A students work without solutions manual 10 problemsnight

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Introduction/ Context

Roman pipes at Pompei Solution Equilibria: Solubility In homes older than 1980 most of the plumbing is lead pipe (Pb = plumbous). Even in newer homes with copper pipe, solder joints are a lead/tin alloy. Even without solder joints, many of the faucet heads are machined with a 10 -20% lead content brass. The limit on lead is set to be <5 g Pb/109 g water < 5 x 10 --9 g Pb/g water < 5 ppb What to do?

Solutions: 1. 2. 3. Take out all plumbing Place water filtration devices at all outlets (sinks, showers, hoses). Have the water department take care of it somehow. What do you think the average homeowner prefers?

Coat the pipes from the inside out with a dense impermeable quasi-permanent layer = insoluble salt What makes insoluble salts? hint: same concepts as govern what ions are or are not spectators.

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Review Charge Density
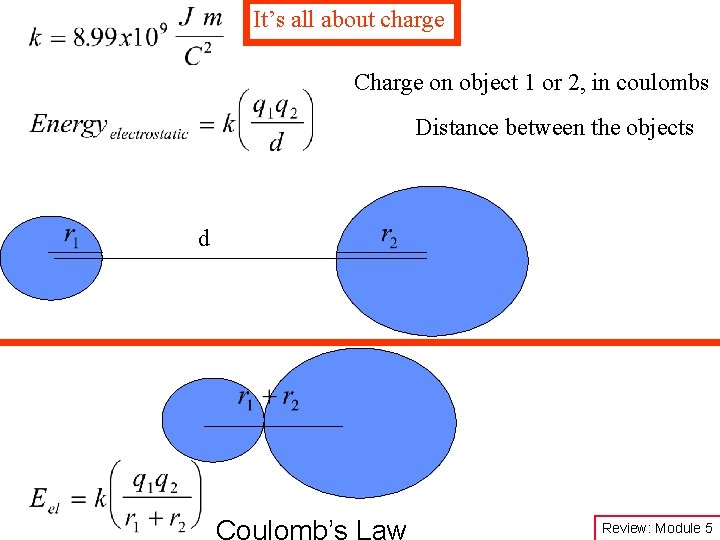
