A students work without solutions manual 10 problemsnight

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Effect of number of Possible configurations (randomness) on reactions

What we’ve Learned So Far Energy Ea Ea endothermic exothermic A+B C + heat A + B + heat C Does not tell us if spontaneous or not
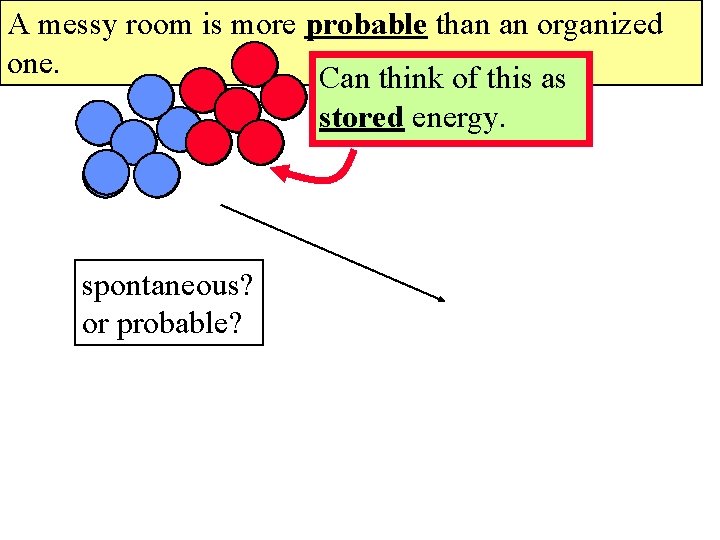
A messy room is more probable than an organized one. Can think of this as stored energy. spontaneous? or probable?
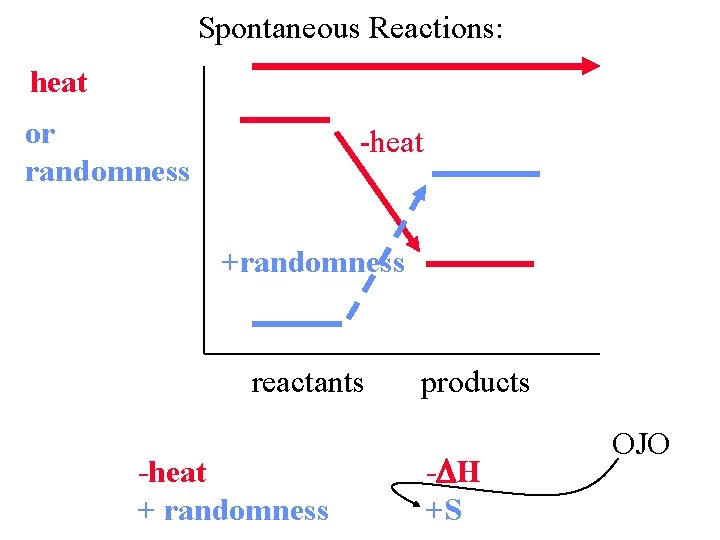
Spontaneous Reactions: heat or randomness -heat +randomness reactants -heat + randomness products - H +S OJO

Let’s consider randomness Each system will change toward a Condition of maximum probability a) Most probable state of 1 quarter Of 2 quarters 3 quarters 4 quarters More probable state, The greater entropy

Randomness = more possibilities = entropy (S) 1 coin =2 sides 2 coins 1 Equal probability In each state 2 configurations =21 2 Most probable 1 4 configurations =22

What will the pattern be for three coins? 1 3 3 coins 8 configurations = 23 4 coins ? configurations = 2? 3 1 Most probable Point: of configurations increases exponentially With #coins (units) (n) 2 points – most probable Log relationship of poss 3 rd point will be explore #configurations contains Information about organization of The system (chemical) (how many “sides”; how many “chemical units” with those sides)

How does randomness associated with a specific Chemical unit (atom or molecule) in different phases Solid Liquid gas
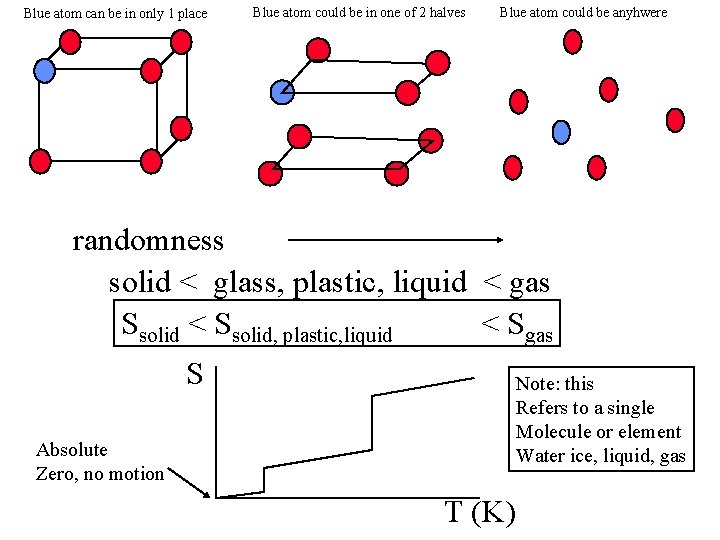
Blue atom can be in only 1 place Blue atom could be in one of 2 halves Blue atom could be anyhwere randomness solid < glass, plastic, liquid < gas Ssolid < Ssolid, plastic, liquid < Sgas S Note: this Refers to a single Molecule or element Water ice, liquid, gas Absolute Zero, no motion T (K)

