A students work without solutions manual 10 problemsnight

“A” students work (without solutions manual) ~ 10 problems/night. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours W – F 2 -3 pm Module #10: Covalent Bonding

Chemistry General FITCH Rules G 1: Suzuki is Success G 2. Slow me down G 3. Scientific Knowledge is Referential G 4. Watch out for Red Herrings G 5. Chemists are Lazy C 1. It’s all about charge C 2. Everybody wants to “be like Mike” (grp. 18) C 3. Size Matters C 4. Still Waters Run Deep C 5. Alpha Dogs eat first

Properties and Measurements Property Size Volume Weight Unit m cm 3 gram Temperature 1. 66053873 x 10 -24 g quantity mole Pressure Energy: Thermal Kinetic Reference State size of earth m mass of 1 cm 3 water at specified Temp (and Pressure) o. C, K boiling, freezing of water (specified Pressure) amu (mass of 1 C-12 atom)/12 atomic mass of an element in grams atm, mm Hg earth’s atmosphere at sea level BTU calorie J Energy, of electrons Electronegativity 1 lb water 1 o. F 1 g water 1 o. C 2 kg mass moving at 1 m/s energy of electron in a vacuum F

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.
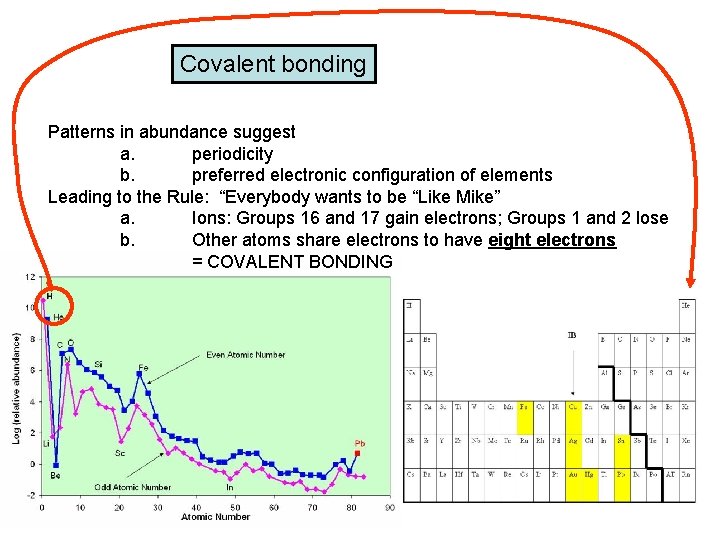
Covalent bonding Patterns in abundance suggest a. periodicity b. preferred electronic configuration of elements Leading to the Rule: “Everybody wants to be “Like Mike” a. Ions: Groups 16 and 17 gain electrons; Groups 1 and 2 lose b. Other atoms share electrons to have eight electrons = COVALENT BONDING
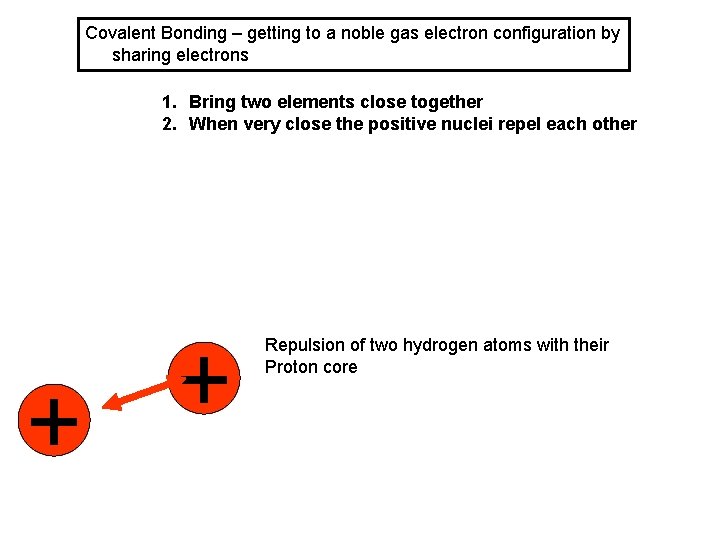
Covalent Bonding – getting to a noble gas electron configuration by sharing electrons 1. Bring two elements close together 2. When very close the positive nuclei repel each other + + Repulsion of two hydrogen atoms with their Proton core
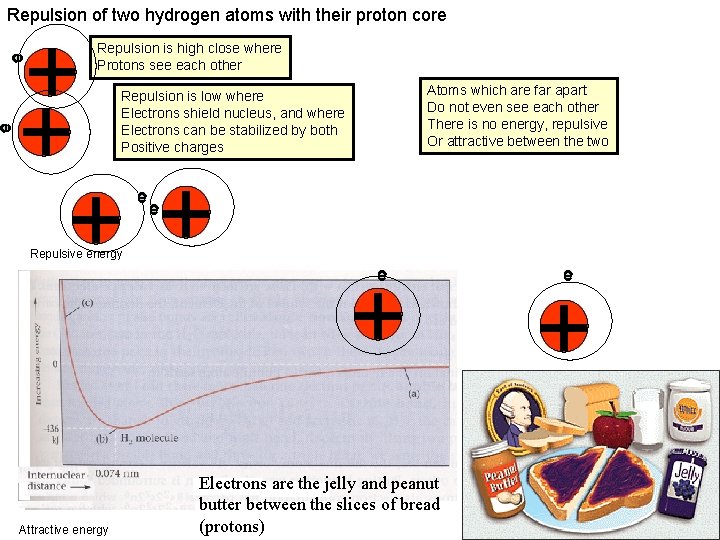
Repulsion of two hydrogen atoms with their proton core e e ++ Repulsion is high close where Protons see each other Atoms which are far apart Do not even see each other There is no energy, repulsive Or attractive between the two Repulsion is low where Electrons shield nucleus, and where Electrons can be stabilized by both Positive charges ++ e e Repulsive energy Attractive energy + e Electrons are the jelly and peanut butter between the slices of bread (protons) e +

“A” students work (without solutions manual) ~ 10 problems/night. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours W – F 2 -3 pm

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.

Lewis dot structure (electron dot Structure or diagram) are diagrams that Show the bonding between atoms of A molecule based on shared “valence” shell (outer shell) electrons and shows the Presence of any “lone pair” of electrons That may exist in the covalently bonded Molecule. Covalent Gilbert Newton Lewis 1875 -1946; Caltech Physical Chemist Valence = outermost shell electrons of an atom shared Latin: valere – to be strong
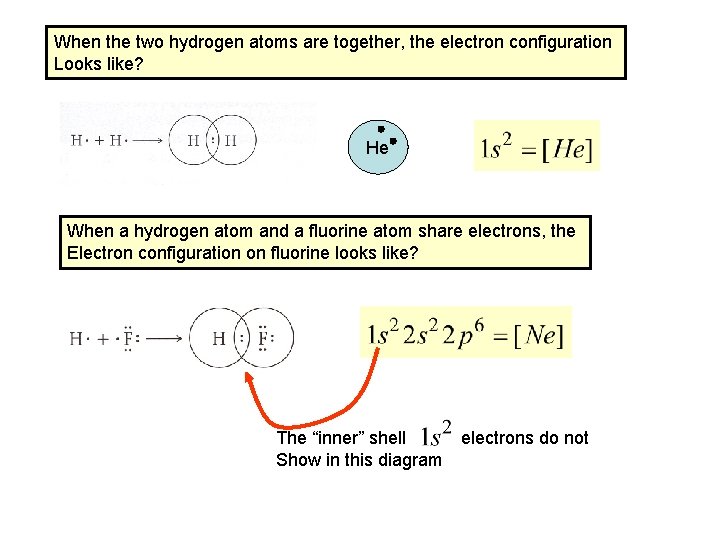
When the two hydrogen atoms are together, the electron configuration Looks like? He When a hydrogen atom and a fluorine atom share electrons, the Electron configuration on fluorine looks like? The “inner” shell electrons do not Show in this diagram
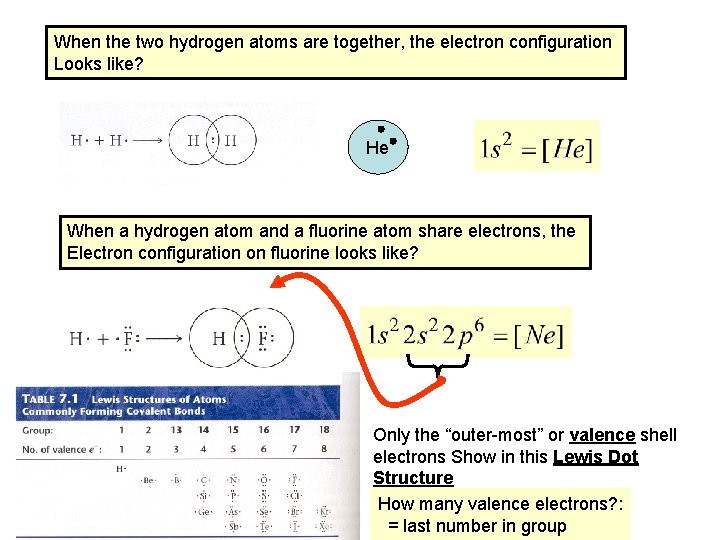
When the two hydrogen atoms are together, the electron configuration Looks like? He When a hydrogen atom and a fluorine atom share electrons, the Electron configuration on fluorine looks like? Only the “outer-most” or valence shell electrons Show in this Lewis Dot Structure How many valence electrons? : = last number in group
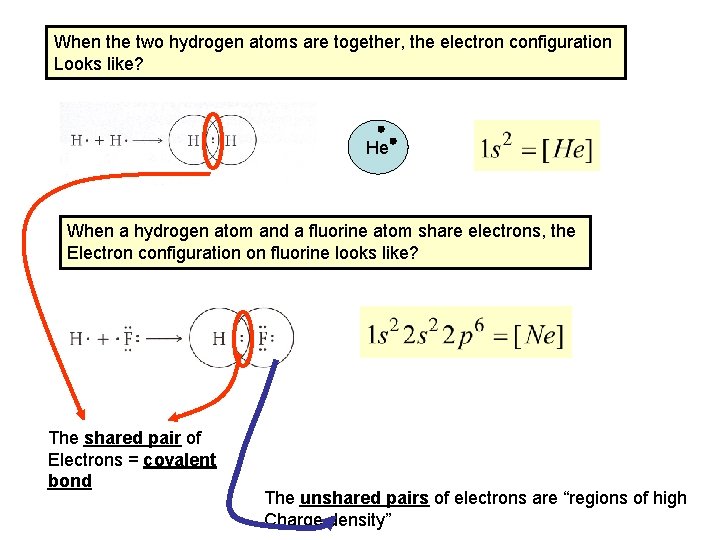
When the two hydrogen atoms are together, the electron configuration Looks like? He When a hydrogen atom and a fluorine atom share electrons, the Electron configuration on fluorine looks like? The shared pair of Electrons = covalent bond The unshared pairs of electrons are “regions of high Charge density”

When a hydrogen atom and an oxygen atom share valence electrons plus an Extra electron, the electron configuration on hydrogen and oxygen look like? Valence electrons on oxygen? Valence electrons on hydrogen? Invoking Rule: Chemists are Lazy the diagram above is too tedious to write out all the time make shared electrons (bond) a line Lewis dot structure for hydroxide The single electron pair shaired between the two bonded atoms Is called a single bond It is drawn as a line.
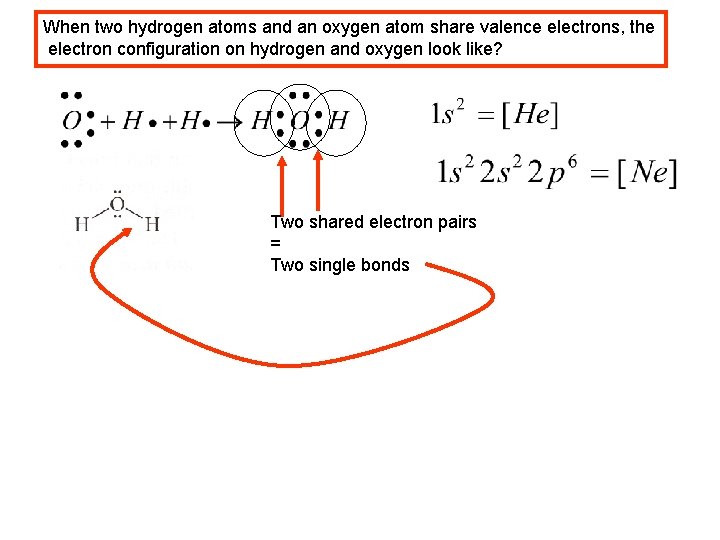
When two hydrogen atoms and an oxygen atom share valence electrons, the electron configuration on hydrogen and oxygen look like? Two shared electron pairs = Two single bonds

When two hydrogen atoms and an oxygen atom share valence electrons, the electron configuration on hydrogen and oxygen look like? When three hydrogen atoms and a nitrogen atom share valence electrons, the electron configuration on hydrogen and nitrogen look like?