It’s all about charge Charge on object 1 or 2, in coulombs Distance between the objects d Coulomb’s Law Review: Module 5
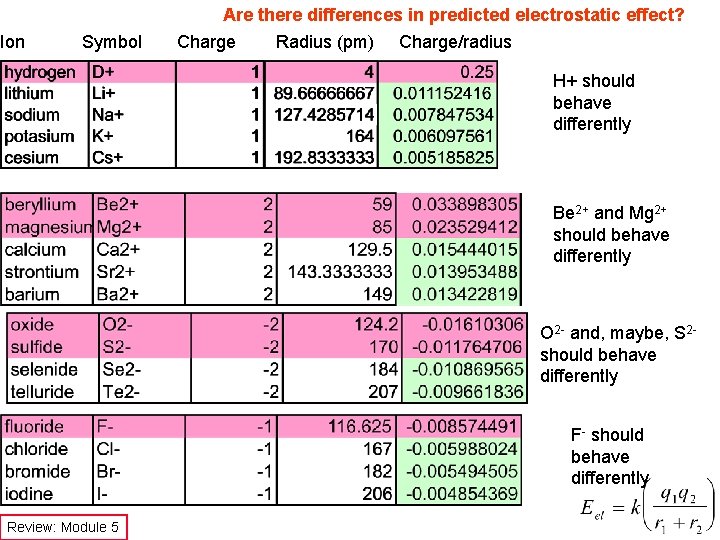
Ion Symbol Are there differences in predicted electrostatic effect? Charge Radius (pm) Charge/radius H+ should behave differently Be 2+ and Mg 2+ should behave differently O 2 - and, maybe, S 2 should behave differently F- should behave differently Review: Module 5
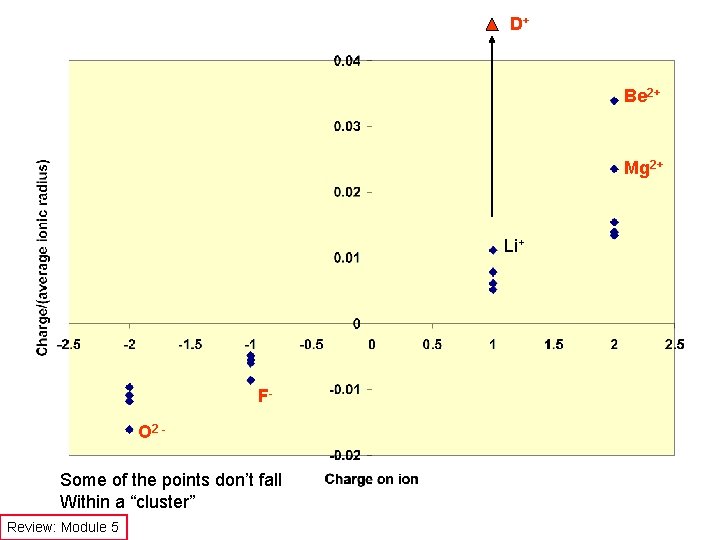
D+ Be 2+ Mg 2+ Li+ FO 2 Some of the points don’t fall Within a “cluster” Review: Module 5
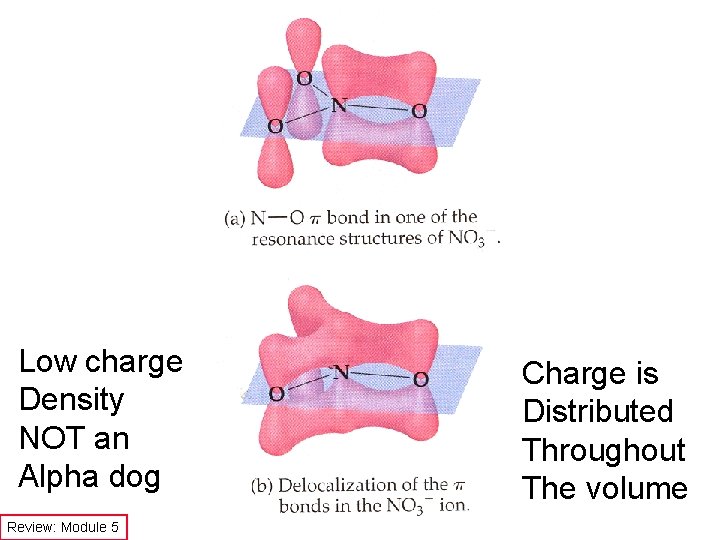
Low charge Density NOT an Alpha dog Review: Module 5 Charge is Distributed Throughout The volume
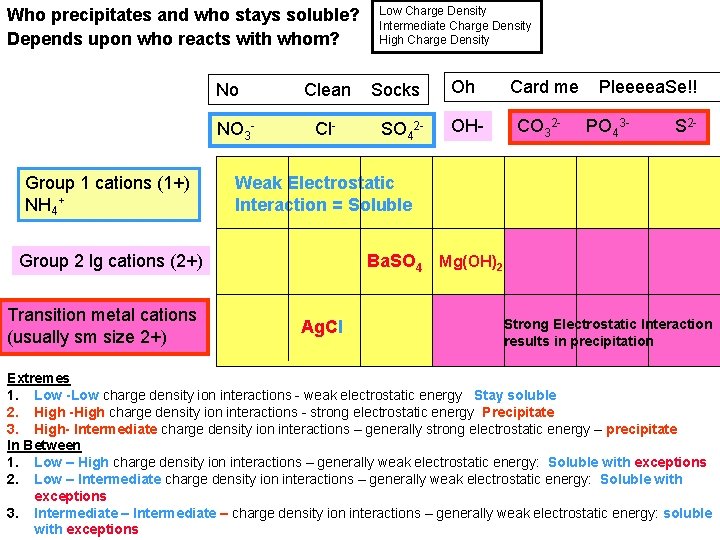
Who precipitates and who stays soluble? Depends upon who reacts with whom? No NO 3 Group 1 cations (1+) NH 4+ Clean Cl- Socks SO 42 - Oh Card me OH- CO 32 - Pleeeea. Se!! PO 43 - S 2 - Weak Electrostatic Interaction = Soluble Group 2 lg cations (2+) Transition metal cations (usually sm size 2+) Low Charge Density Intermediate Charge Density High Charge Density Ba. SO 4 Mg(OH)2 Ag. Cl Strong