What about randomness within a chemical unit Randomness associated with a chemical unit 1. related to number of subunits protons, nuetrons, electrons in an atoms within a molecule 2. related to the organization of those
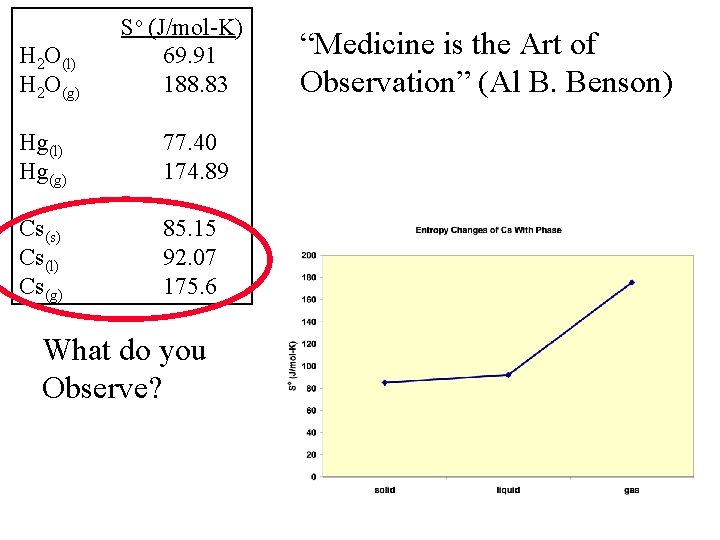
H 2 O(l) H 2 O(g) So (J/mol-K) 69. 91 188. 83 Hg(l) Hg(g) 77. 40 174. 89 Cs(s) Cs(l) Cs(g) 85. 15 92. 07 175. 6 What do you Observe? “Medicine is the Art of Observation” (Al B. Benson)
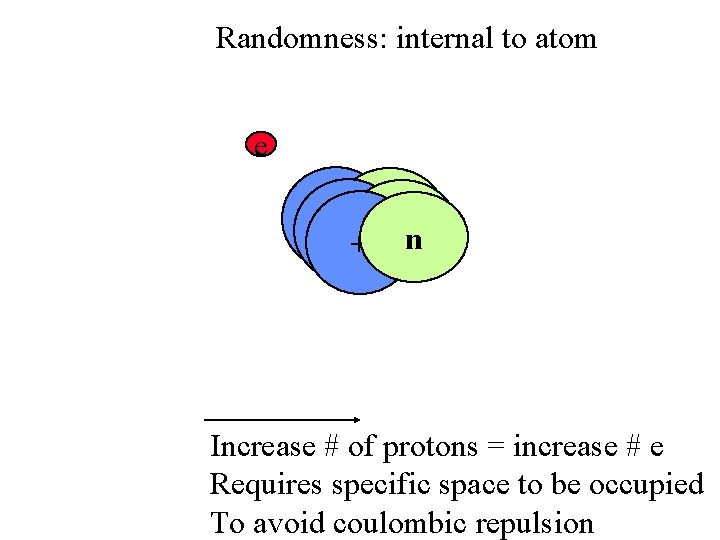
Randomness: internal to atom e ++ nn + n Increase # of protons = increase # e Requires specific space to be occupied To avoid coulombic repulsion
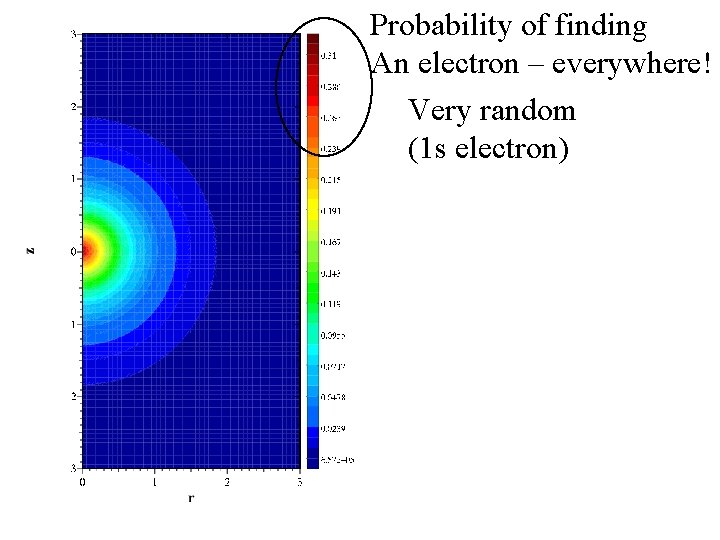
Probability of finding An electron – everywhere! Very random (1 s electron)
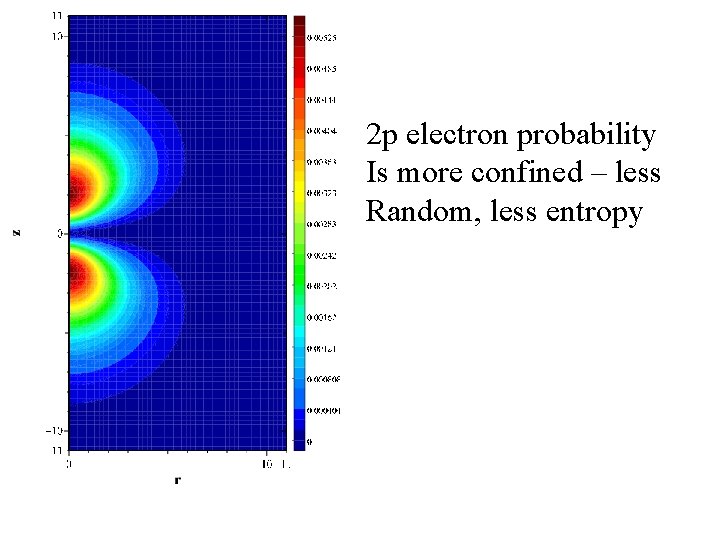
2 p electron probability Is more confined – less Random, less entropy
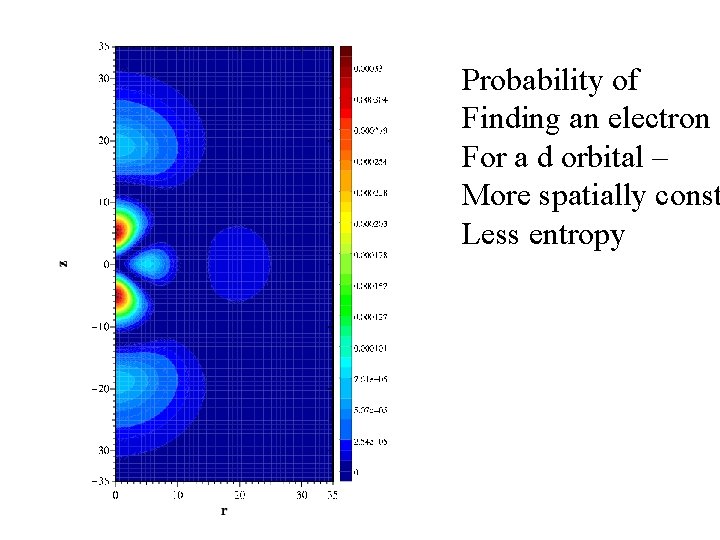
Probability of Finding an electron For a d orbital – More spatially const Less entropy
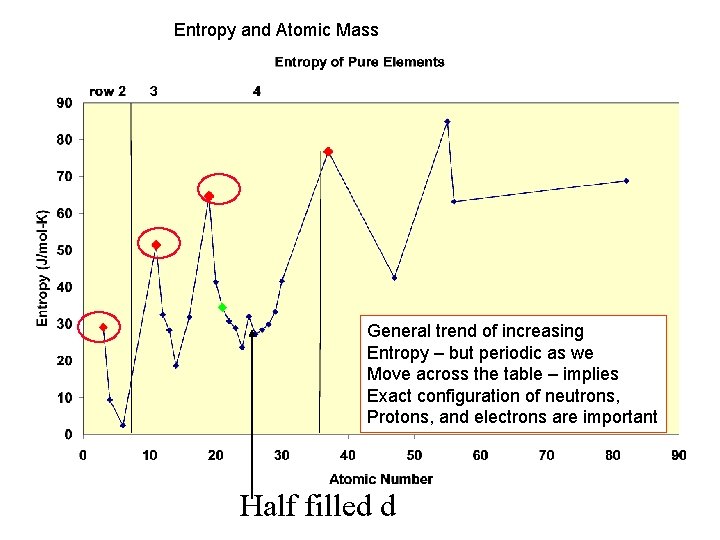
Entropy and Atomic Mass General trend of increasing Entropy – but periodic as we Move across the table – implies Exact configuration of neutrons, Protons, and electrons are important Half filled d
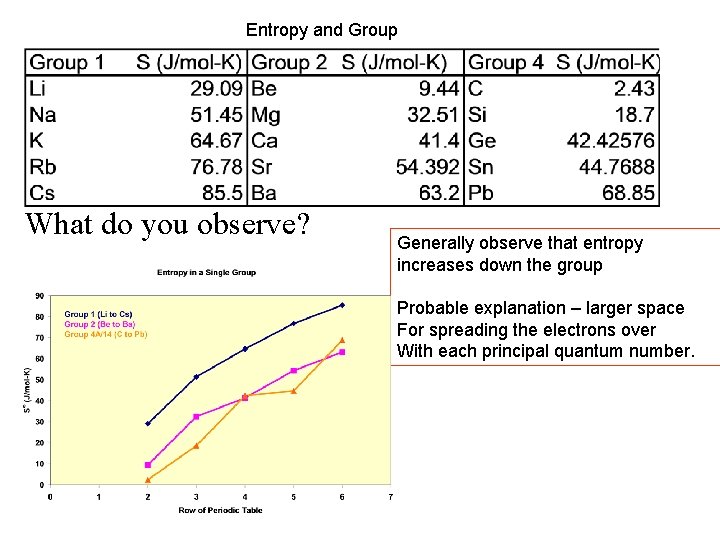
Entropy and Group What do you observe? Generally observe that entropy increases down the group Probable explanation – larger space For spreading the electrons over With each principal quantum number.
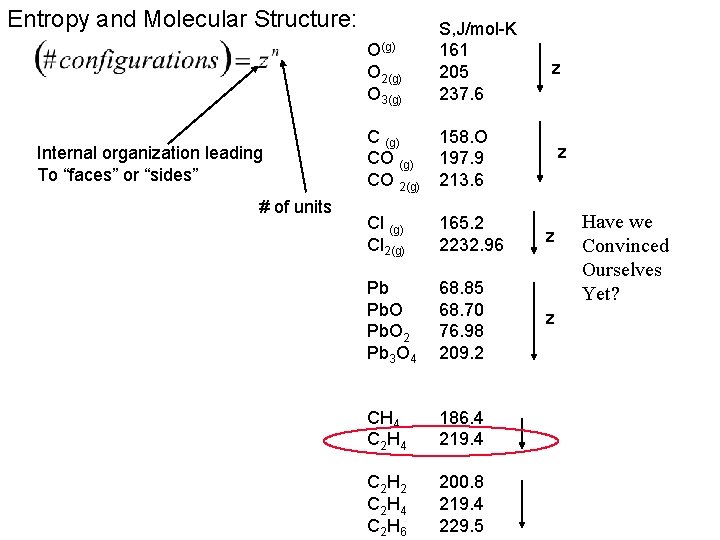
Entropy and Molecular Structure: Internal organization leading To “faces” or “sides” # of units O(g) O 2(g) O 3(g) S, J/mol-K 161 205 237. 6 C (g) CO 2(g) 158. O 197. 9 213. 6 Cl (g) Cl 2(g) 165. 2 2232. 96 z Pb Pb. O 2 Pb 3 O 4 68. 85 68. 70 76. 98 209. 2 z CH 4 C 2 H 4 186. 4 219. 4 C 2 H 2 C 2 H 4 C 2 H 6 200. 8 219. 4 229. 5 z z Have we Convinced Ourselves Yet?
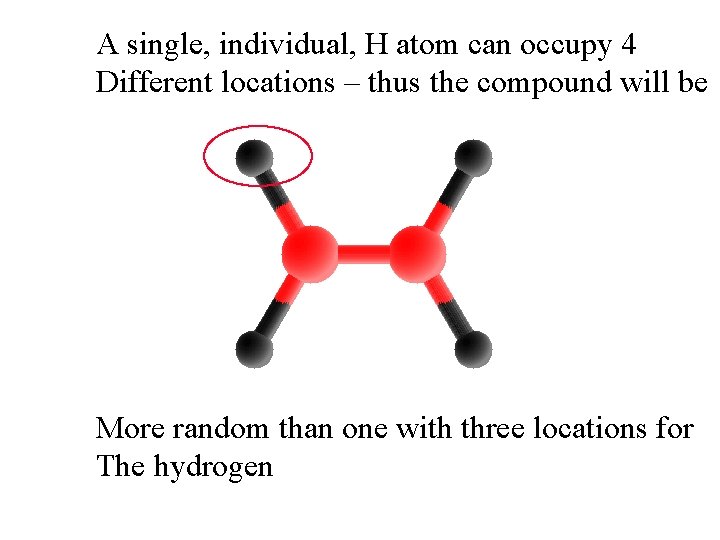
A single, individual, H atom can occupy 4 Different locations – thus the compound will be More random than one with three locations for The hydrogen
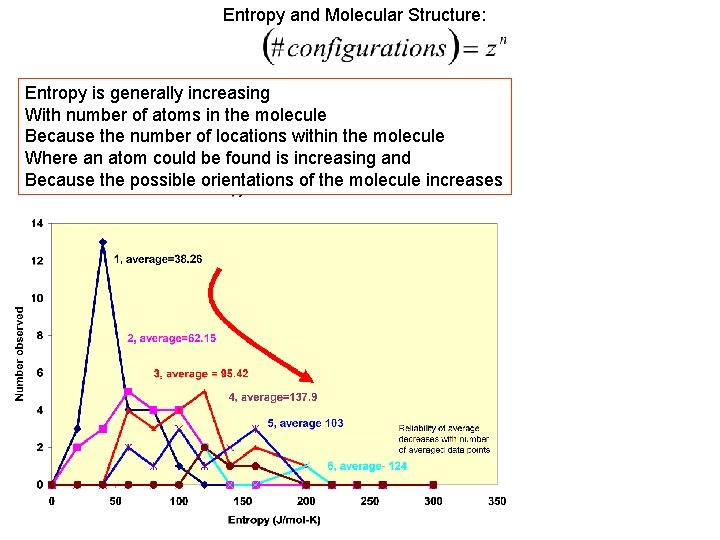
Entropy and Molecular Structure: Entropy is generally increasing With number of atoms in the molecule Because the number of locations within the molecule Where an atom could be found is increasing and Because the possible orientations of the molecule increases