When two hydrogen atoms and an oxygen atom share valence electrons, the electron configuration on hydrogen and oxygen look like? When three hydrogen atoms and a nitrogen atom share valence electrons, the electron configuration on hydrogen and nitrogen look like? Valence shell of nitrogen? Three pairs of shared electrons = three single bonds
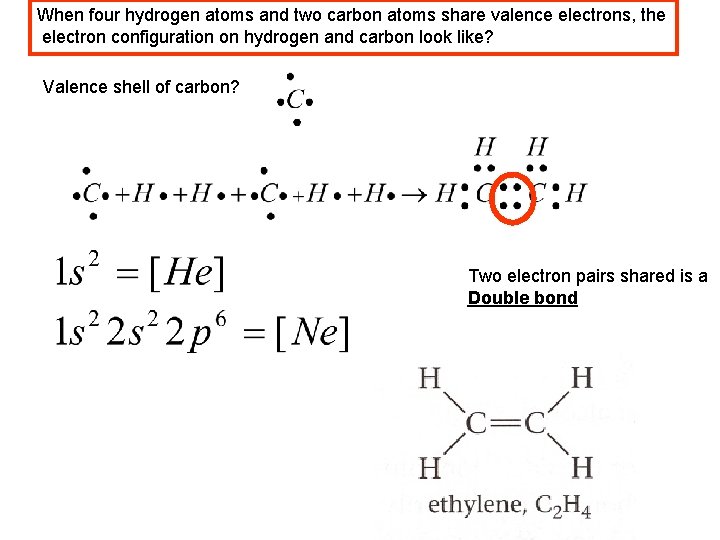
When four hydrogen atoms and two carbon atoms share valence electrons, the electron configuration on hydrogen and carbon look like? Valence shell of carbon? Two electron pairs shared is a Double bond

When two hydrogen atoms and two carbon atoms share valence electrons, the electron configuration on hydrogen and carbon look like? Three electron pairs shared is a Triple bond

Rules for Writing Lewis Dot Structures 1. Count the number of valence electrons (last number of group) of all atoms a. For an anion add the appropriate extra number of electrons b. For a cation subtract the appropriate extra number of electrons 2. Draw a molecular skeleton, joining by single bonds to the central atom. a. The central is usually the atom written first in the formula (N in NH 4+, S in SO 2, and C in CCl 4). b. The terminal atoms are usually H, O. c. Halogens are always terminal atoms. 3. Determine the number of valence electrons still available for distribution after subtracting two electrons for each single bond. 4. Determine the number of electrons required to complete the octet a. H gets only two electrons b. Other exceptions to be noted below 5. Fill in the region required for the octet. 6. Make up deficit of electrons by creating double bonds a. C, N, O, S H can only have one bond because it can share only one Electron. Poor H. Halogens have lots of electrons but really do not like to share. Greedy halogens All they want is one more to make up the Mike configuration
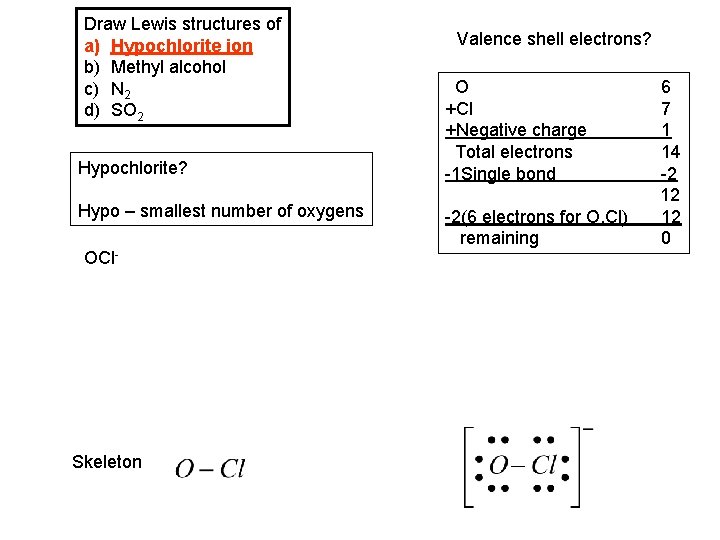
Draw Lewis structures of a) Hypochlorite ion b) Methyl alcohol c) N 2 d) SO 2 Hypochlorite? Hypo – smallest number of oxygens OCl- Skeleton Valence shell electrons? O +Cl +Negative charge Total electrons -1 Single bond -2(6 electrons for O, Cl) remaining 6 7 1 14 -2 12 12 0

Draw Lewis structures of a) Hypochlorite ion b) Methyl alcohol, CH 3 OH c) N 2 d) SO 2 Skeleton Carbon is first in formula Hydrogen is always terminal Valence shell electrons? O +C +4(H) Negative charge Total electrons -5 single bonds remaining -octet for oxygen remaining 6 4 4 0 14 -10 4 -4 0 Octets Carbon has its octet Hydrogen has its duet Oxygen requires 4 more electrons

Draw Lewis structures of a) Hypochlorite ion b) Methyl alcohol, CH 3 OH c) N 2 d) SO 2 Skeleton Octets Each nitrogen requires 6 more Valence shell electrons? 2 N Negative charge Total electrons -1 single bond Remaining Octet completion Difference 10 0 10 -2 8 -12 -4 2 N Negative charge Total electrons -3 single bonds Remaining 10 0 10 -6 4 We are short 4 electrons for the octet, The only way to get extra ones is to Share four more electrons = triple Bond. Place the remaining 4 electrons equally On the two equal nitrogens

Draw Lewis structures of a) Hypochlorite ion b) Methyl alcohol, CH 3 OH c) N 2 d) SO 2 Valence shell electrons? 2 O +1 S Negative charge Total electrons -2(single bonds) Remaining electrons Octet for S 2(Octet for each O) Deficit? 12 6 0 18 -4 14 -4 -12 -2 2 O +1 S Negative charge Total electrons -3(single bonds) 12 6 0 18 -6 12 Skeleton, First atom in formula is central Octets We are short 2 electrons for the octet, The only way to get extra ones is to Share two more electrons = double bond. Place the remaining 12 electrons to fill octets
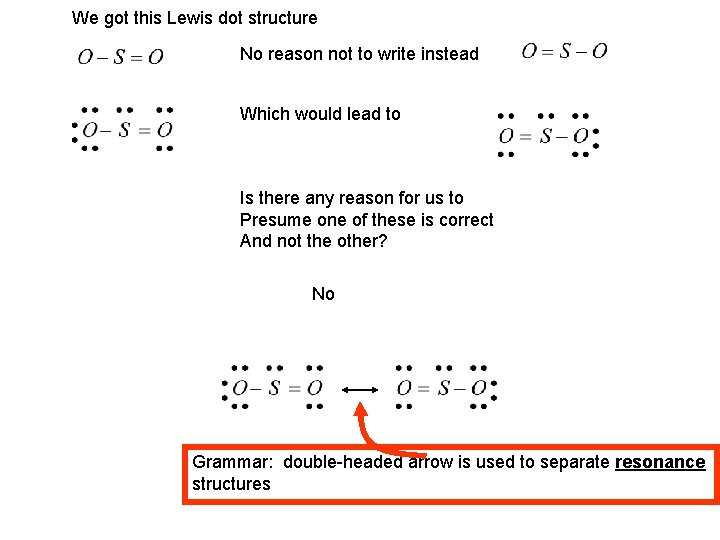
We got this Lewis dot structure No reason not to write instead Which would lead to Is there any reason for us to Presume one of these is correct And not the other? No Grammar: double-headed arrow is used to separate resonance structures

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.
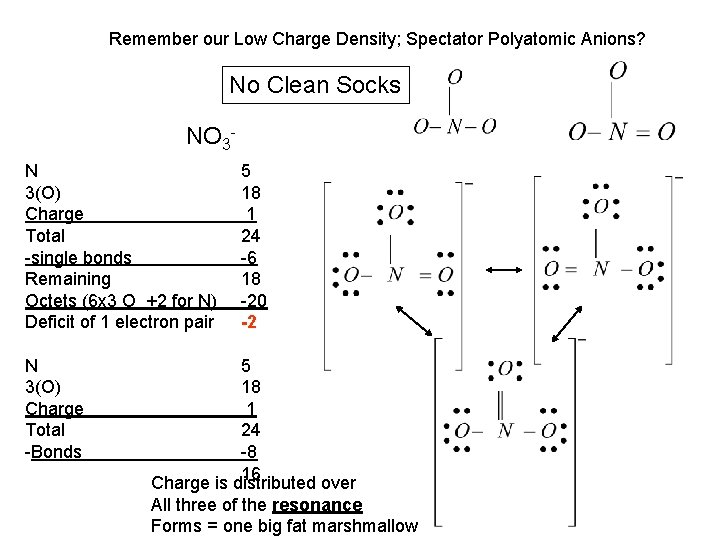
Remember our Low Charge Density; Spectator Polyatomic Anions? No Clean Socks NO 3 N 3(O) Charge Total -single bonds Remaining Octets (6 x 3 O +2 for N) Deficit of 1 electron pair N 3(O) Charge Total -Bonds 5 18 1 24 -6 18 -20 -2 5 18 1 24 -8 16 Charge is distributed over All three of the resonance Forms = one big fat marshmallow

Resonance 1. The “real” molecule is none of the three nitrates we drew but something intermediate to the three. 2. Resonance can be “assumed” or “predicted” when there are equally plausible Lewis dot structures. 3. Resonance forms differ only in the distribution of electrons and not in the arrangement of atoms.
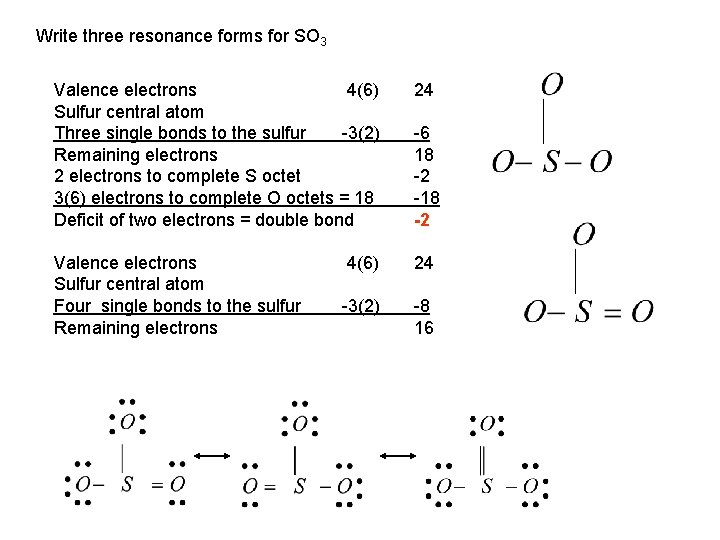
Write three resonance forms for SO 3 Valence electrons 4(6) Sulfur central atom Three single bonds to the sulfur -3(2) Remaining electrons 2 electrons to complete S octet 3(6) electrons to complete O octets = 18 Deficit of two electrons = double bond 24 Valence electrons Sulfur central atom Four single bonds to the sulfur Remaining electrons 4(6) 24 -3(2) -8 16 -6 18 -2 -18 -2

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.

Formal Charge helps determine the correct Lewis Dot Structure X=number of valence e- in the free atom (last number of group) Y = number of unshared e- owned by the atom in the Lewis structure Z = number of bonding e- shared by the atom in the Lewis structure The correct Lewis dot structure is generally the one in which a. The formal charges are as close to zero as possible b. Negative charge is located on the more electronegative atom

X=number of valence e- in the free atom (last number of group) Y = number of unshared e- owned by the atom in the Lewis structure Z = number of bonding e- shared by the atom in the Lewis structure Which is correct?

