A Tour of the Periodic Table Packet 11

A Tour of the Periodic Table Packet #11

Mendeleev’s Periodic Table • From 1829 – 1869, different systems were proposed. • In 1869, Russian chemist and teacher, Dmitri Mendeleev, published a table of elements.

Mendeleev’s Periodic Table • Mendeleev arranged the elements in his periodic table in order of increasing atomic mass. • This caused problems. • Scientist had not yet determined the structure of the atom. • Mendeleev predicted that elements would be discovered and used to fill spaces where he had placed question marks.
Modern Periodic Table • In 1913, Henry Moseley organized the elements by increasing atomic and resolved discrepancies in Mendeleev’s arrangement.
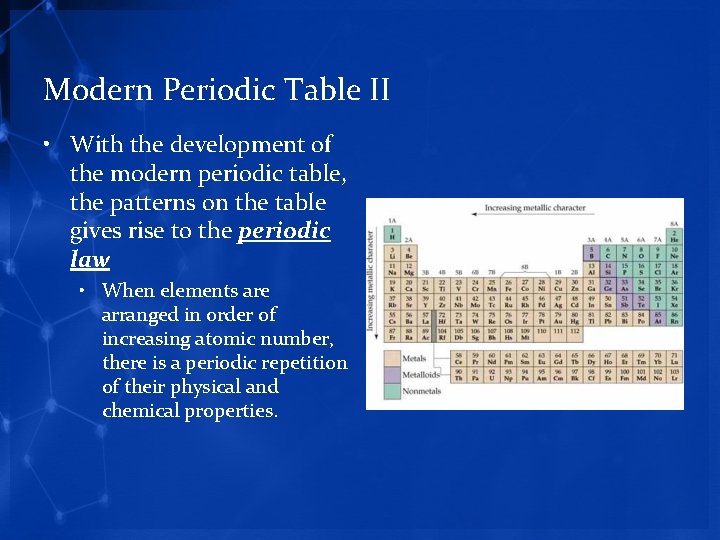
Modern Periodic Table II • With the development of the modern periodic table, the patterns on the table gives rise to the periodic law • When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties.

Modern Periodic Table III • The International Union of Pure and Applied Chemistry (IUPAC) is an orgnaization that sets the standatds for chemistry. • In 1985, the IUPAC proposed a new system for labeling groups on the periodic table. • The groups are numbered 1 – 18. • Dividing the elements into the groups is not the only way to classify them based on their properties.

CLASSES OF ELEMENTS

Classes of Elements • There are three main “classes” of elements. • Metals • Nonmetals • Metalloids • Across the periodic table, the properties of the elements become less metallic and more non-metallic

Physical Properties of Metals • • Good conductors of heat and electric current. Malleability • Most metals can be hammered into thin sheets without breaking. • Ductility • Most metals can be put into wires • Lustrous Appearance • A freshly cleaned or cut surface will have a high luster, or sheen. • Sheen is caused by the metal’s ability to reflect light.

Properties of Non-metals (Elements of the Upper Right Corner) • • Greater variation in physical properties when compared to metals. Most are gases at room temperature. • • Few are solids at room temperature • • • Dark-red liquid In general, poor conductors of heat and electricity. • • Sulfur Phosphorus Bromine • • Nitrogen, oxygen, chlorine & neon Carbon exception to rule. Non-metals are brittle.

Metalloids (Semimetals) {Stair-step line that separates metals from nonmetals} • • • Has properties similar to metals and non-metals. Depending on conditions, may behave as a metal while as a non-metal in others. Example • Silicon is a poor conductor of electric current. • However, if boron is mixed with silicon, the mixture is a good conductor of electricity. • Silicon can be cut into wafers.