Entropy changes in a chemical reaction Calculations here

Reaction Entropy Example Calculation Compare the total entropy change for the following reaction at 0 o. C and 600 o. C Substance O 2(gas) Pb. S(solid) Pb. O(solid) SO 2(gas) S 0 (J/K-mole) 205 91 66. 5 248 Do you See any Trend in This data? S is greater for the gases than for the solids!

Do an entropy change for gas phase reaction Or for the change in structure of lead oxide. Cu. S + Cu – Cu 2 S 66. 5 33. 2 120. 9



Reaction Entropy Example Calculation Compare the total entropy change for the following reaction at 0 o. C and 600 o. C Substance O 2(gas) Pb. S(solid) Pb. O(solid) SO 2(gas) S 0 (J/K-mole) 205 91 66. 5 248 Do you See any Trend in This data? S is greater for the gases than for the solids!

Reaction Entropy Example Calculation 3 Compare the total entropy change for the following reaction at 25 o. C and 600 o. C Substance O 2(gas) Pb. S(solid) Pb. O(solid) SO 2(gas) S 0 (J/K-mole) 205 91 66. 5 248

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Entropy of the surroundings

Spontaneous Process: Universal entropy increases (The universe is winding down. )
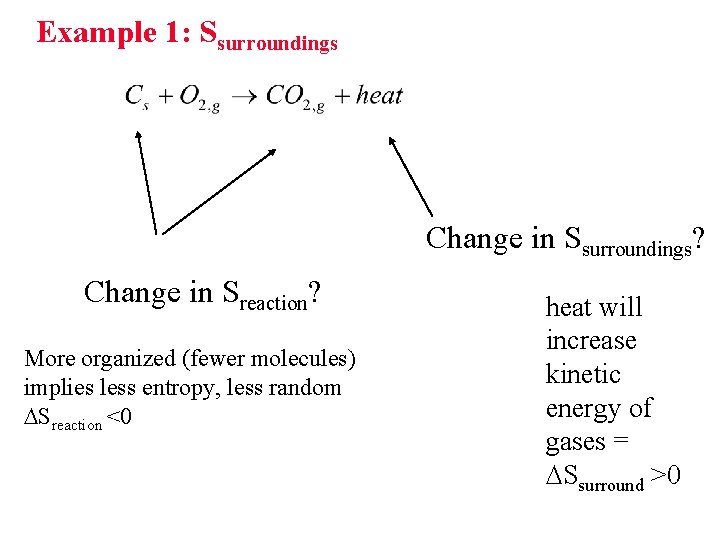
Example 1: Ssurroundings Change in Ssurroundings? Change in Sreaction? More organized (fewer molecules) implies less entropy, less random Sreaction <0 heat will increase kinetic energy of gases = Ssurround >0