Electrostatic Interaction results in precipitation Extremes 1. Low -Low charge density ion interactions - weak electrostatic energy Stay soluble 2. High -High charge density ion interactions - strong electrostatic energy Precipitate 3. High- Intermediate charge density ion interactions – generally strong electrostatic energy – precipitate In Between 1. Low – High charge density ion interactions – generally weak electrostatic energy: Soluble with exceptions 2. Low – Intermediate charge density ion interactions – generally weak electrostatic energy: Soluble with exceptions 3. Intermediate – charge density ion interactions – generally weak electrostatic energy: soluble with exceptions
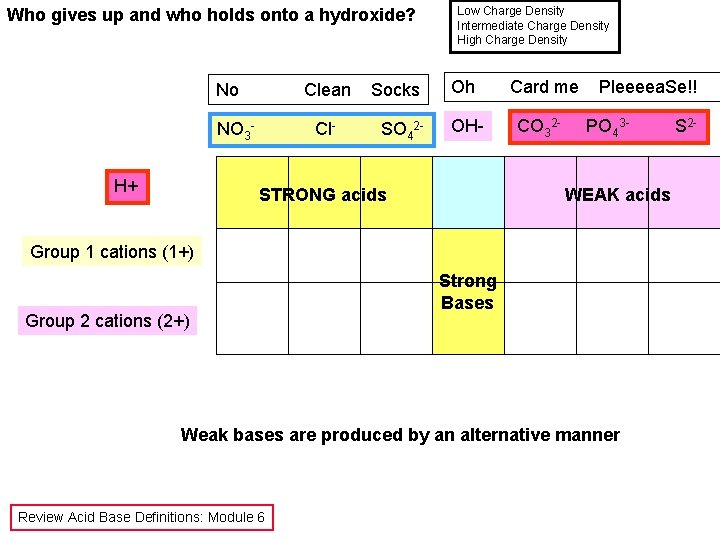
Who gives up and who holds onto a hydroxide? No Clean NO 3 H+ Cl- Socks SO 42 - Low Charge Density Intermediate Charge Density High Charge Density Oh Card me OH- CO 32 - STRONG acids Pleeeea. Se!! PO 43 - WEAK acids Group 1 cations (1+) Group 2 cations (2+) Strong Bases Weak bases are produced by an alternative manner Review Acid Base Definitions: Module 6 S 2 -