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.
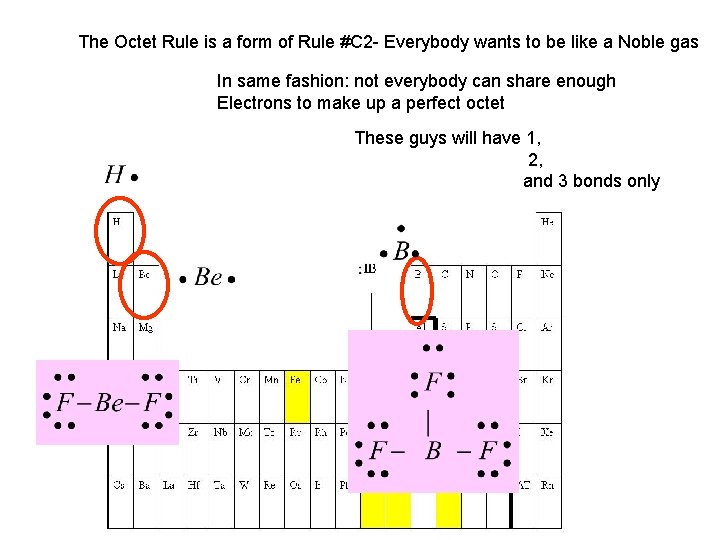
The Octet Rule is a form of Rule #C 2 - Everybody wants to be like a Noble gas In same fashion: not everybody can share enough Electrons to make up a perfect octet These guys will have 1, 2, and 3 bonds only
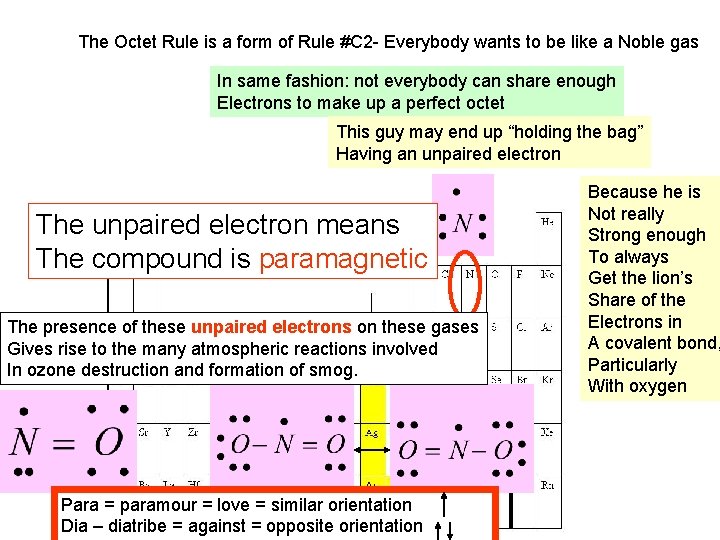
The Octet Rule is a form of Rule #C 2 - Everybody wants to be like a Noble gas In same fashion: not everybody can share enough Electrons to make up a perfect octet This guy may end up “holding the bag” Having an unpaired electron The unpaired electron means The compound is paramagnetic The presence of these unpaired electrons on these gases Gives rise to the many atmospheric reactions involved In ozone destruction and formation of smog. Para = paramour = love = similar orientation Dia – diatribe = against = opposite orientation Because he is Not really Strong enough To always Get the lion’s Share of the Electrons in A covalent bond, Particularly With oxygen
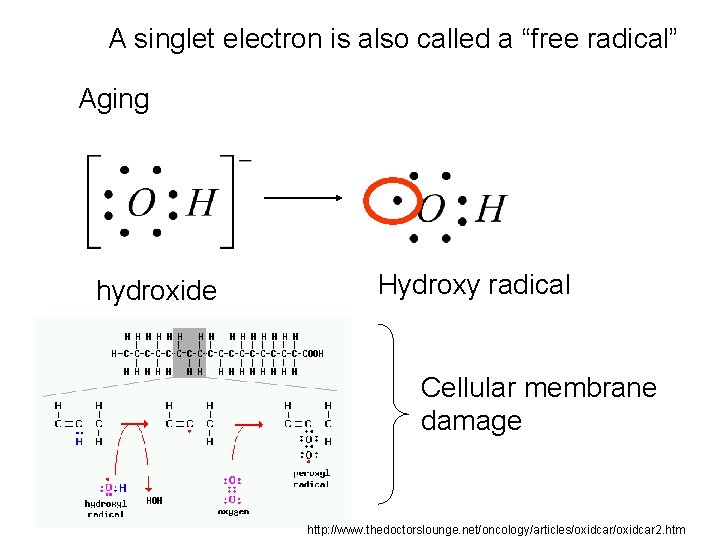
A singlet electron is also called a “free radical” Aging hydroxide Hydroxy radical Cellular membrane damage http: //www. thedoctorslounge. net/oncology/articles/oxidcar 2. htm
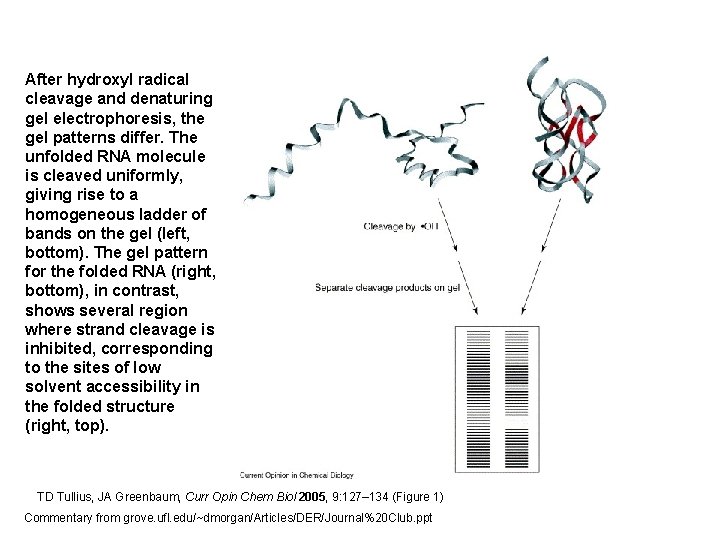
After hydroxyl radical cleavage and denaturing gel electrophoresis, the gel patterns differ. The unfolded RNA molecule is cleaved uniformly, giving rise to a homogeneous ladder of bands on the gel (left, bottom). The gel pattern for the folded RNA (right, bottom), in contrast, shows several region where strand cleavage is inhibited, corresponding to the sites of low solvent accessibility in the folded structure (right, top). TD Tullius, JA Greenbaum, Curr Opin Chem Biol 2005, 9: 127– 134 (Figure 1) Commentary from grove. ufl. edu/~dmorgan/Articles/DER/Journal%20 Club. ppt
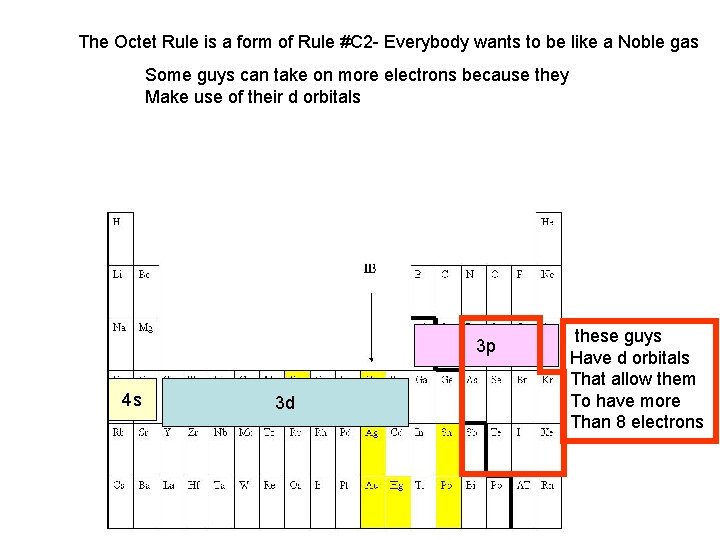
The Octet Rule is a form of Rule #C 2 - Everybody wants to be like a Noble gas Some guys can take on more electrons because they Make use of their d orbitals 3 p 4 s 3 d these guys Have d orbitals That allow them To have more Than 8 electrons
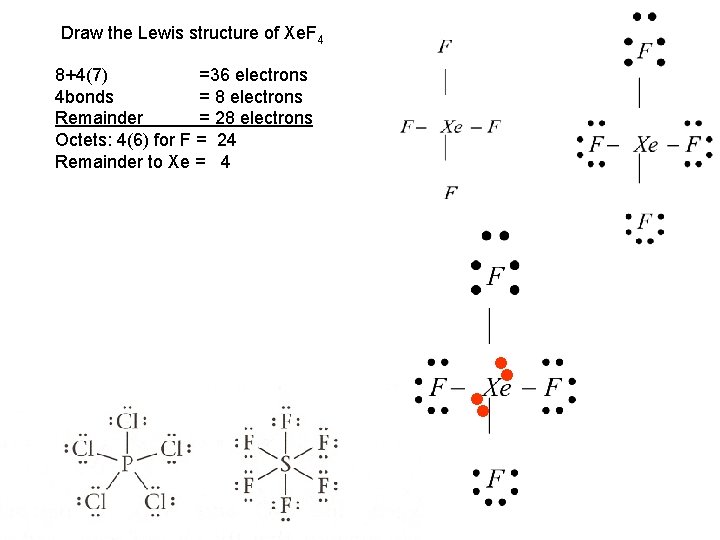
Draw the Lewis structure of Xe. F 4 8+4(7) =36 electrons 4 bonds = 8 electrons Remainder = 28 electrons Octets: 4(6) for F = 24 Remainder to Xe = 4
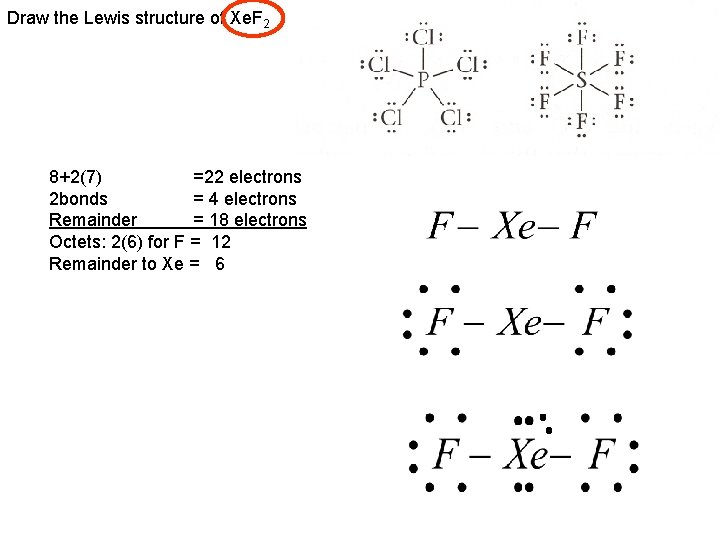
Draw the Lewis structure of Xe. F 2 8+2(7) =22 electrons 2 bonds = 4 electrons Remainder = 18 electrons Octets: 2(6) for F = 12 Remainder to Xe = 6

“A” students work (without solutions manual) ~ 10 problems/night. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours W – F 2 -3 pm

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.
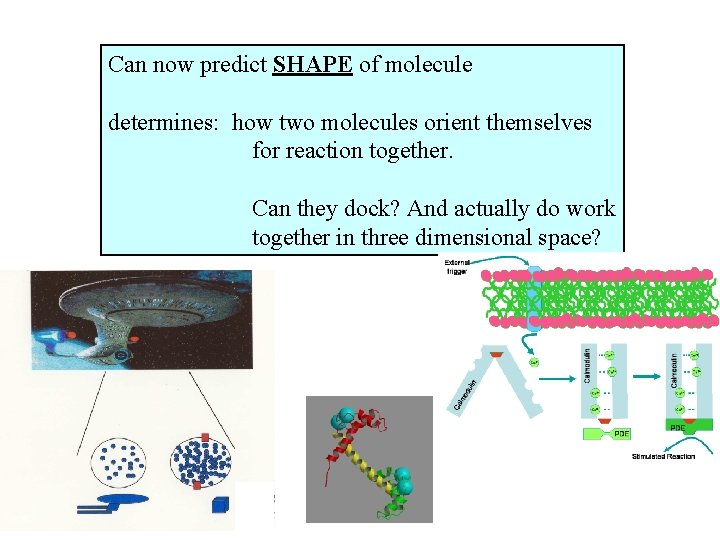
Can now predict SHAPE of molecule determines: how two molecules orient themselves for reaction together. Can they dock? And actually do work together in three dimensional space?
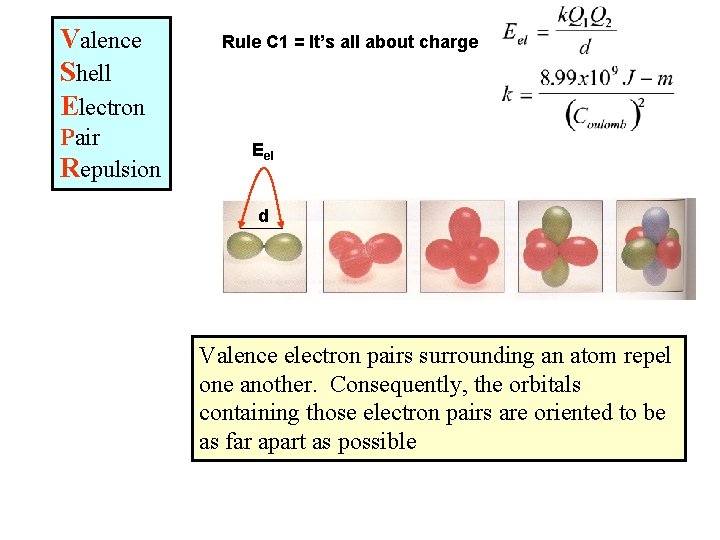
Valence Shell Electron Pair Repulsion Rule C 1 = It’s all about charge Eel d Valence electron pairs surrounding an atom repel one another. Consequently, the orbitals containing those electron pairs are oriented to be as far apart as possible
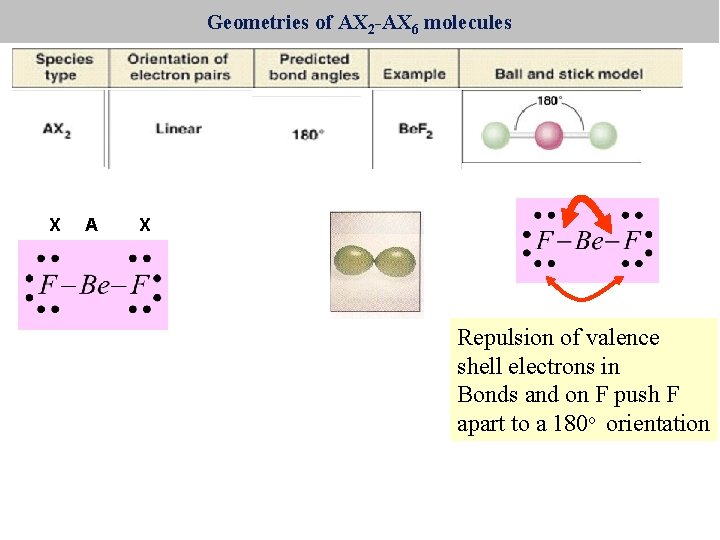
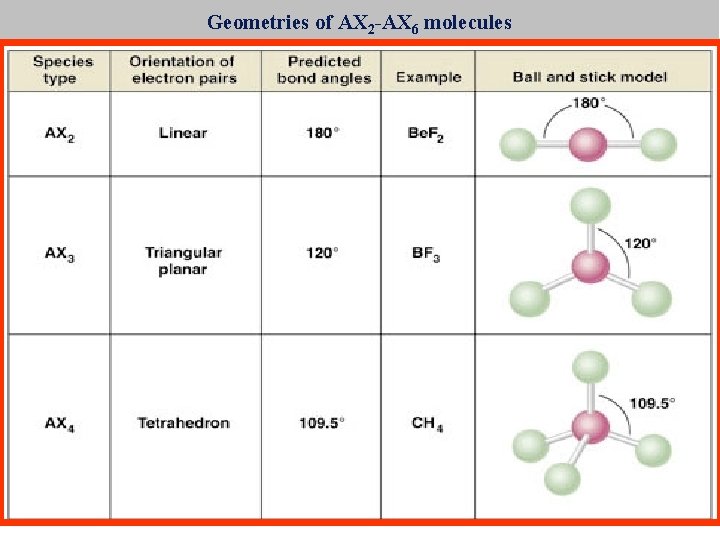
Geometries of AX 2 -AX 6 molecules X A X Repulsion of valence shell electrons in Bonds and on F push F apart to a 180 o orientation
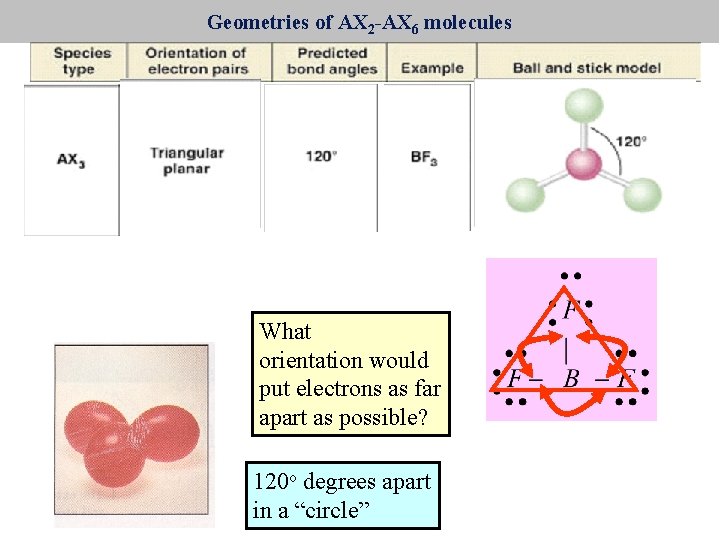
Geometries of AX 2 -AX 6 molecules What orientation would put electrons as far apart as possible? 120 o degrees apart in a “circle”