Example 2: Ssurroundings From surroundings, withdraw heat, Less kinetic energy, less motion, less entropy + heat Although this process requires heat, it is spontaneous, driven by entropy of chemical reaction Reaction less random rx more random

The two reactions (the system) Interact with the surroundings by exchange of heat Heat of reaction must be related to entropy of surroundings

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Randomness of the “surroundings” affected By enthalpy

related to enthalpy or heat of reaction proportional Where will impact on Ssurroundings be greatest? a. 1 J at 600 o. C b. 1 J at 25 o. C
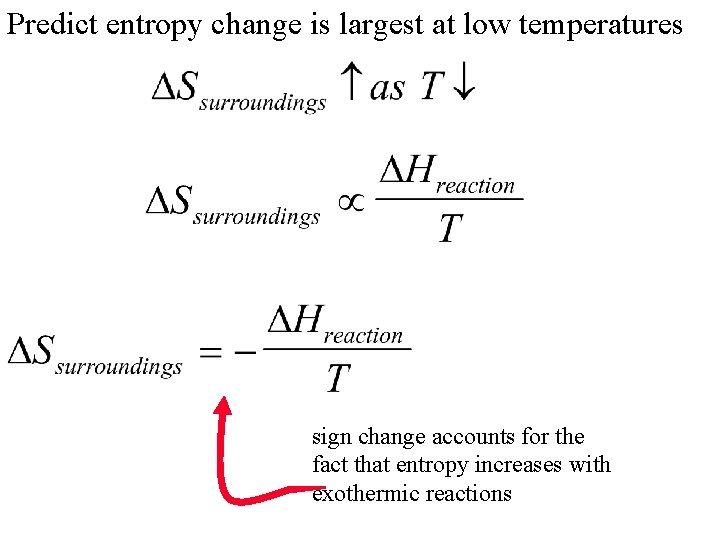
Predict entropy change is largest at low temperatures sign change accounts for the fact that entropy increases with exothermic reactions
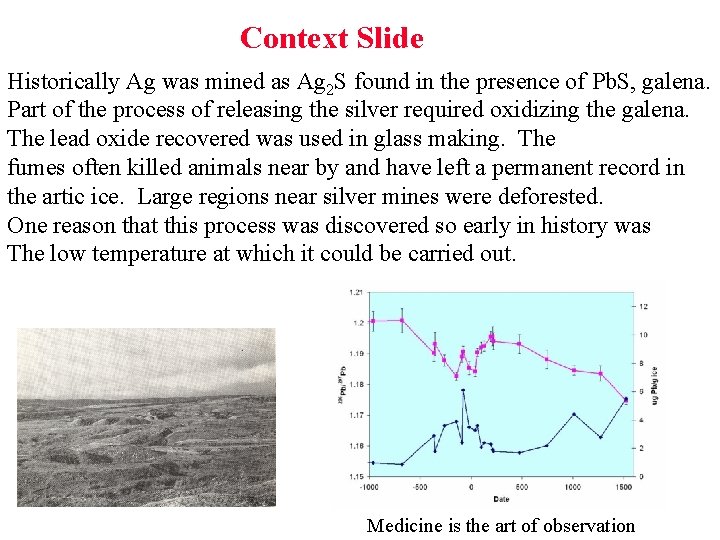
Context Slide Historically Ag was mined as Ag 2 S found in the presence of Pb. S, galena. Part of the process of releasing the silver required oxidizing the galena. The lead oxide recovered was used in glass making. The fumes often killed animals near by and have left a permanent record in the artic ice. Large regions near silver mines were deforested. One reason that this process was discovered so early in history was The low temperature at which it could be carried out. Medicine is the art of observation

Calculating Ssurrounding Example 1 Compare the change in entropy of the surroundings for this reaction at room temperature and at the temperature of a campfire (~600 o. C). Know: reaction Don’t know entropy red herrings? none
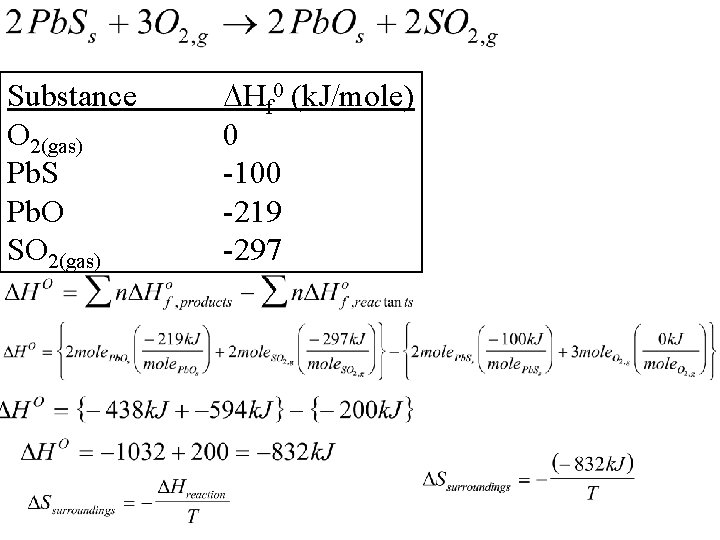
Substance O 2(gas) Pb. S Pb. O SO 2(gas) Hf 0 (k. J/mole) 0 -100 -219 -297

Calculating Ssurrounding Example 1 Compare the change in entropy of the surroundings for this reaction at room temperature and at the temperature of a campfire (~600 o. C). Our prediction was right! Larger at low T

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Total Entropy change With reaction enthalpy

This number, for a limited set of reactions, experimentally determined by varying T. Other values calculated by Hess’s Law

Reaction Entropy Example Calculation Compare the total entropy change for the following reaction at 0 o. C and 600 o. C Substance O 2(gas) Pb. S(solid) Pb. O(solid) SO 2(gas) S 0 (J/K-mole) 205 91 66. 5 248 Do you See any Trend in This data? S is greater for the gases than for the solids!

Reaction Entropy Example Calculation 3 Compare the total entropy change for the following reaction at 25 o. C and 600 o. C Substance O 2(gas) Pb. S(solid) Pb. O(solid) SO 2(gas) S 0 (J/K-mole) 205 91 66. 5 248

Spontaneous Less spontaneous

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity “Free energy” is a Way of accounting For contribution of randomness

Define Gibb’s free energy a) enthalpy of bonds b) organization of atoms Spontaneous reaction

Galen, 170 Marie the Jewess, 300 Charles Augustin James Watt Coulomb 1735 -1806 1736 -1819 Justus von Thomas Graham Liebig (1803 -1873 1805 -1869 Ludwig Boltzman 1844 -1906 Gilbert N Lewis 1875 -1946 Henri Louis Le. Chatlier 1850 -1936 Johannes Bronsted 1879 -1947 Jabir ibn Hawan, 721 -815 Luigi Galvani 1737 -1798 Richard AC E Erlenmeyer 1825 -1909 An alchemist Count Alessandro G A A Volta, 1747 -1827 James Joule (1818 -1889) Henri Bequerel 1852 -1908 Lawrence Henderson 1878 -1942 Galileo Galili Evangelista Torricelli 1564 -1642 1608 -1647 Amedeo Avogadro 1756 -1856 Rudolph Clausius 1822 -1888 Jacobus van’t Hoff 1852 -1911 Niels Bohr 1885 -1962 John Dalton 1766 -1844 William Thompson Lord Kelvin, 1824 -1907 Johannes Rydberg 1854 -1919 William Henry 1775 -1836 Johann Balmer 1825 -1898 J. J. Thomson 1856 -1940 Erwin Schodinger Louis de Broglie 1887 -1961 (1892 -1987) Fitch Rule G 3: Science is Referential Jean Picard 1620 -1682 Jacques Charles 1778 -1850 Francois-Marie Raoult 1830 -1901 Heinrich R. Hertz, 1857 -1894 Friedrich H. Hund 1896 -1997 Daniel Fahrenheit 1686 -1737 Max Planck 1858 -1947 Rolf Sievert, 1896 -1966 Blaise Pascal 1623 -1662 Georg Simon Ohm 1789 -1854 James Maxwell 1831 -1879 Robert Boyle, 1627 -1691 Isaac Newton 1643 -1727 Michael Faraday 1791 -1867 B. P. Emile Clapeyron 1799 -1864 Dmitri Mendeleev 1834 -1907 Svante Arrehenius Walther Nernst 1859 -1927 1864 -1941 Fritz London 1900 -1954 Wolfgang Pauli 1900 -1958 Johannes D. Van der Waals 1837 -1923 Marie Curie 1867 -1934 Anders Celsius 1701 -1744 Germain Henri Hess 1802 -1850 J. Willard Gibbs 1839 -1903 Fritz Haber 1868 -1934 Thomas M Lowry 1874 -1936 Werner Karl Linus Pauling Louis Harold Gray 1905 -1965 Heisenberg 1901 -1994 1901 -1976

