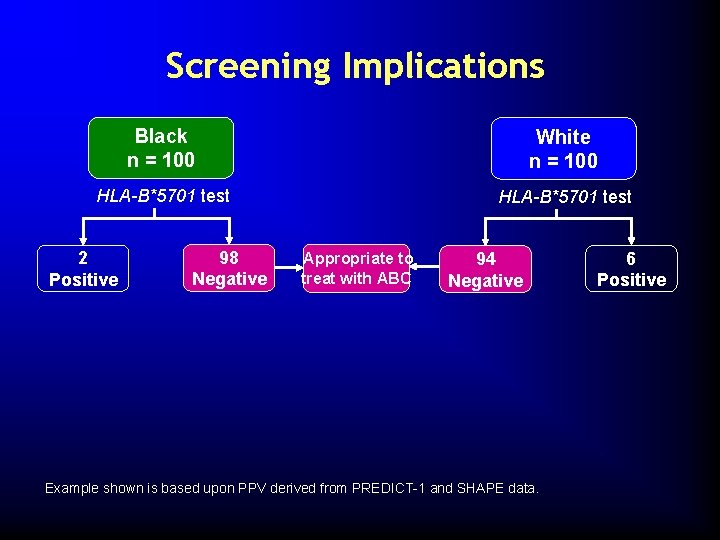
Screening Implications Black n = 100 White n = 100 HLA-B*5701 test 2 Positive 98 Negative Appropriate to treat with ABC 94 Negative Example shown is based upon PPV derived from PREDICT-1 and SHAPE data. 6 Positive
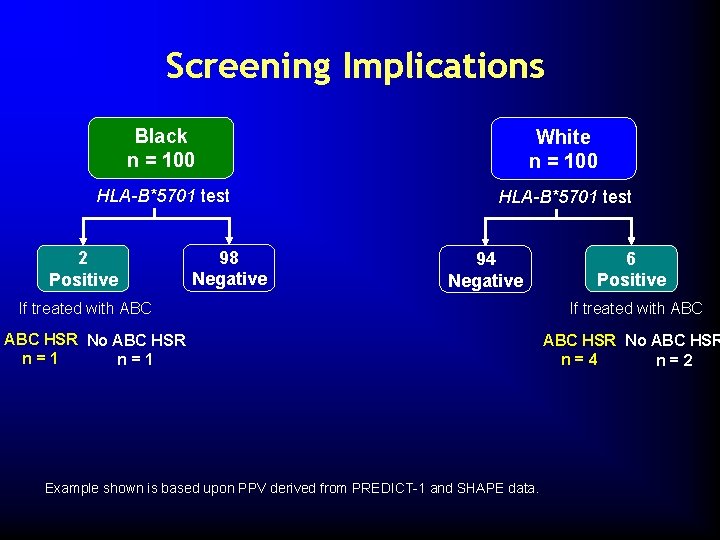
Screening Implications Black n = 100 White n = 100 HLA-B*5701 test 2 Positive 98 Negative 94 Negative If treated with ABC HSR No ABC HSR n=1 Example shown is based upon PPV derived from PREDICT-1 and SHAPE data. 6 Positive If treated with ABC HSR No ABC HSR n=4 n=2
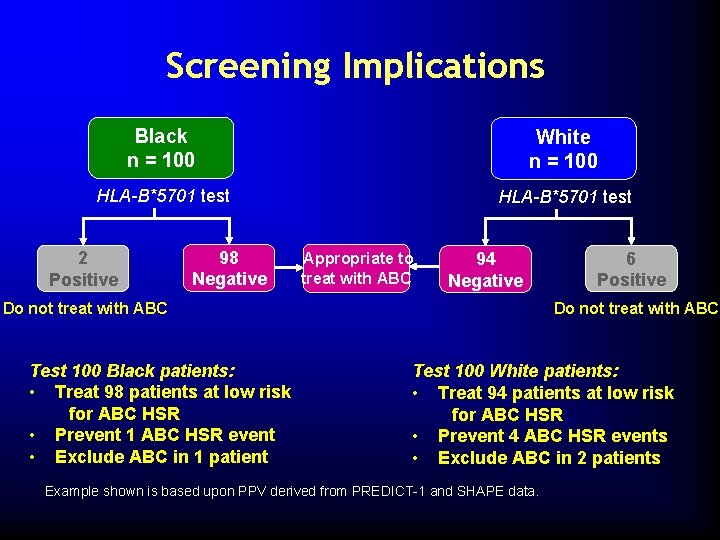
Screening Implications Black n = 100 White n = 100 HLA-B*5701 test 2 Positive 98 Negative Appropriate to treat with ABC 94 Negative Do not treat with ABC Test 100 Black patients: • Treat 98 patients at low risk for ABC HSR • Prevent 1 ABC HSR event • Exclude ABC in 1 patient 6 Positive Test 100 White patients: • Treat 94 patients at low risk for ABC HSR • Prevent 4 ABC HSR events • Exclude ABC in 2 patients Example shown is based upon PPV derived from PREDICT-1 and SHAPE data.

Conclusions · Results in Whites consistent with the randomized study PREDICT-1 (Abstract WESS 101) that demonstrated pretherapy screening for HLA-B*5701 significantly reduced both clinical and immunologically confirmed ABC HSR. · Similar results seen in Blacks imply that screening for HLAB*5701 is a broadly generalizable approach for reducing ABC HSR rates.

Acknowledgments All patients who participated in the study. Study investigators: Clinical Operations: Statistics: O Adeyemi; B Akil; D Antoniskis; D Berger; G Blick; M Borucki; P Brachman; W Burman, P Cohen; P Cook; R Corales; D Coulston, L Crane; C Damodaran; E De. Jesus; J Eron; T File; J Gallant; J Giron; S Green; R Greenberg; R Hsu; T Jefferson; A La. Marca; R Liporace; C Martorell; C Mc. Donald; D Mc. Donough; P Mc. Leroth; A Mills; K Mounzer; K Mullane; C Newman, S O’Brien; T Overton; A Petroll; A Ramani; G Richmond; J Rodriguez; M Saag; P Salvato; C Schiessl; S Schrader; A Scribner; P Shalit; L Sloan; C Small; D Stein; M Thompson; J Torres; W Towner; A Voskanian; D Walshe; W Weinberg; D Wright; B Young; C Zurawski Annie Cameron; Steve Chriscoe; Susan Porter. Atkinson; Camden Breeden Britt Stancil; Tiffany Davis; Mark Edwards; Shannon La. Belle; Amy Cutrell Safety: Christopher Stainsby; Helen Goumeas; Lloyd Curtis Pharmacogenetics support: Anita Nelsen; Keith Nangle; Morlisa Dixon; Jill Ratchford Data Management: Shannon Gooding; Ying Zhang Expert Committee: Neil Shear; Bridgette Milpied; Derk Bruynzeel Clinical input: Denise Sutherland-Phillips; Jaime Hernandez PREDICT-1: Nick Fitch Regulatory input: Patch testing supplies: Martha Anne Moore Sunnybrook Hospital, Toronto Monitoring: Amy Schecterson; Tonya Lai
- Slides: 20