Draw Lewis structures of a) CH 4 Valence shell electrons? +C +4(H) Negative charge Total electrons -4 single bonds remaining 4 4 0 8 -8 0 Skeleton Carbon is first in formula Hydrogen is always terminal Octets Carbon has its octet Hydrogen has its duet
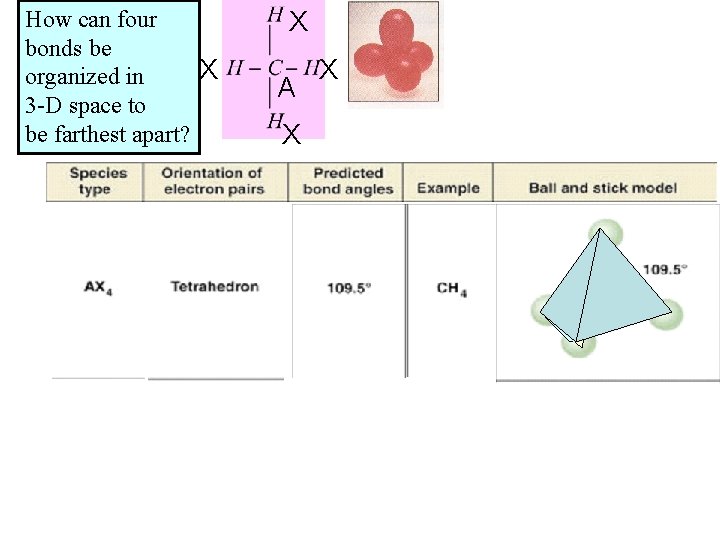
How can four bonds be X organized in 3 -D space to be farthest apart? X A X X
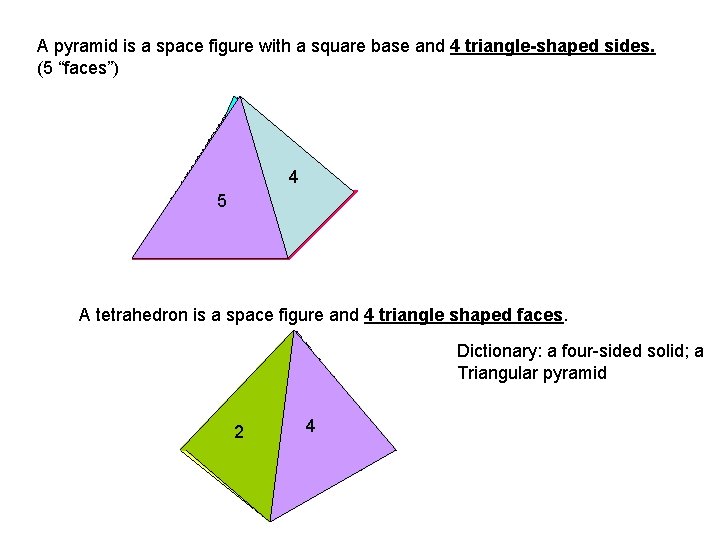
A pyramid is a space figure with a square base and 4 triangle-shaped sides. (5 “faces”) 3 2 5 4 1 A tetrahedron is a space figure and 4 triangle shaped faces. Dictionary: a four-sided solid; a Triangular pyramid 3 2 1 4
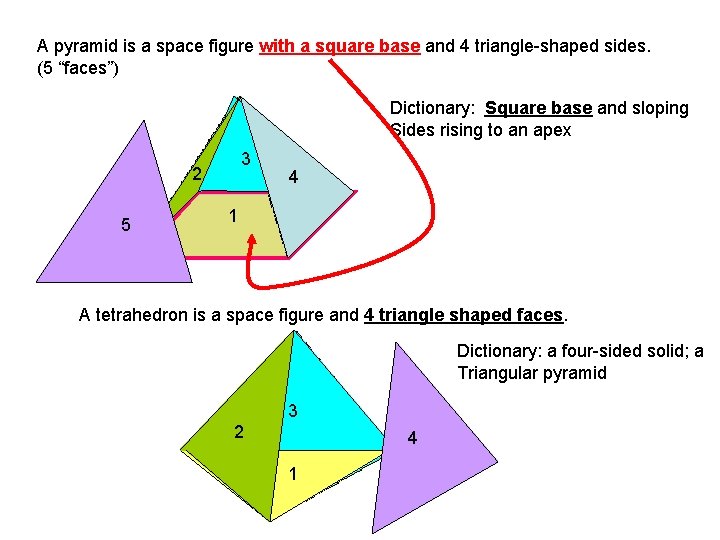
A pyramid is a space figure with a square base and 4 triangle-shaped sides. (5 “faces”) Dictionary: Square base and sloping Sides rising to an apex 3 2 5 4 1 A tetrahedron is a space figure and 4 triangle shaped faces. Dictionary: a four-sided solid; a Triangular pyramid 3 2 4 1
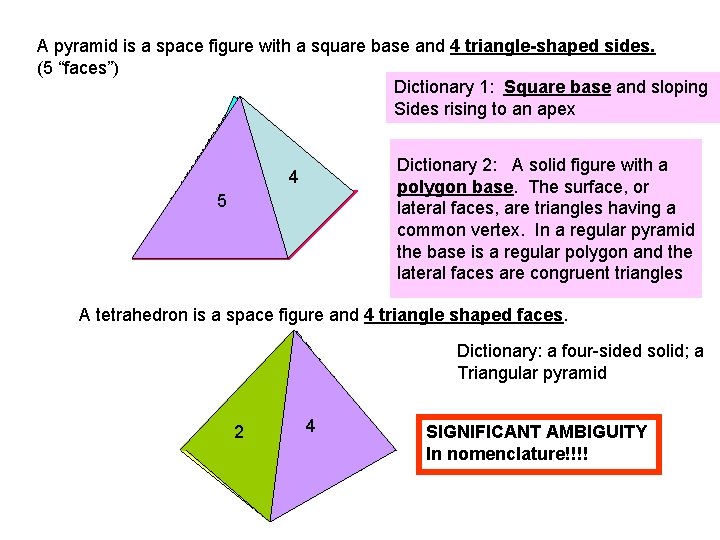
A pyramid is a space figure with a square base and 4 triangle-shaped sides. (5 “faces”) Dictionary 1: Square base and sloping Sides rising to an apex 3 2 5 Dictionary 2: A solid figure with a polygon base. The surface, or lateral faces, are triangles having a common vertex. In a regular pyramid the base is a regular polygon and the lateral faces are congruent triangles 4 1 A tetrahedron is a space figure and 4 triangle shaped faces. Dictionary: a four-sided solid; a Triangular pyramid 3 2 1 4 SIGNIFICANT AMBIGUITY In nomenclature!!!!
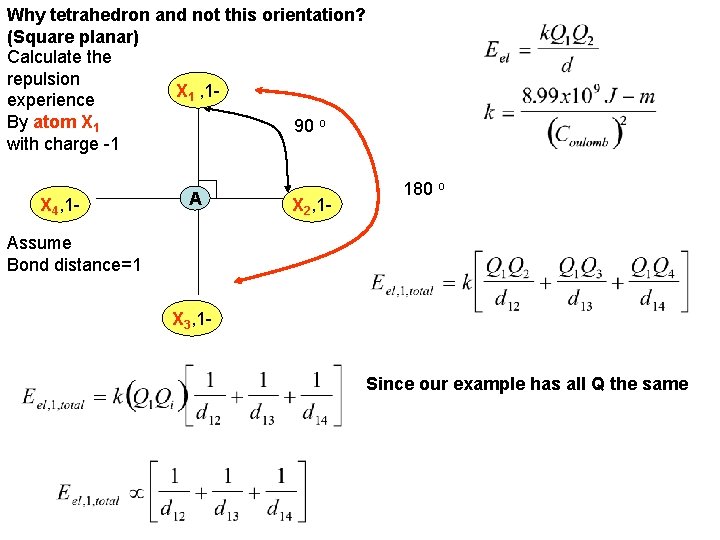
Why tetrahedron and not this orientation? (Square planar) Calculate the repulsion X 1 , 1 experience By atom X 1 90 o with charge -1 X 4, 1 - A X 2, 1 - 180 o Assume Bond distance=1 X 3, 1 Since our example has all Q the same
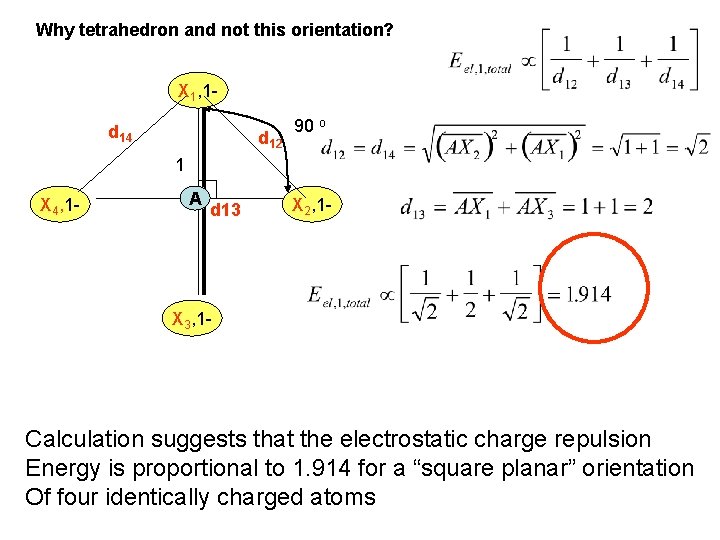
Why tetrahedron and not this orientation? X 1, 1 d 14 d 12 90 o 1 X 4, 1 - A d 13 X 2, 1 - X 3, 1 - Calculation suggests that the electrostatic charge repulsion Energy is proportional to 1. 914 for a “square planar” orientation Of four identically charged atoms
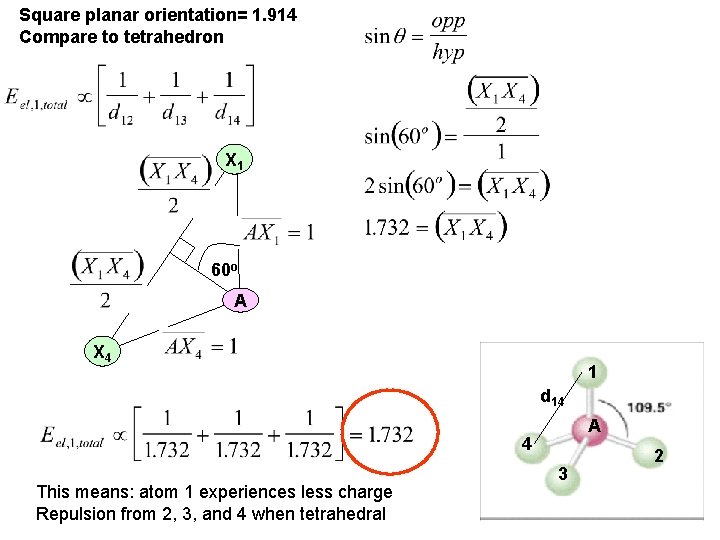
Square planar orientation= 1. 914 Compare to tetrahedron X 1 60 o A X 4 1 d 14 A 4 This means: atom 1 experiences less charge Repulsion from 2, 3, and 4 when tetrahedral 3 2
Geometries of AX 2 -AX 6 molecules
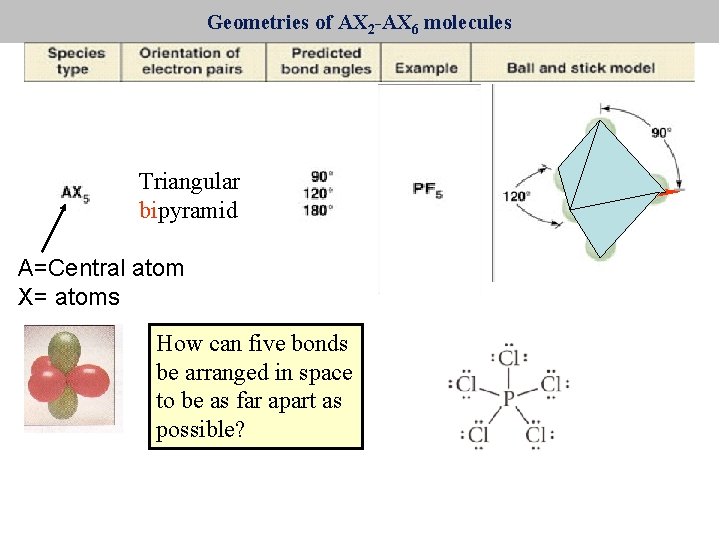
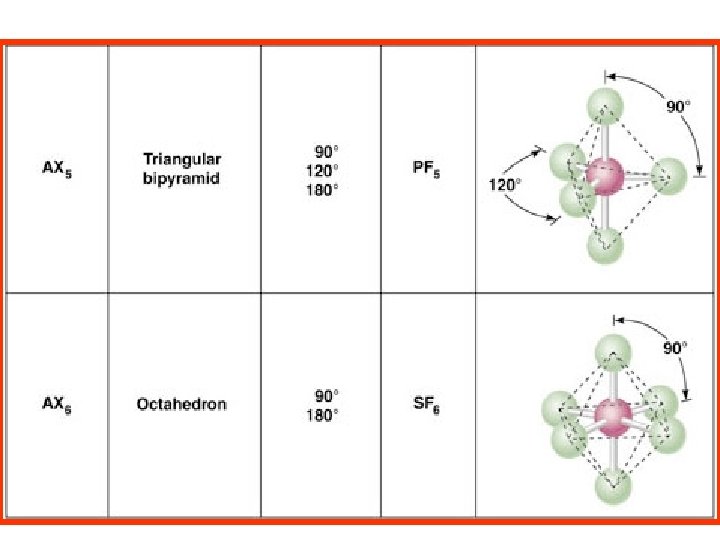
Geometries of AX 2 -AX 6 molecules Triangular pyramid bipyramid A=Central atom X= atoms How can five bonds be arranged in space to be as far apart as possible?
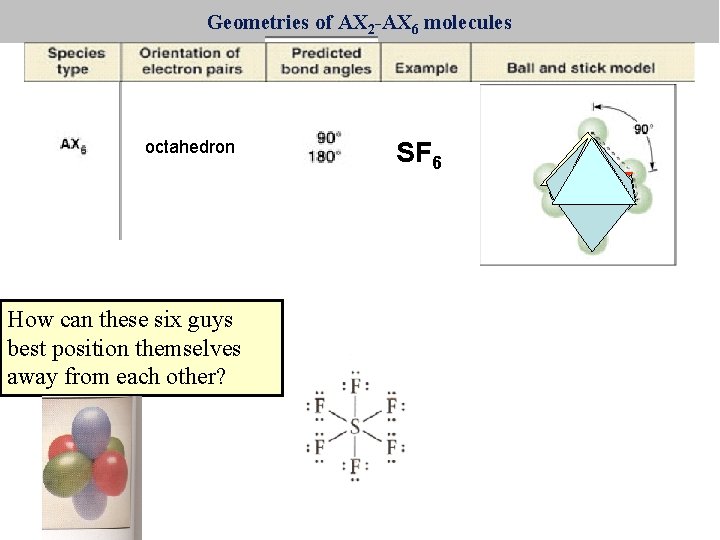
Geometries of AX 2 -AX 6 molecules octahedron How can these six guys best position themselves away from each other? SF 6