Conceptually: H reaction + + Sreaction + + - Spontaneous? always at high T, 2 nd term lg. at low. T, 2 nd term sm never

Gibbs Free Energy Example 1 When will this reaction be spontaneous? H reaction + + Sreaction + + - Spontaneous? always at high T, 2 nd term lg. at low. T, 2 nd term sm never At Low. T!!!
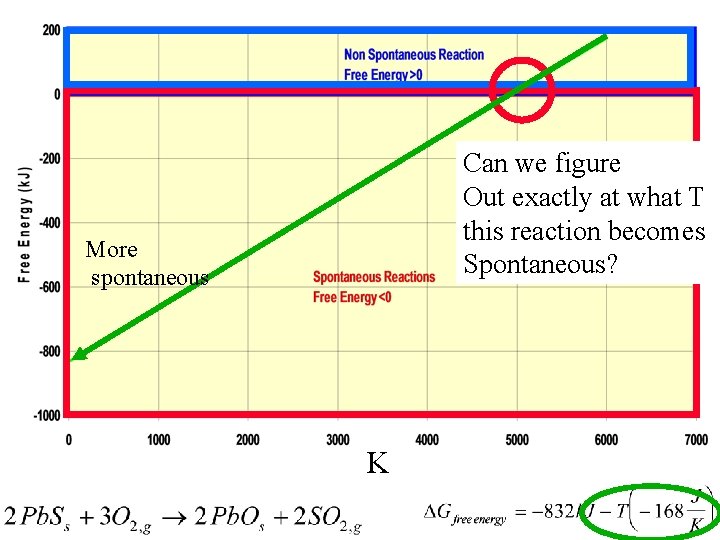
Can we figure Out exactly at what T this reaction becomes Spontaneous? More spontaneous K

To find when a reaction will just go Spontaneous (or not) 1. Use the equation: 2. Set Go to zero (equilibrium) 3. Solve for T. 4. Depending upon sign of enthalpy entropy determine if temperature decrease/increase causes Go to go negative

Gibbs Free Energy Example 2 At what T will this reaction become change between Spontaneous and non-spontaneous? Rx spontaneous at T<4952 K

Gibbs Free Energy Example 2: The only good substitute for Pb. CO 3 for white paint is Ti. O 2. To manufacture this paint need to be able to process titanium ore. At what temperature does the following reaction become spontaneous? Substance Ti. O 2 solid Ti O 2(gas) SO 2(gas) Csolid CO(gas) Hf 0 (k. J/mole) S 0(J/K-mole) -945 50 485 179. 45 0 205 -297 248 0 0 -110. 5 198
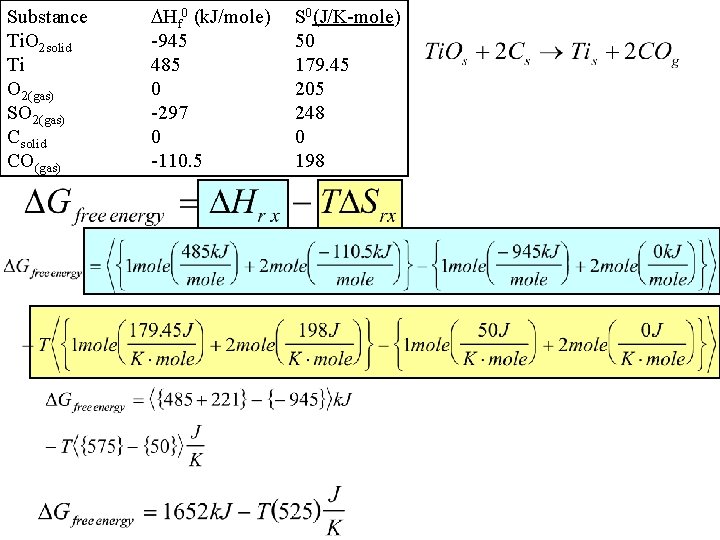
Substance Ti. O 2 solid Ti O 2(gas) SO 2(gas) Csolid CO(gas) Hf 0 (k. J/mole) -945 485 0 -297 0 -110. 5 S 0(J/K-mole) 50 179. 45 205 248 0 198

Substance Ti. O 2 solid Ti O 2(gas) SO 2(gas) Csolid CO(gas) Hf 0 (k. J/mole) -945 485 0 -297 0 -110. 5 S 0(J/K-mole) 50 179. 45 205 248 0 198 When is this reaction spontaneous: at high or low temp? Rx spontaneous > 3144 K
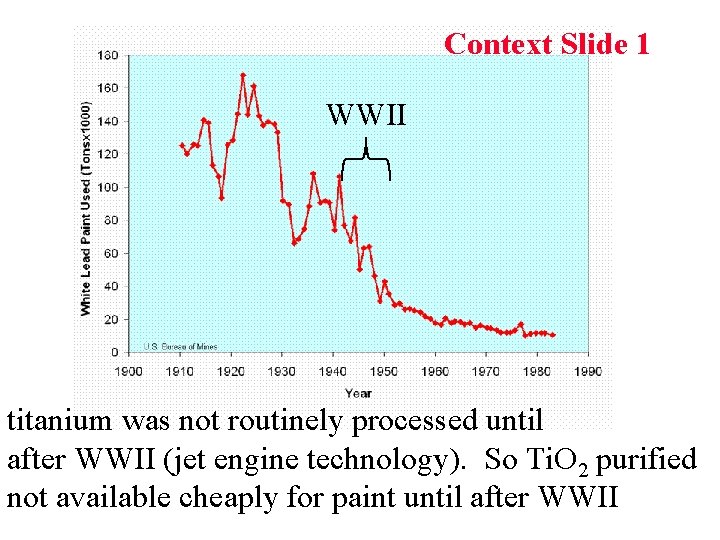
Context Slide 1 WWII titanium was not routinely processed until after WWII (jet engine technology). So Ti. O 2 purified not available cheaply for paint until after WWII