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Solubility Product, Ksp

Mathematically Express These Concepts: (Memorization Table is a short hand) Didn’t we learn A trick about this? The smaller K, the less aquated ions, the less soluble the material

Solubility Constants Numbers are determined from measuring the amount of stuff in solution. Mm+ Xx-

Example 1: Calculate the Ksp of Bismuth sulfide if there is 1. 0 x 10 -15 mol/L of the compound in solution at 25 o. C. Bismuth is a post transition metal with the electronic configuration of? : Bi s 2 d 10 p 3 What do you think it will do to become a cation? Calculate the Ksp of Bismuth sulfide if there is 1. 0 x 10 -15 mol/L of the compound in solution at 25 o. C.

Lose three e Bi s 2 d 10 p 3 Bi 3+ s 2 d 10 p 0 The electron configuration on S is: S s 2 p 4 What will it do to get to the noble gas? Calculate the Ksp of Bismuth sulfide if there is 1. 0 x 10 -15 mol/L of the compound in solution at 25 C.

Gain two e: S s 2 p 4 S 2 - s 2 p 6 Formula? : Bi 3+ with S 2 Bi 2 S 3 Calculate the Ksp of Bismuth sulfide if there is 1. 0 x 10 -15 mol/L of the compound in solution at 25 C.

Reaction? : Now What? What do we know/don’t know/want? Calculate the Ksp of Bismuth sulfide if there is 1. 0 x 10 -15 mol/L of the compound in solution at 25 C.

stoic Init Change Equil. Bi 2 S 3(solid) 1 solid -x 1. 0 x 10 -15 2 Bi 3+aquated + 3 S 2 -aquated 2 3 0 0 +2 x +3 x 2. 0 x 10 -15 3. 0 x 10 -15 OJO! Or you can do it: Calculate the Ksp of Bismuth sulfide if there is 1. 0 x 10 -15 mol/L of the compound in solution at 25 C.

For an enormous list of Ksp: http: //www. northland. cc. mn. us/chemistry/solubility_products. htm

Example Calculation 2 Calculate Solubility, s, and ion concentrations from Ksp Candidates for water treatment for lead? Ksp Pb. F 2 4 x 10 -8 Criteria you will Pb. Cl 2 1. 6 x 10 -5 use? Pb. I 2 1. 4 x 10 -8 Pb. SO 4 1. 3 x 10 -8 Pb. Cr. O 4 2 x 10 -16 Solubility = s= Pb. CO 3 1. 5 x 10 -15 amount of compound Pb(OH)2 1. 2 x 10 -15 that is soluble in Pb. S 7 x 10 -29 water Pb 3(PO 4)2 1 x 10 -54 Do our “rules” clue us to Ksp values?

stoic Init Change Equil? Pb 3(PO 4)2 1 solid -x - 3 Pb 2+ + 2 PO 433 2 0 0 3 x 2 x OJO! Are we done? No, need [Pb 2+]

Is this below the federal standards (5 ppb)? Yes:

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Common Ion Effects
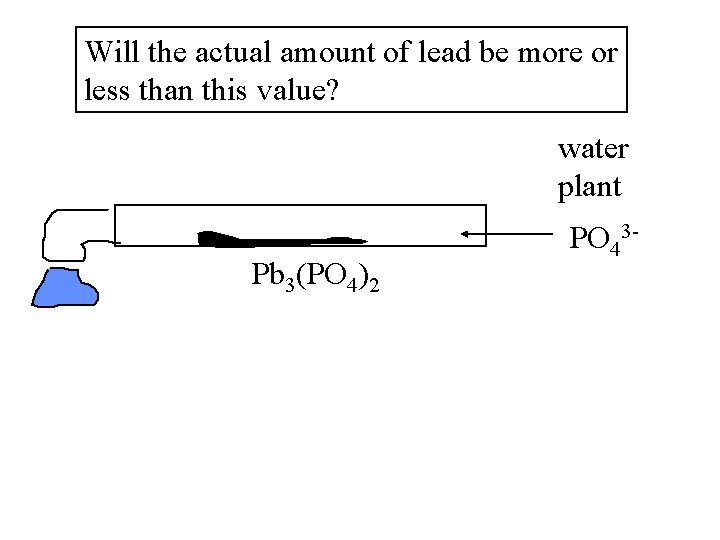
Will the actual amount of lead be more or less than this value? water plant Pb 3(PO 4)2 PO 43 -

Le. C principle: Common ion effect Constant flow of phosphate will suppress lead dissociation, value should be even lower. http: //www. caruschem. com/phosphate_zpoly. htm
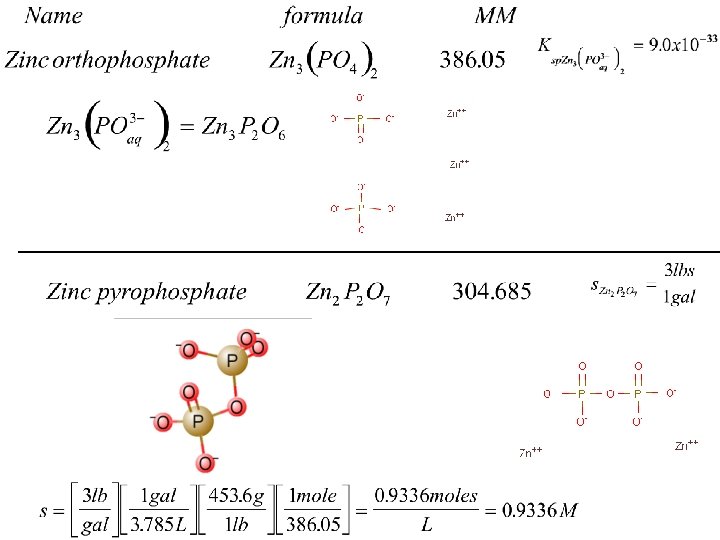