Some of the molecules we constructed using Lewis Dot structures had UNSHARED PAIRS of electrons on the CENTRAL ATOM What effect will this have on the geometry? . AX 2 E Unshared electron pairs orient themselves pretty much the same as single bonds. The observed molecular geometry (invisible electrons) is very different
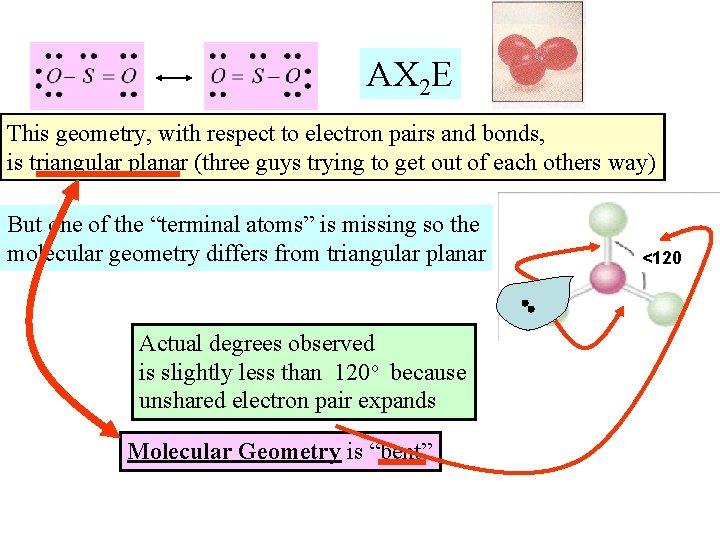
AX 2 E This geometry, with respect to electron pairs and bonds, is triangular planar (three guys trying to get out of each others way) But one of the “terminal atoms” is missing so the molecular geometry differs from triangular planar Actual degrees observed is slightly less than 120 o because unshared electron pair expands Molecular Geometry is “bent” <120
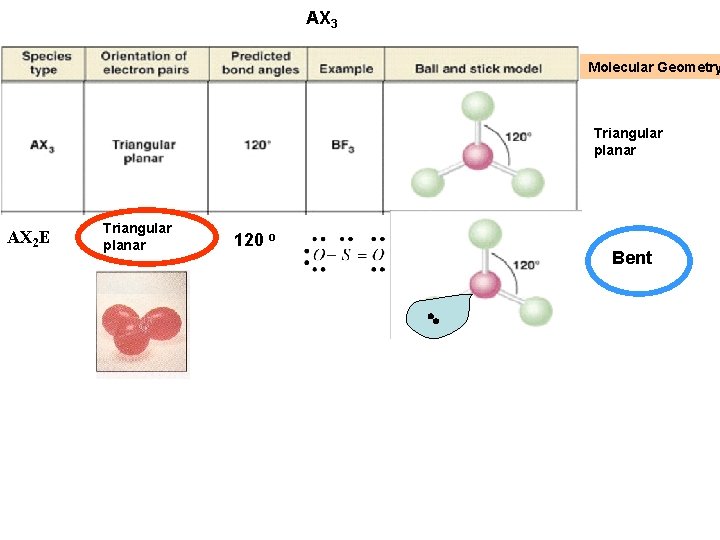
AX 3 Molecular Geometry Triangular planar AX 2 E Triangular planar 120 o Bent
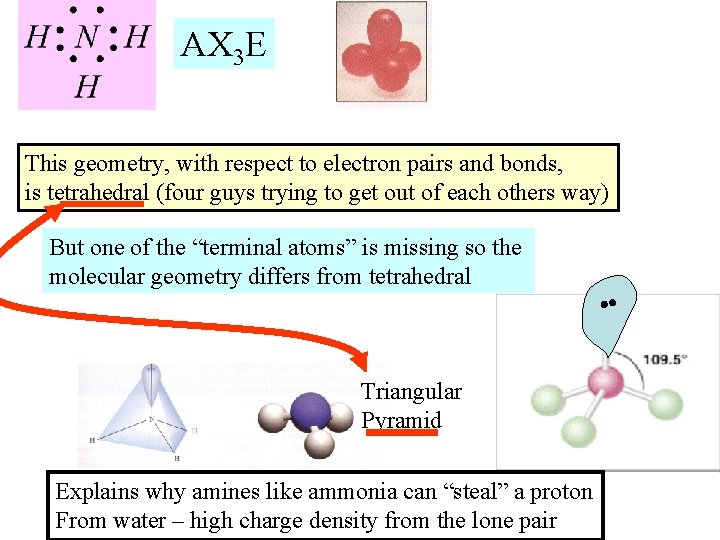
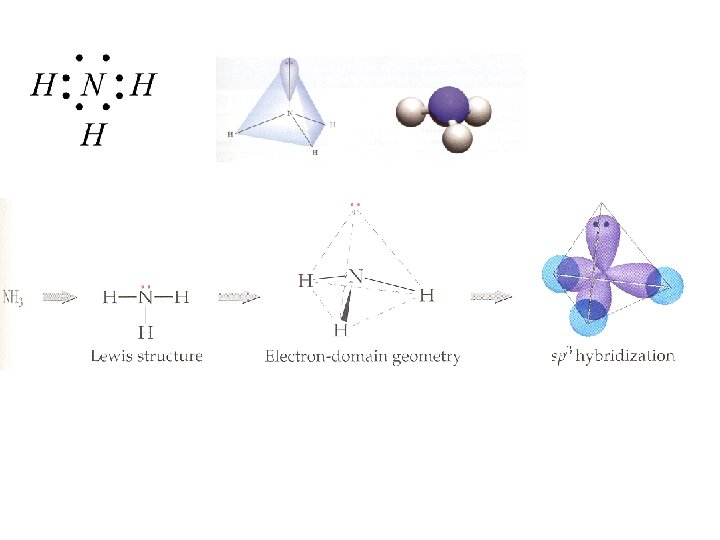
AX 3 E This geometry, with respect to electron pairs and bonds, is tetrahedral (four guys trying to get out of each others way) But one of the “terminal atoms” is missing so the molecular geometry differs from tetrahedral Triangular Pyramid Explains why amines like ammonia can “steal” a proton From water – high charge density from the lone pair
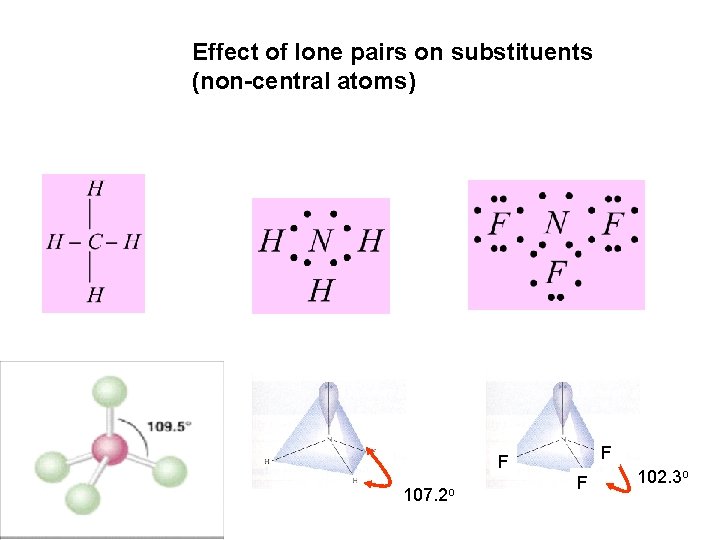
Effect of lone pairs on substituents (non-central atoms) F F 107. 2 o F 102. 3 o
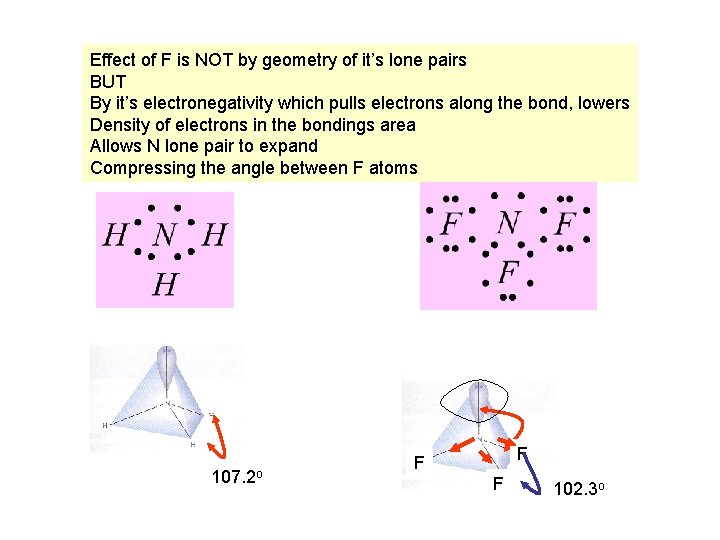
Effect of F is NOT by geometry of it’s lone pairs BUT By it’s electronegativity which pulls electrons along the bond, lowers Density of electrons in the bondings area Allows N lone pair to expand Compressing the angle between F atoms 107. 2 o F F F 102. 3 o
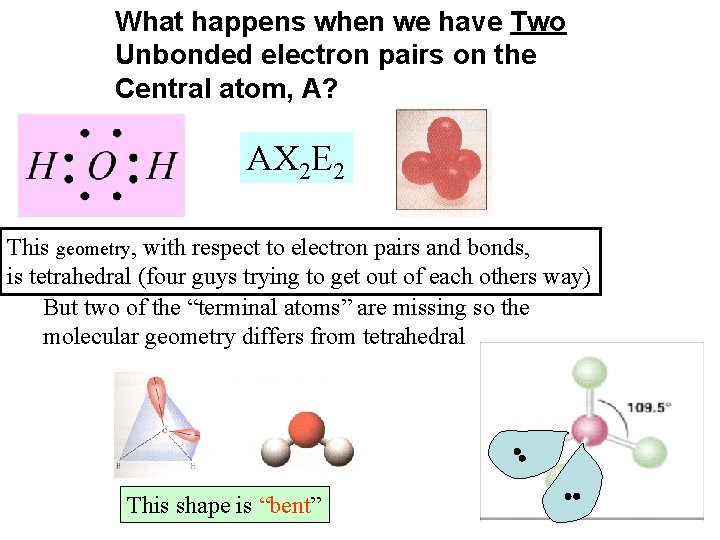
What happens when we have Two Unbonded electron pairs on the Central atom, A? AX 2 E 2 This geometry, with respect to electron pairs and bonds, is tetrahedral (four guys trying to get out of each others way) But two of the “terminal atoms” are missing so the molecular geometry differs from tetrahedral This shape is “bent”
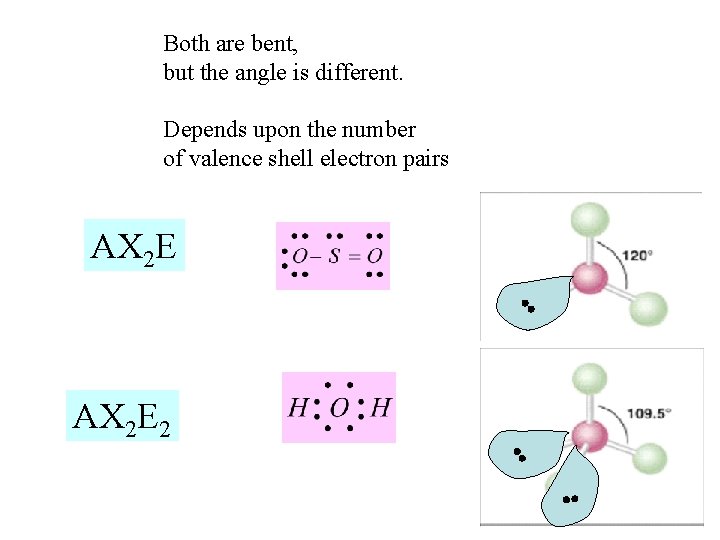
Both are bent, but the angle is different. Depends upon the number of valence shell electron pairs AX 2 E 2
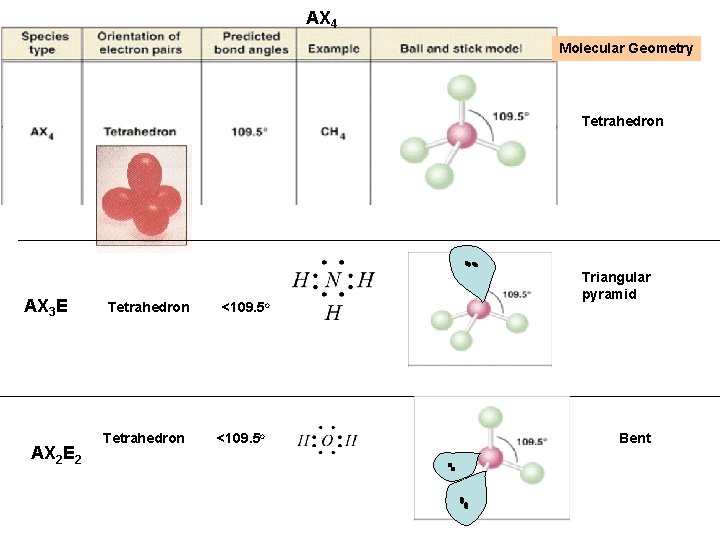
AX 4 Molecular Geometry Tetrahedron AX 3 E AX 2 E 2 Tetrahedron <109. 5 o Triangular pyramid Bent
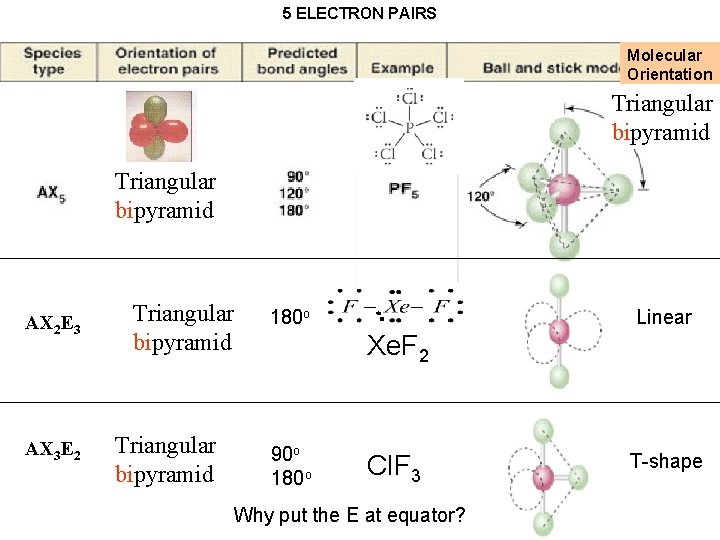
5 ELECTRON PAIRS Molecular Orientation Triangular bipyramid Triangular pyramid bipyramid AX 2 E 3 AX 3 E 2 Triangular bipyramid 180 o Linear Xe. F 2 90 o 180 o Cl. F 3 Why put the E at equator? T-shape
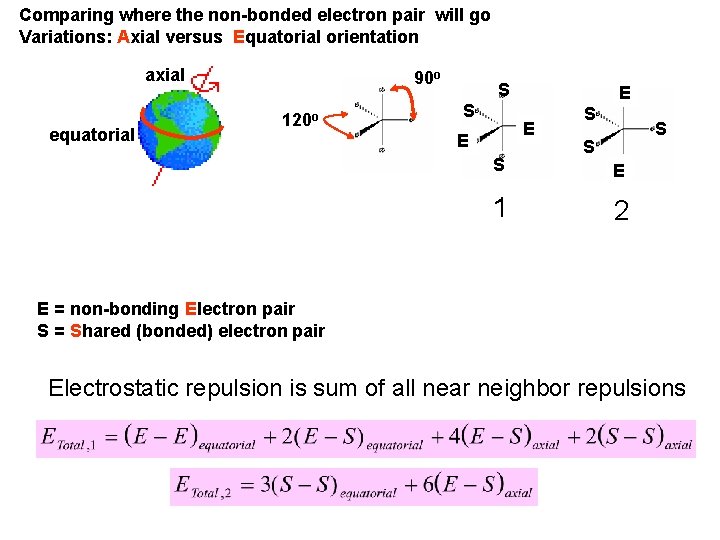
Comparing where the non-bonded electron pair will go Variations: Axial versus Equatorial orientation axial equatorial 90 o 120 o S S E E E S 1 S S S E 2 E = non-bonding Electron pair S = Shared (bonded) electron pair Electrostatic repulsion is sum of all near neighbor repulsions
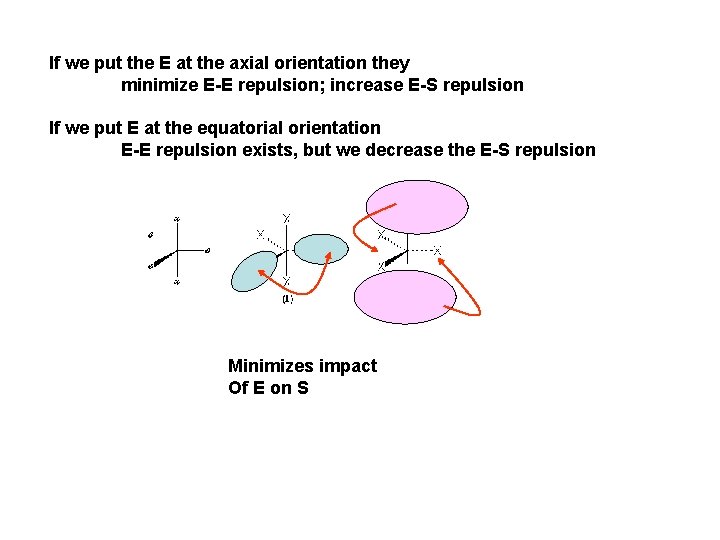
If we put the E at the axial orientation they minimize E-E repulsion; increase E-S repulsion If we put E at the equatorial orientation E-E repulsion exists, but we decrease the E-S repulsion E Minimizes impact Of E on S E
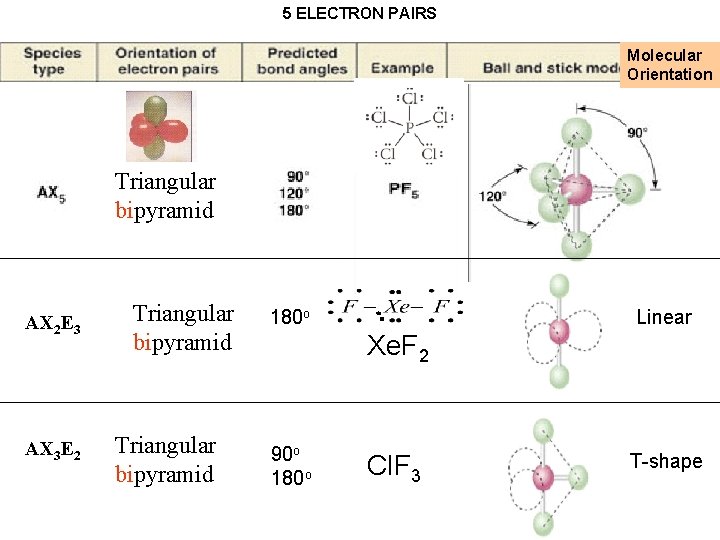
5 ELECTRON PAIRS Molecular Orientation Triangular pyramid bipyramid AX 2 E 3 AX 3 E 2 Triangular bipyramid 180 o Linear Xe. F 2 90 o 180 o Cl. F 3 T-shape
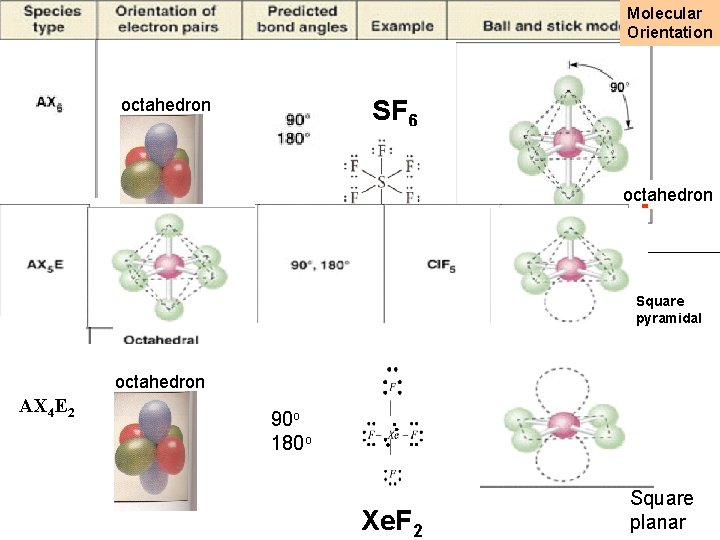
Molecular Orientation SF 6 octahedron Square pyramidal octahedron AX 4 E 2 90 o 180 o Xe. F 2 Square planar
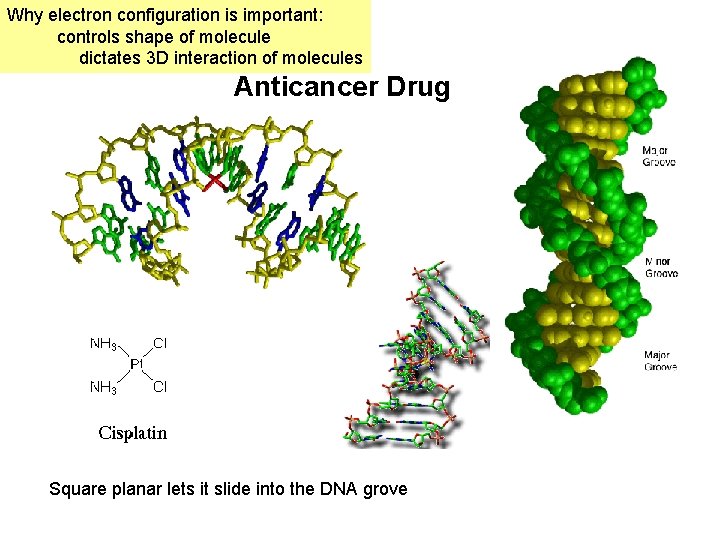
Why electron configuration is important: controls shape of molecule dictates 3 D interaction of molecules Anticancer Drug Square planar lets it slide into the DNA grove
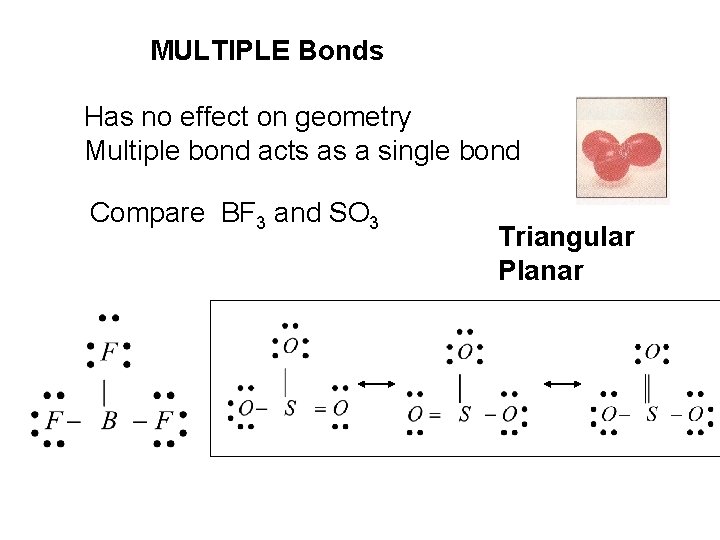
MULTIPLE Bonds Has no effect on geometry Multiple bond acts as a single bond Compare BF 3 and SO 3 Triangular Planar