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Reference states for Free Energy
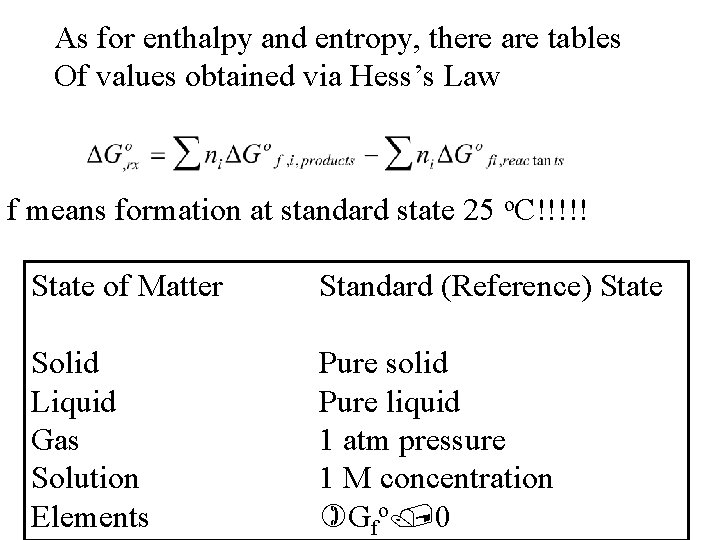
As for enthalpy and entropy, there are tables Of values obtained via Hess’s Law f means formation at standard state 25 o. C!!!!! State of Matter Standard (Reference) State Solid Liquid Gas Solution Elements Pure solid Pure liquid 1 atm pressure 1 M concentration Gfo 0

Properties and Measurements Property Size Volume Weight Temperature Unit m cm 3 gram Reference State size of earth m mass of 1 cm 3 water at specified Temp (and Pressure) o. C, K boiling, freezing of water (specified Pressure) amu (mass of 1 C-12 atom)/12 atomic mass of an element in grams atm, mm Hg earth’s atmosphere at sea level 1. 66053873 x 10 -24 g quantity mole Pressure Energy, General Animal hp heat BTU calorie Kinetic J Electrostatic electronic states in atom Electronegativity F horse on tread mill 1 lb water 1 o. F 1 g water 1 o. C m, kg, s 1 electrical charge against 1 V Energy of electron in vacuum

Gibbs Standard Free Energy Example Calc. 1: What Is the standard free energy change of the following Reaction? Substance O 2(gas) Pb. S Pb. O SO 2(gas) Gf 0 (k. J/mole) 0 -99 -188. 9 -300

Gibbs Standard Free Energy Example Calc. 1: What Is the standard free energy change of the following Reaction? For comparison

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Summing Reactions
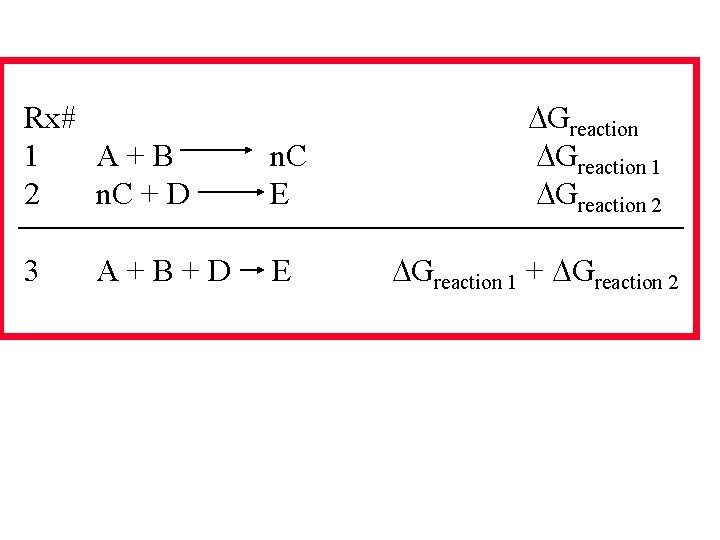
Rx# 1 A+B 2 n. C + D n. C E 3 E A+B+D Greaction 1 Greaction 2 Greaction 1 + Greaction 2

Summing Free Energy Example Calculation Why was lead one of the first elements first processed by man? A. Calculate the standard free energy of the Combined reactions. B. Calculate the free energy of the reaction at 600 o. C (campfire temp).

Summing Free Energy Example Calculation Why was lead one of the first elements first processed by man? A. Calculate the standard free energy of the Combined reactions. B. Calculate the free energy of the reaction at 600 o. C (campfire temp). Need standard free energy to solve A But! Will also need standard enthalpy and S To solve B – so solve for those
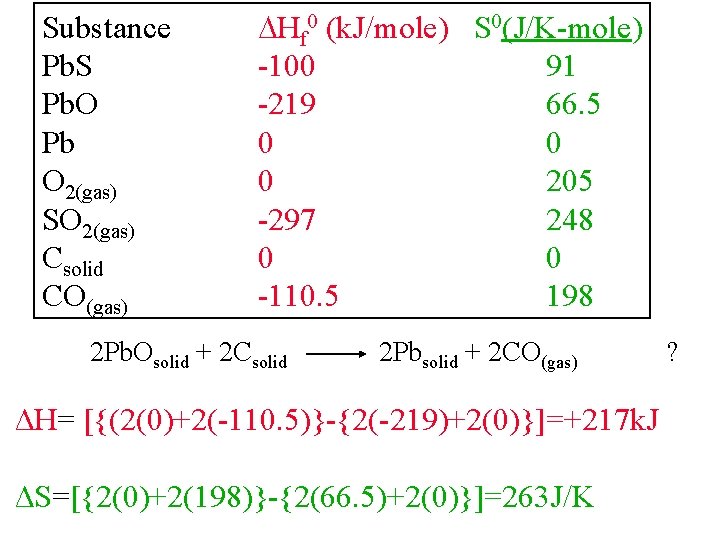
Substance Pb. S Pb. O Pb O 2(gas) SO 2(gas) Csolid CO(gas) Hf 0 (k. J/mole) S 0(J/K-mole) -100 91 -219 66. 5 0 0 0 205 -297 248 0 0 -110. 5 198 2 Pb. Osolid + 2 Csolid 2 Pbsolid + 2 CO(gas) ΔH= [{(2(0)+2(-110. 5)}-{2(-219)+2(0)}]=+217 k. J ΔS=[{2(0)+2(198)}-{2(66. 5)+2(0)}]=263 J/K ?

At standard conditions At campfire conditions

Net reaction at 25 o. C 2 Pb. S(solid) + 3 O 2(gas) Pb. O(solid) + 2 SO 2(gas) 2 Pb. Osoloid + 2 Csolid 2 Pbsolid + 2 CO(gas) 2 Pb. S + 3 O 2(gas) + 2 Csolid Grx -779. 8 k. J +138. 6 k. J 2 Pbsolid + 2 SO 2 gas + 2 COgas sum = -641 k. J The net standard free energy for the coupled two reactions is -641 k. J, spontaneous

Net reaction at 600 o. C 2 Pb. S(solid) + 3 O 2(gas) Pb. O(solid) + 2 SO 2(gas) 2 Pb. Osoloid + 2 Csolid 2 Pbsolid + 2 CO(gas) 2 Pb. S + 3 O 2(gas) + 2 Csolid Grx -685 k. J -12. 6 k. J 2 Pbsolid + 2 SO 2 gas + 2 COgas sum = -697 k. J The reduction of Pb in Pb. S to metal and oxidation of S in Pb. S to sulfur dioxide gas is spontaneous at campfire temperatures of 600 o. C