GROUPS ON THE PERIODIC TABLE

Groups on the Periodic Table I • Families of elements with similar chemical properties that lie in the same vertical column in the periodic table are called groups. • Often referred to by the number over the column.
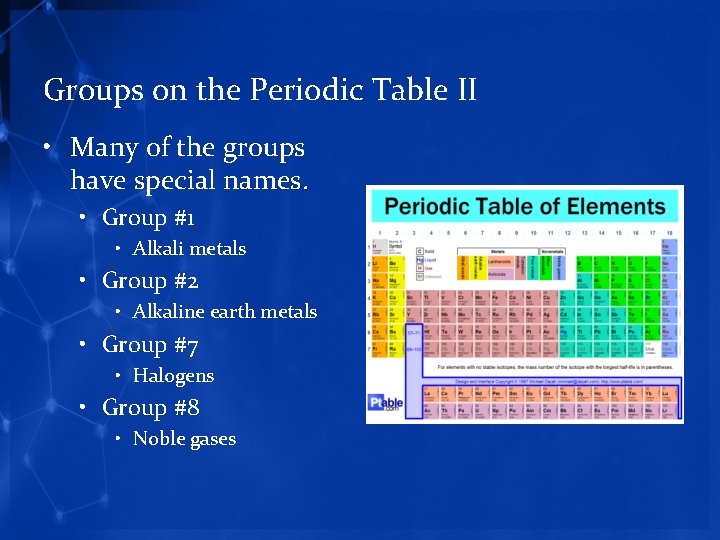
Groups on the Periodic Table II • Many of the groups have special names. • Group #1 • Alkali metals • Group #2 • Alkaline earth metals • Group #7 • Halogens • Group #8 • Noble gases
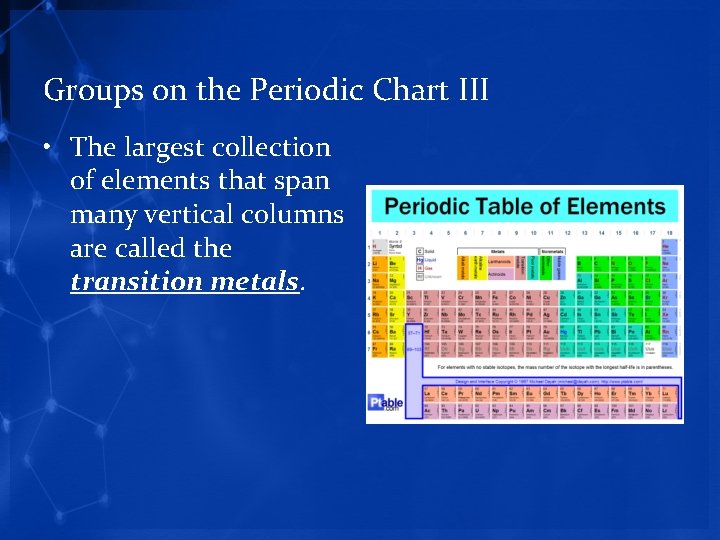
Groups on the Periodic Chart III • The largest collection of elements that span many vertical columns are called the transition metals.

NATURAL STATES OF ELEMENTS

Natural States I • Most matter is in the form of a mixture. • Mentioned previously, most mixtures contain compounds. • Atoms from different elements bounded together • Therefore, most elements are “reactive. ” • Most elements are not found, in nature, in a pure form.

Natural States II • Exceptions to the rule • Gold rush of late 1800’s • Gold nuggets were found in purest form. • Platinum & Silver • Often nearly found in the pure form. • Gold, platinum and silver are in a group of metals called noble metals. • These are relatively un-reactive. • Similar to the behavior of group #8 elements—the noble gases.

Natural States III—Diatomic Molecules • Diatomic molecules • Molecules made up of two atoms. • Examples • Nitrogen • N 2 • Oxygen • O 2 • Hydrogen* • H 2 • Sodium Chloride • Na. Cl • Group #7

Natural State IV—Liquid Form • Only two elements are in liquid form when in their elemental form (most natural form) • Bromine • B 2 • Non-metal • Also diatomic • Mercury • Metal

Natural State V—Unique Facts • Carbon • Allotrope • Solid carbon exist in three different forms • Diamond • Hardest natural substance known • Graphite • Soft material (lead in pencils) • Buckminsterfullerene • Dome shape composed of ≈60 carbon atoms.

REVIEW
- Slides: 22