Compare Molecular Geometries for Be. F 2 and CO 2 1. We already did Be. F 2
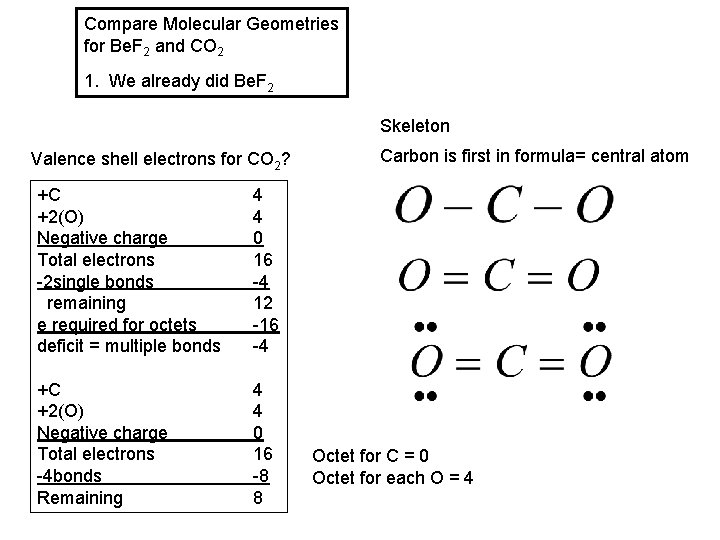
Compare Molecular Geometries for Be. F 2 and CO 2 1. We already did Be. F 2 Skeleton Valence shell electrons for CO 2? +C +2(O) Negative charge Total electrons -2 single bonds remaining e required for octets deficit = multiple bonds 4 4 0 16 -4 12 -16 -4 +C +2(O) Negative charge Total electrons -4 bonds Remaining 4 4 0 16 -8 8 Carbon is first in formula= central atom Octet for C = 0 Octet for each O = 4
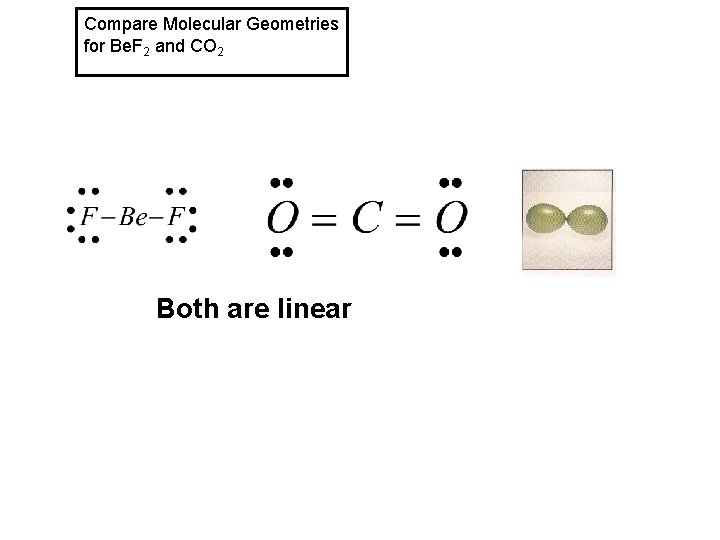
Compare Molecular Geometries for Be. F 2 and CO 2 Both are linear
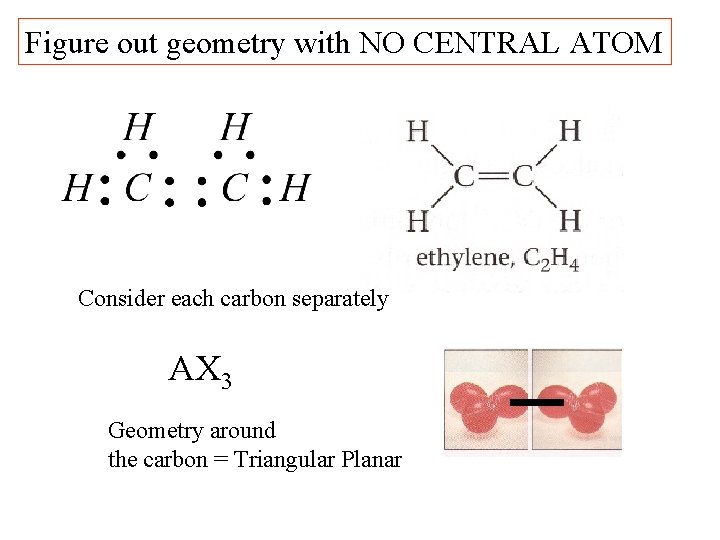
Figure out geometry with NO CENTRAL ATOM Consider each carbon separately AX 3 Geometry around the carbon = Triangular Planar

Figure out geometry with NO CENTRAL ATOM Consider each carbon separately AX 2 Geometry around the carbon =Linear
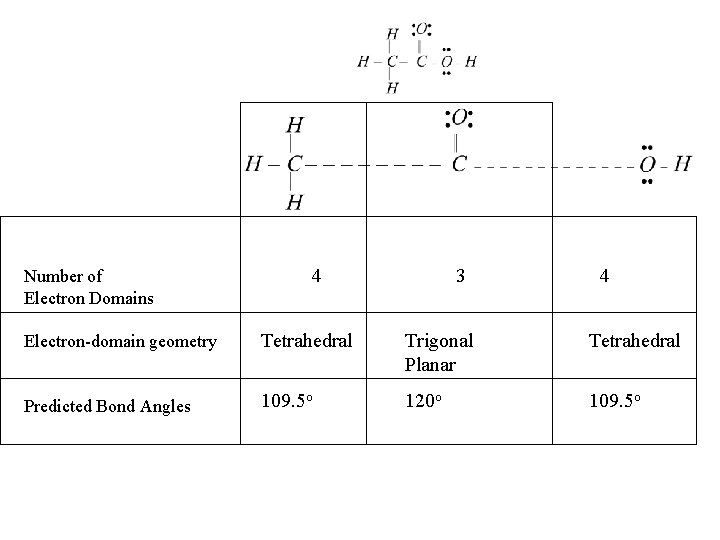
Number of Electron Domains 4 3 4 Electron-domain geometry Tetrahedral Trigonal Planar Tetrahedral Predicted Bond Angles 109. 5 o 120 o 109. 5 o

“A” students work (without solutions manual) ~ 10 problems/night. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours W – F 2 -3 pm

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.