Context Slide
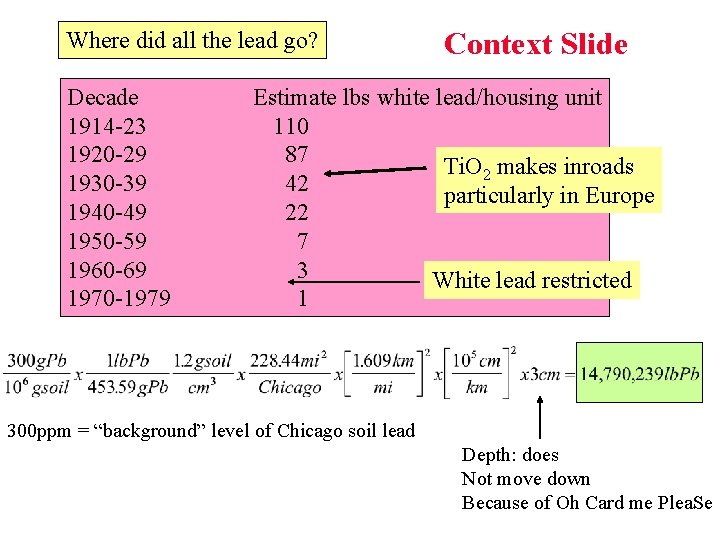
Where did all the lead go? Decade 1914 -23 1920 -29 1930 -39 1940 -49 1950 -59 1960 -69 1970 -1979 Context Slide Estimate lbs white lead/housing unit 110 87 Ti. O 2 makes inroads 42 particularly in Europe 22 7 3 White lead restricted 1 300 ppm = “background” level of Chicago soil lead Depth: does Not move down Because of Oh Card me Plea. Se
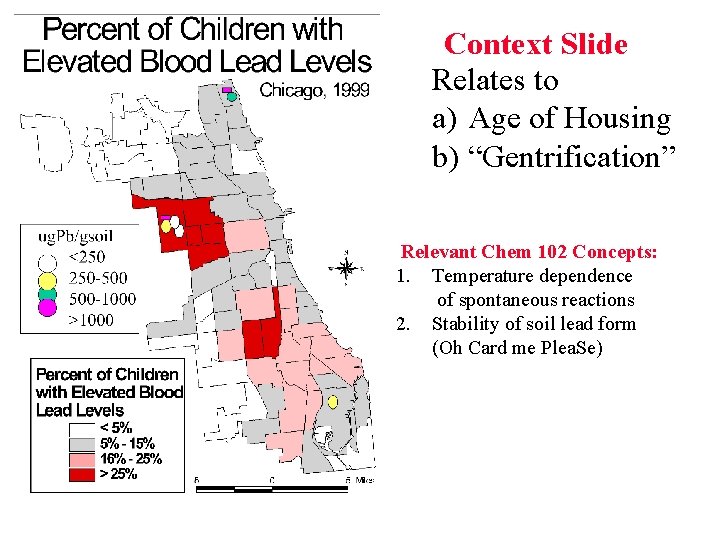
Context Slide Relates to a) Age of Housing b) “Gentrification” Relevant Chem 102 Concepts: 1. Temperature dependence of spontaneous reactions 2. Stability of soil lead form (Oh Card me Plea. Se)

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #20 Spontaneity Relating Free Energy To Concentrations

The free energy of the reaction related to a) standard free energy change b) and the ratio of concentrations of products to reactants, Q In this equation you can use (simulataneously) Pressures Concentrations The ln(Q) is treated as unitless
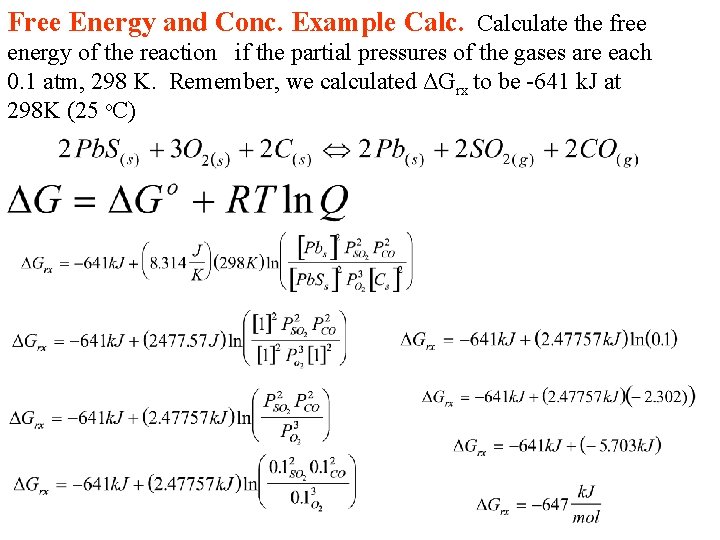
Free Energy and Conc. Example Calculate the free energy of the reaction if the partial pressures of the gases are each 0. 1 atm, 298 K. Remember, we calculated ΔGrx to be -641 k. J at 298 K (25 o. C)

When Q = K (equilibrium): 0 K At equilbrium no Energy to drive Rx one way or other K=1 -RT ln (1) = 0 K >1 -RT ln (>1) = -(+) < 0 K<1 -RT ln (<1) = -(-) > 0

Example Problem 2 Free Energy and Equilibrium: What is the equilibrium constant for the reaction at a campfire temperature? Go = -697 k. J/mol rx

Example 3 Free Energy and Equilibrium: The corrosion of Fe at 298 K is K = 10261. What is the equilibrium constant for corrosion of lead? 2 Pbsolid + O 2 gas 2 Pb. Osolid We don’t have any K values so we need To go to appendix for various enthalpy and Entropies to come at K from the backside
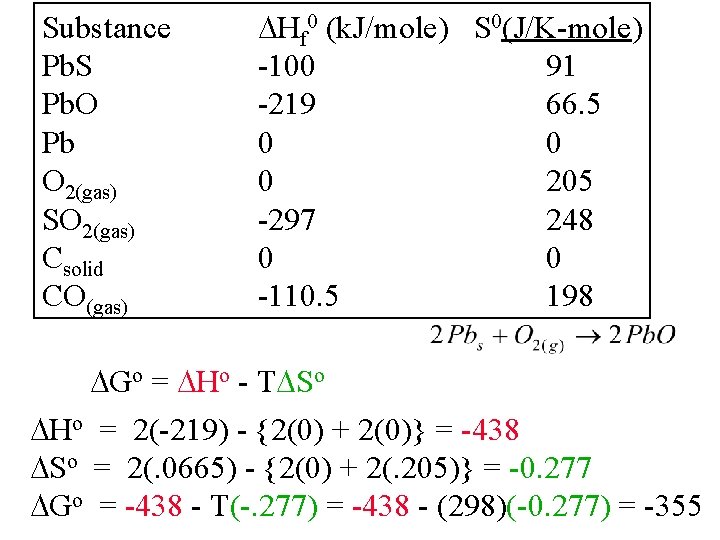
Substance Pb. S Pb. O Pb O 2(gas) SO 2(gas) Csolid CO(gas) Hf 0 (k. J/mole) S 0(J/K-mole) -100 91 -219 66. 5 0 0 0 205 -297 248 0 0 -110. 5 198 Go = Ho - T So Ho = 2(-219) - {2(0) + 2(0)} = -438 So = 2(. 0665) - {2(0) + 2(. 205)} = -0. 277 Go = -438 - T(-. 277) = -438 - (298)(-0. 277) = -355

RT ln K - Go K for rusting of Fe = 10261 K for rusting of Pb = 1. 27 x 1062 so: even though the reaction is favorable it is less so than for iron. Lead rusts less than iron = used for plumbing

“A” students work (without solutions manual) ~7 problems/night. Module #20 Spontaneity What you need to know

1. Be able to rank the entropy of various phases of materials 2. Be able to rank the entropy of various compounds 3. Relate entropy to enthalpy 4. Explain entropy concepts as related to chemical geometry 5. Calc. standard entropy change for a reaction 6. Calc. temperature at which a reaction becomes spontaneous 7. Explain why Ti. O 2 was relatively late in replacing Pb. CO 3 as a white pigment 8. Convert standard free energy to equilibrium constant