Bond Polarity – distribution of electrons in the bond 1. Depends upon the difference in electronegativity of bonded atoms 2. If two atoms in the bond are identical =nonpolar 3. Otherwise all bonds are polar
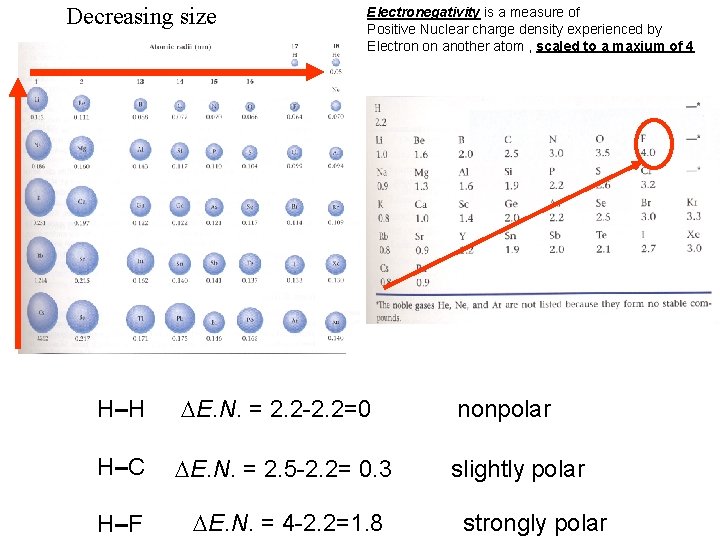
Decreasing size Electronegativity is a measure of Positive Nuclear charge density experienced by Electron on another atom , scaled to a maxium of 4 H–H E. N. = 2. 2 -2. 2=0 nonpolar H–C E. N. = 2. 5 -2. 2= 0. 3 slightly polar H–F E. N. = 4 -2. 2=1. 8 strongly polar
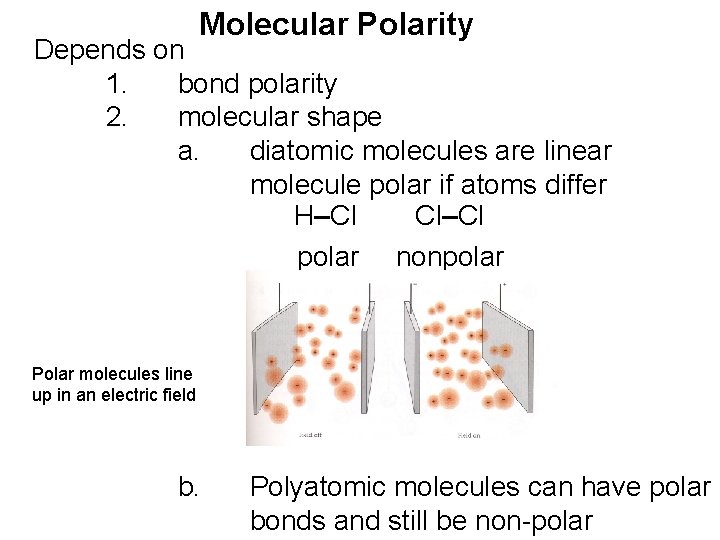
Molecular Polarity Depends on 1. bond polarity 2. molecular shape a. diatomic molecules are linear molecule polar if atoms differ H–Cl Cl–Cl polar nonpolar Polar molecules line up in an electric field b. Polyatomic molecules can have polar bonds and still be non-polar
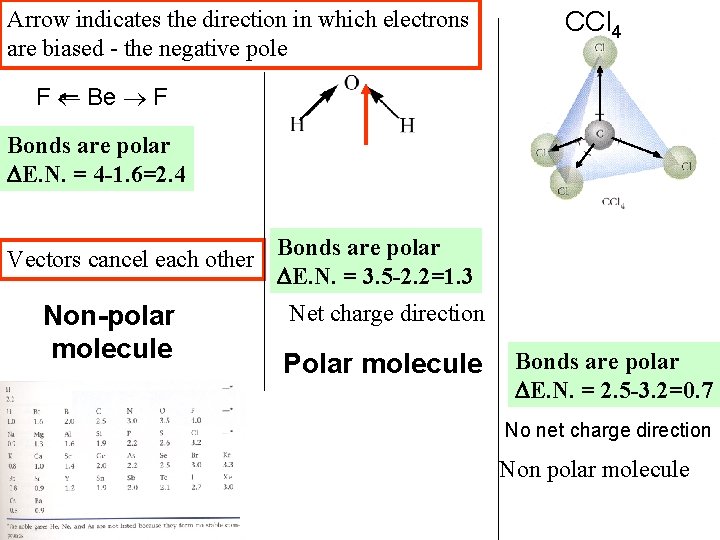
Arrow indicates the direction in which electrons are biased - the negative pole CCl 4 F Be F Bonds are polar E. N. = 4 -1. 6=2. 4 Vectors cancel each other Bonds are polar E. N. = 3. 5 -2. 2=1. 3 Non-polar molecule Net charge direction Polar molecule Bonds are polar E. N. = 2. 5 -3. 2=0. 7 No net charge direction Non polar molecule
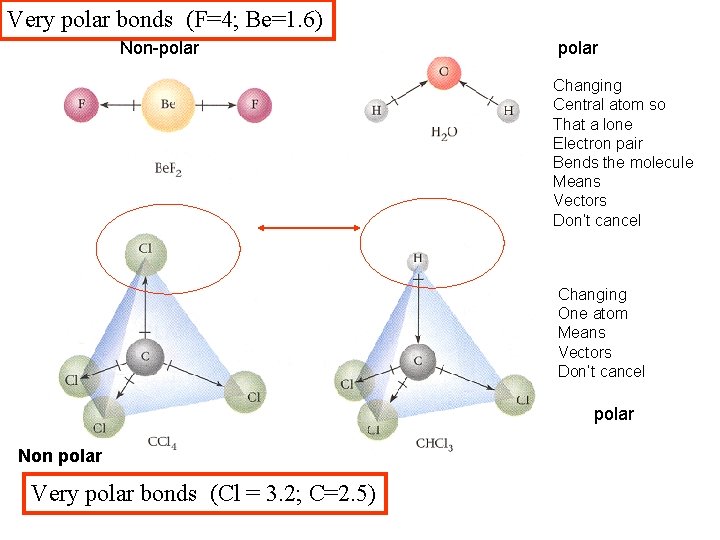
Very polar bonds (F=4; Be=1. 6) Non-polar Changing Central atom so That a lone Electron pair Bends the molecule Means Vectors Don’t cancel Changing One atom Means Vectors Don’t cancel polar Non polar Very polar bonds (Cl = 3. 2; C=2. 5)

“A” students work (without solutions manual) ~ 10 problems/night. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours W – F 2 -3 pm

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.

The “fly in the ointment” VSEPR AX 2 Linear Repulsion of valence shell electrons pushes F apart to a 180 o orientation VSEPR model suggests that once Be bonds to F the orbitals are “equivalent” and therefore are equidistant from each other. But the electron orbital diagram suggests otherwise that Be has paired electrons and would not make bonds at all.
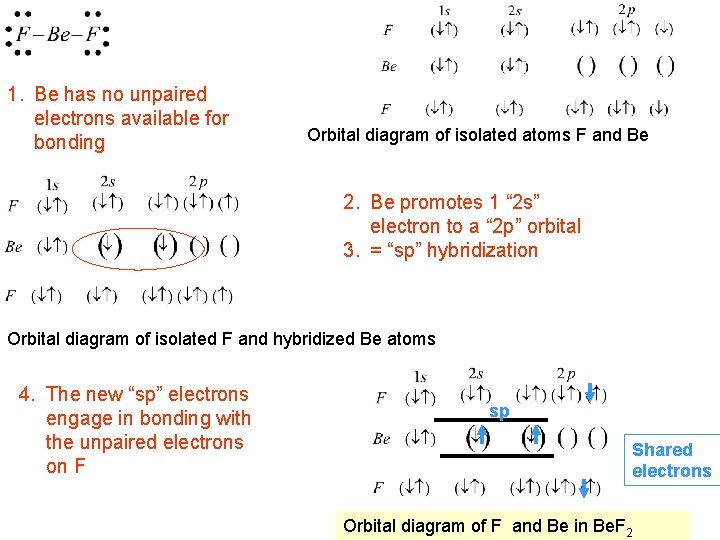
1. Be has no unpaired electrons available for bonding Orbital diagram of isolated atoms F and Be 2. Be promotes 1 “ 2 s” electron to a “ 2 p” orbital 3. = “sp” hybridization Orbital diagram of isolated F and hybridized Be atoms 4. The new “sp” electrons engage in bonding with the unpaired electrons on F sp Shared electrons Orbital diagram of F and Be in Be. F 2
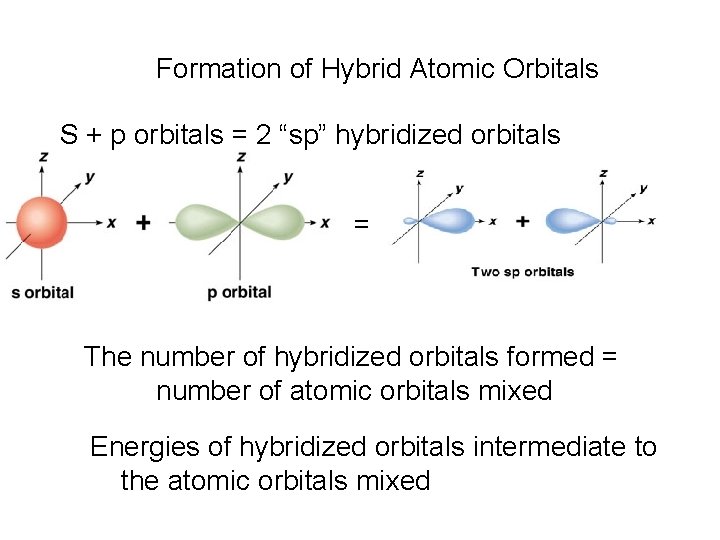
Formation of Hybrid Atomic Orbitals S + p orbitals = 2 “sp” hybridized orbitals = The number of hybridized orbitals formed = number of atomic orbitals mixed Energies of hybridized orbitals intermediate to the atomic orbitals mixed
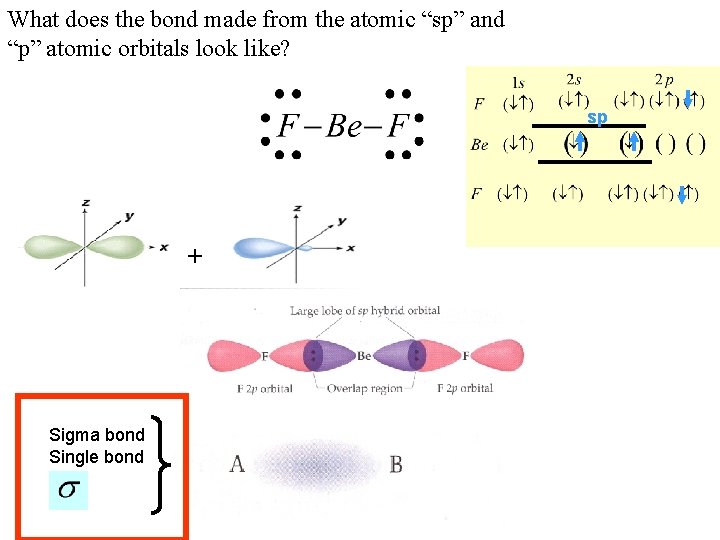
What does the bond made from the atomic “sp” and “p” atomic orbitals look like? sp + Sigma bond Single bond
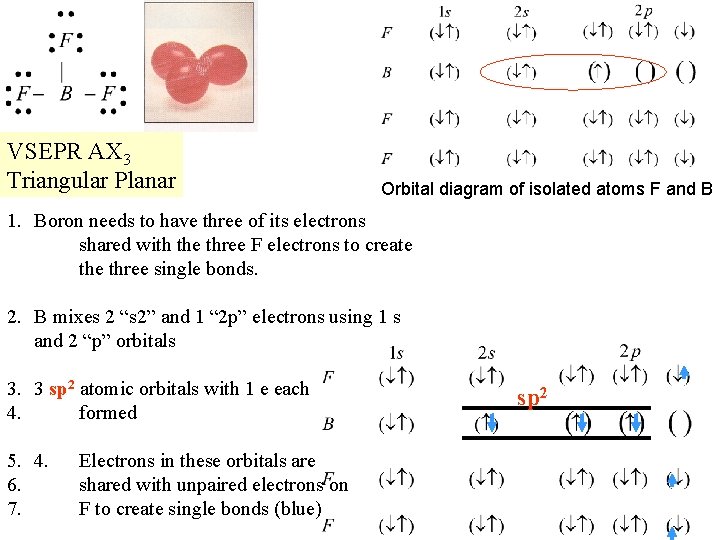
VSEPR AX 3 Triangular Planar Orbital diagram of isolated atoms F and B 1. Boron needs to have three of its electrons shared with the three F electrons to create three single bonds. 2. B mixes 2 “s 2” and 1 “ 2 p” electrons using 1 s and 2 “p” orbitals 3. 3 sp 2 atomic orbitals with 1 e each 4. formed 5. 4. 6. 7. Electrons in these orbitals are shared with unpaired electrons on F to create single bonds (blue) sp 2
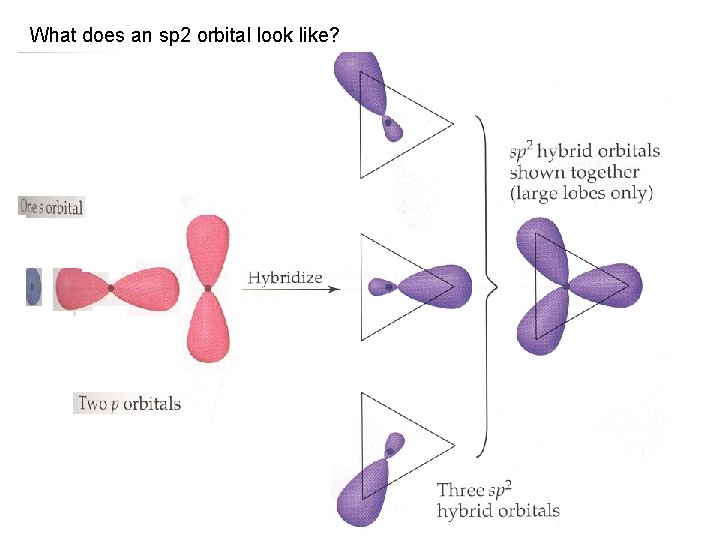
What does an sp 2 orbital look like?