“A” students work (without solutions manual) ~7 problems/night. Module #20 Spontaneity END
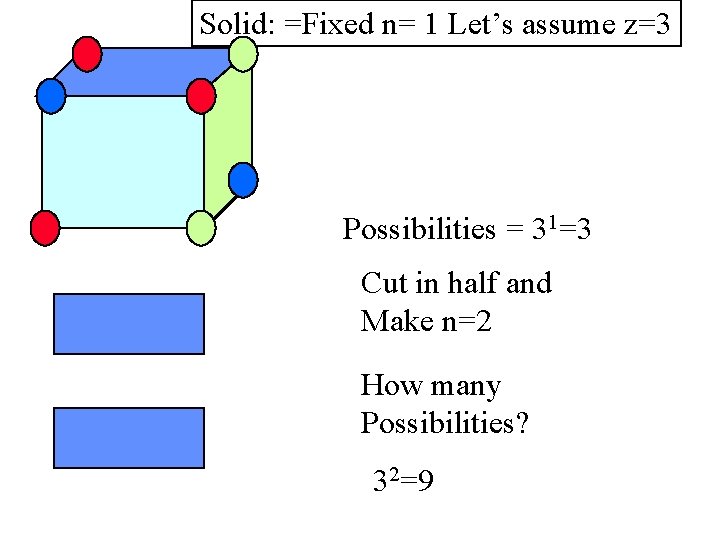
Solid: =Fixed n= 1 Let’s assume z=3 Possibilities = 31=3 Cut in half and Make n=2 How many Possibilities? 32=9
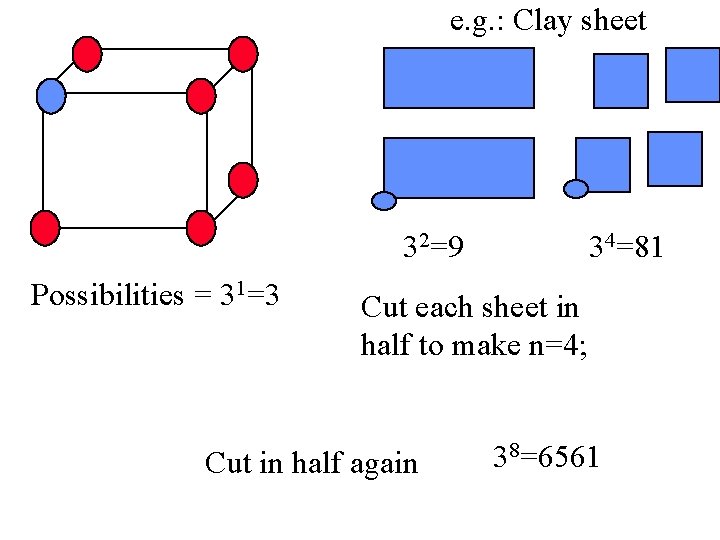
e. g. : Clay sheet 32=9 Possibilities = 31=3 34=81 Cut each sheet in half to make n=4; Cut in half again 38=6561

Entropy and Molecular Structure: Internal organization leading To “faces” or “sides” “z” = 2 # of units Number of “sides” increases with Number of atoms in the molecule “z” = 4 So we may make a general rule: Increasing atoms should increase The entropy of an individual the molecule Because it can have more possible Locations within the molecule for The atom, as well as more possible “sides” Or oriengations ins pacestates. “z” =6 “z” = 8
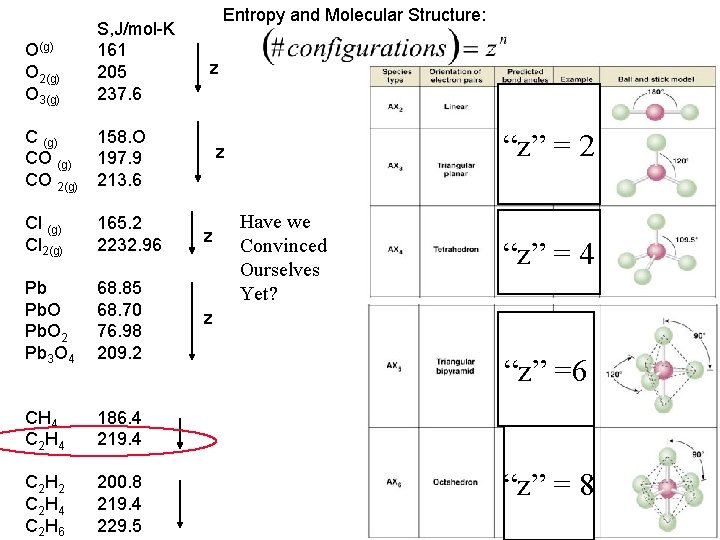
Entropy and Molecular Structure: O(g) O 2(g) O 3(g) S, J/mol-K 161 205 237. 6 C (g) CO 2(g) 158. O 197. 9 213. 6 Cl (g) Cl 2(g) 165. 2 2232. 96 z Pb Pb. O 2 Pb 3 O 4 68. 85 68. 70 76. 98 209. 2 z CH 4 C 2 H 4 186. 4 219. 4 C 2 H 2 C 2 H 4 C 2 H 6 200. 8 219. 4 229. 5 z “z” = 2 z Have we Convinced Ourselves Yet? “z” = 4 “z” =6 “z” = 8
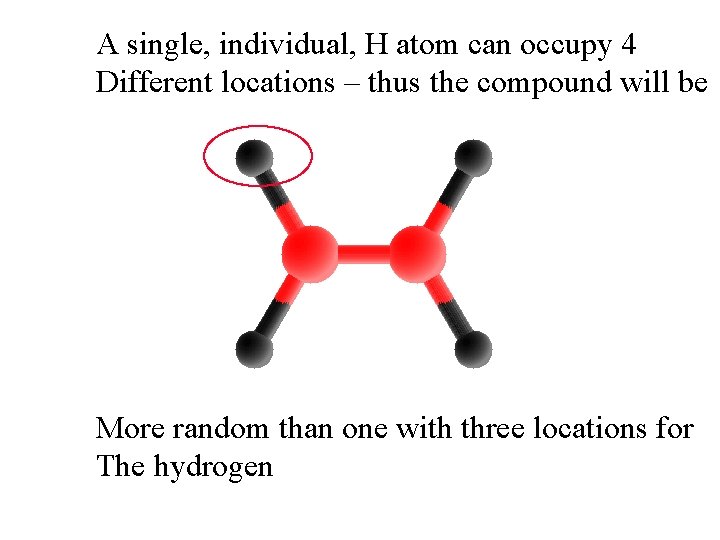
A single, individual, H atom can occupy 4 Different locations – thus the compound will be More random than one with three locations for The hydrogen
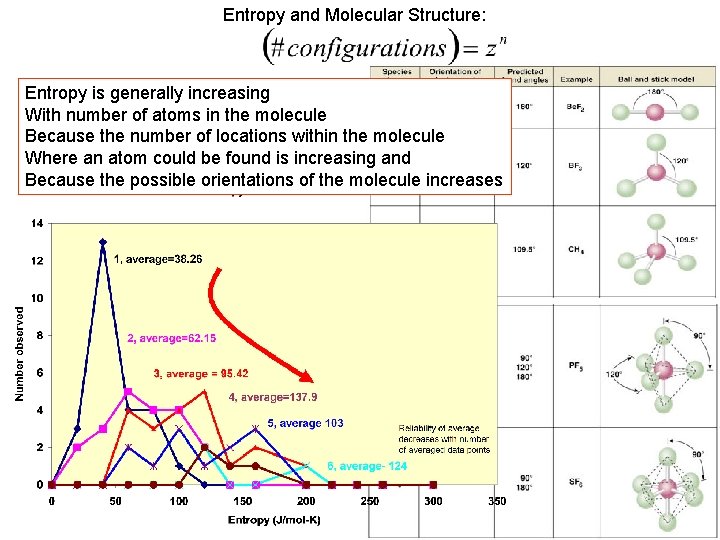
Entropy and Molecular Structure: Entropy is generally increasing With number of atoms in the molecule Because the number of locations within the molecule Where an atom could be found is increasing and Because the possible orientations of the molecule increases
- Slides: 89