VSEPR AX 4 Tetrahedral 1. C needs four energetically equivalent bonds Orbital diagram of isolated atoms H and C 2. C mixes 1 “ 2 s” and 3 “ 2 p” atomic orbitals 3. 4 sp 3 atomic orbitals formed 4. sp 3 orbitals used to create bonds sp 3
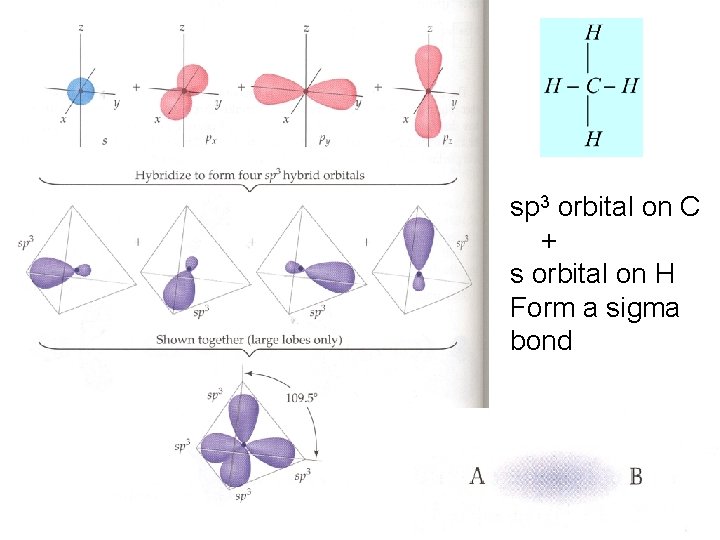
sp 3 orbital on C + s orbital on H Form a sigma bond
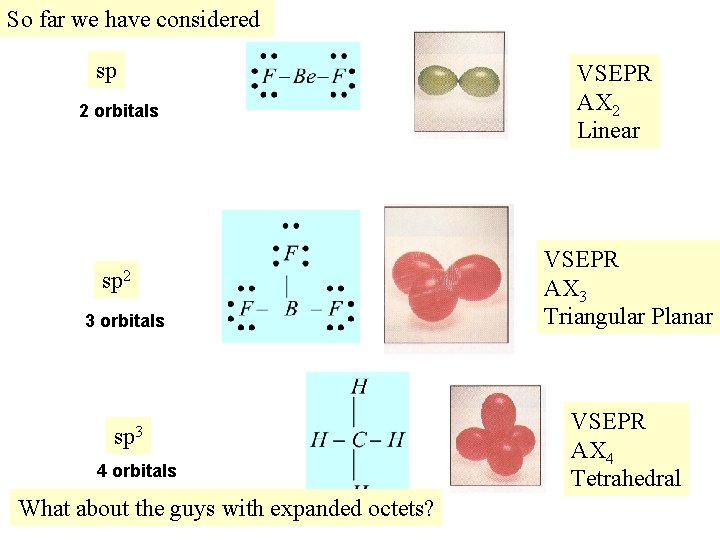
So far we have considered sp 2 orbitals sp 2 3 orbitals sp 3 4 orbitals What about the guys with expanded octets? VSEPR AX 2 Linear VSEPR AX 3 Triangular Planar VSEPR AX 4 Tetrahedral
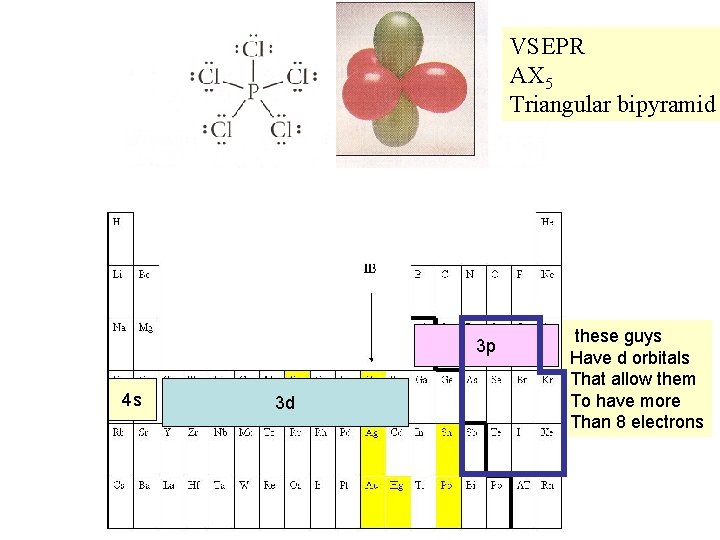
VSEPR AX 5 Triangular bipyramid 3 p 4 s 3 d these guys Have d orbitals That allow them To have more Than 8 electrons
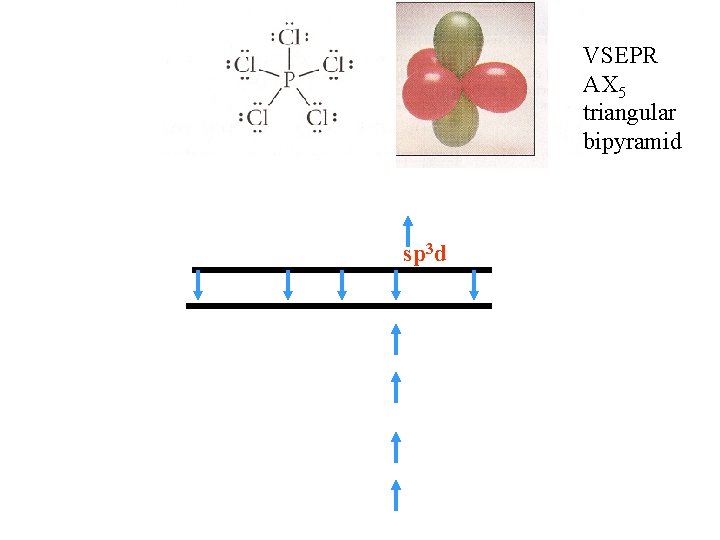
VSEPR AX 5 triangular bipyramid sp 3 d

VSEPR AX 2 AX 3 AX 4 AX 5 AX 6 Nothing new here – same as we got with VSEPR Note to myself from 2006 – F does not hybridize – has one unpaired electron Forming one bond
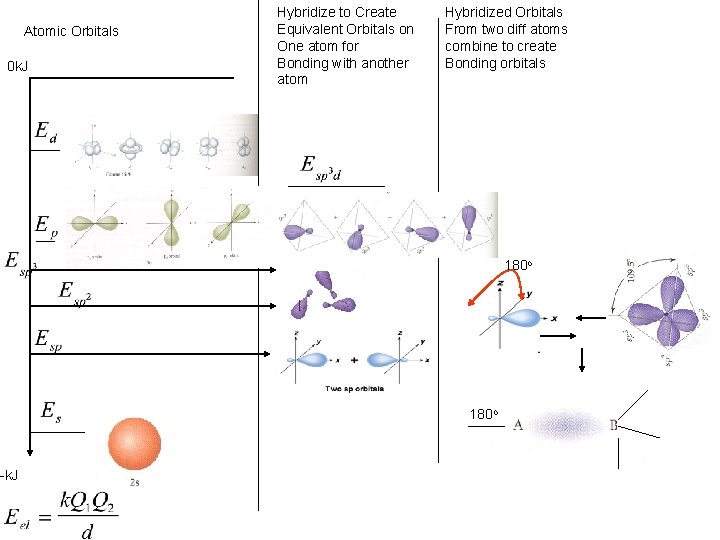
Atomic Orbitals 0 k. J -k. J Hybridize to Create Equivalent Orbitals on One atom for Bonding with another atom Hybridized Orbitals From two diff atoms combine to create Bonding orbitals 180 o

Double and Triple Bonds Whenever we have a “single” bond we can assume that it has the sigma shape, resulting from hybridization between atomic orbitals Sigma bond Single bond For double and triple bonds, we do not need to create more equivalent bonds which can be moved as far apart as predicted by Valence Shell Electron Pair Repulsion. We need to simply create additional bonds within the shape predicted by VSEPR Pi bond Double bond around single bond
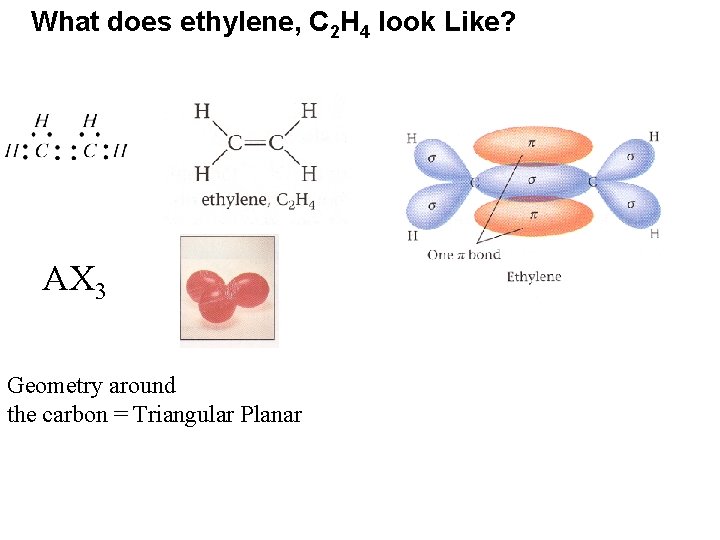
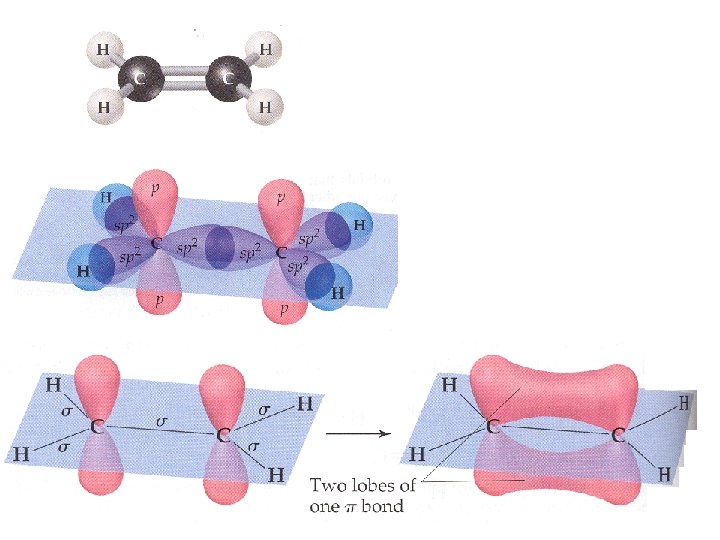
What does ethylene, C 2 H 4 look Like? AX 3 Geometry around the carbon = Triangular Planar
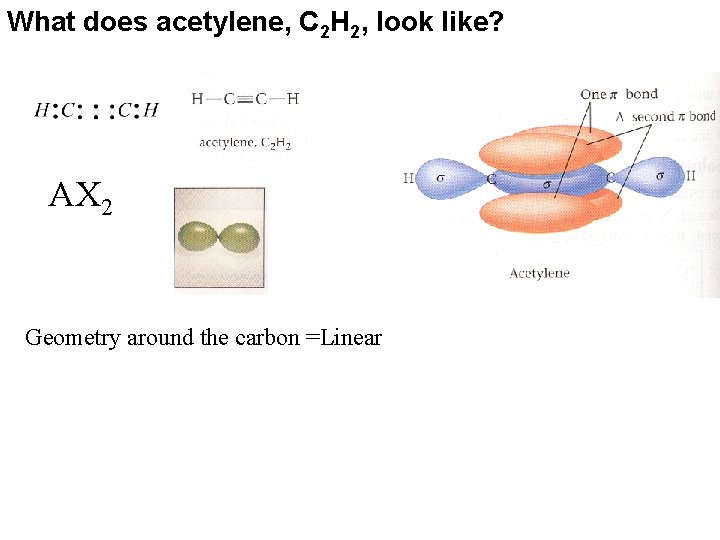
What does acetylene, C 2 H 2, look like? AX 2 Geometry around the carbon =Linear

Example problem: What is the hybridization of nitrogen in a) NO 3 -, b) NH 2 Cl, c) N 2, d) and N 2 O? 1. Make the Lewis dot structure 2. Count sigma bonds on central atom 3. Count the unbonded electron pairs on central atom 4. Sum the 2 (n) 5. Number of hybridized orbitals =n Cmpd σ Bonds Unbonded electron pairs on on N N n Hybridization on N NO 33 -- 3 0 3 sp 2 NH 2 Cl 3 1 4 sp 3 N 2 1 1 2 sp N 2 O a 1 1 2 sp N 2 O b 2 0 2 sp

RESONANCE
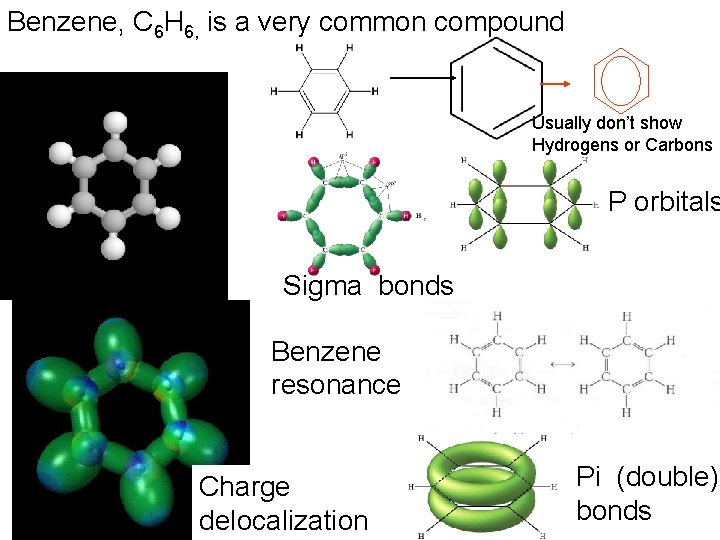
Benzene, C 6 H 6, is a very common compound Usually don’t show Hydrogens or Carbons P orbitals Sigma bonds Benzene resonance Charge delocalization Pi (double) bonds
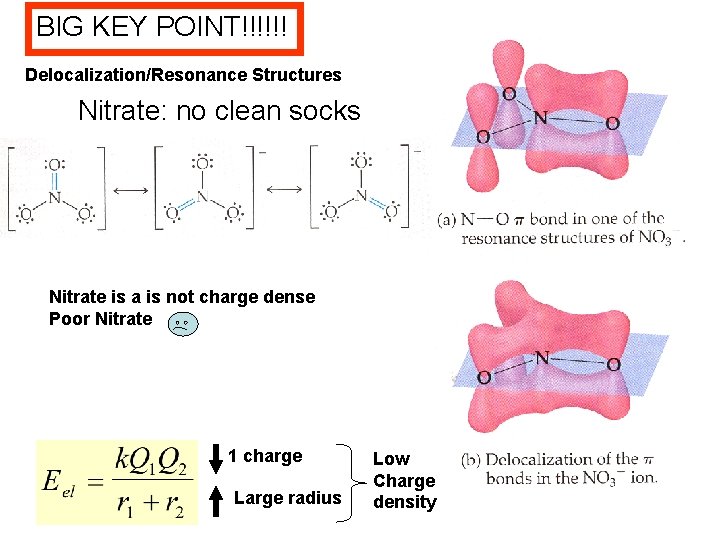
BIG KEY POINT!!!!!! Delocalization/Resonance Structures Nitrate: no clean socks Nitrate is a is not charge dense Poor Nitrate 1 charge Large radius Low Charge density

1. Bonding = sharing –electrons between repulsive + nuclei 2. Lewis Dot structures help us visualize sharing of electrons Octets Double and triple bonds Resonance structures and No Clean Socks 3. Formal Charge to help distinguish between alternatives 4. Violations of the Octet Rule 2 electrons >8 electrons 5. Using electrons to predict the SHAPE of the molecules VESPR Effect of unpaired electrons on the central atom on molecular shape Effect of Multiple bonds How to deal with “no central atom” 6. Bond polarity Effect of electronegativity difference between atoms in bond Effect of molecular shape How to symbolize bond polarity 7. Discrepancies between Electron Orbital Diagrams and VESPR Introducing orbital hybridization, s, sp 2, sp 3 Using orbital hybridization to visualize resonance or smearing out of electron density in the molecule.

“A” students work (without solutions manual) ~ 10 problems/night. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours W – F 2 -3 pm
- Slides: 113