Water quality plants use either polyphosphate Or pyrophosphate (NOT orthophosphate) polyphosphate pyrophosphate

Example Calculation 3 Common Ion What will be the solubility of lead if a constant stream of 0. 001 M phosphate is fed through the water system? Notice the set up here 1. Looks like accounting for H+ from water 2. Called a “common ion” (ion from another rx) Zn 2 P 2 O 7 stoic Init Change Equil? Assume Pb 3(PO 4)2 1 solid -x - 2 Zn 2+ 2 PO 430. 001 3 Pb 2+ + 2 PO 433 2 0 0. 001 +3 x +2 x 3 x 0. 001

What will be the solubility of lead if a constant stream of 0. 001 M phosphate is fed through the water system? Zn 2 P 2 O 7 stoic Init Change Equil? Assume Pb 3(PO 4)2 1 solid -x - 2 Zn 2+ 2 PO 430. 001 3 Pb 2+ + 2 PO 433 2 0 0. 001 3 x 2 x 3 x 0. 001+2 x 3 x 0. 001
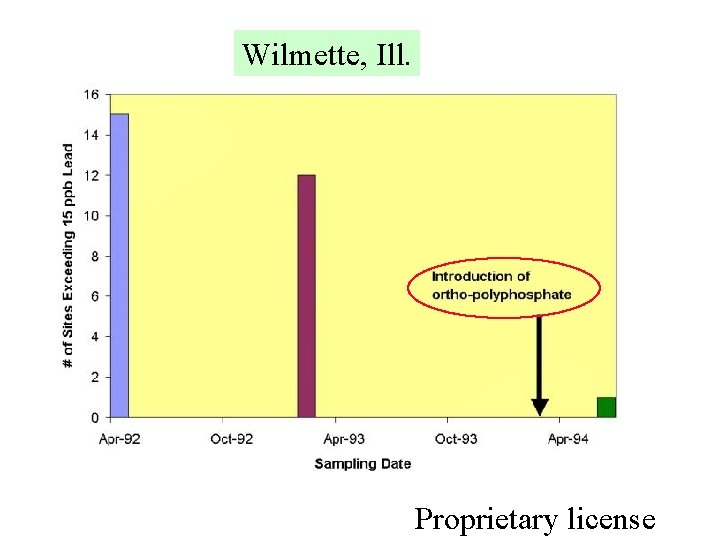
Wilmette, Ill. Proprietary license
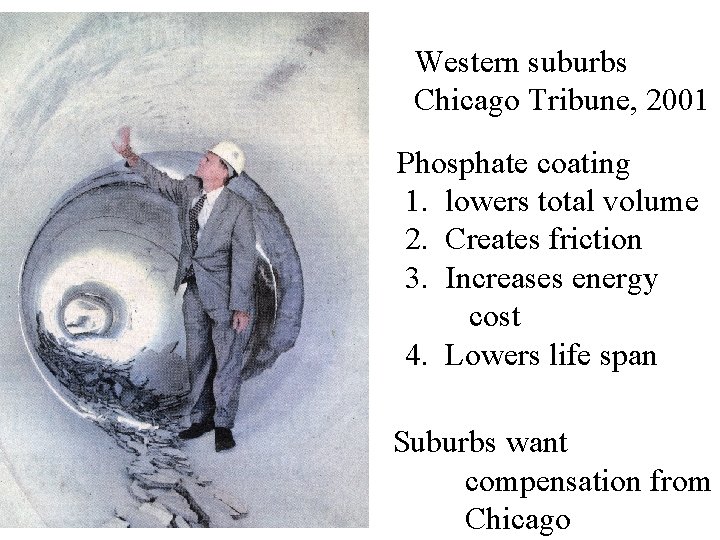
Western suburbs Chicago Tribune, 2001 Phosphate coating 1. lowers total volume 2. Creates friction 3. Increases energy cost 4. Lowers life span Suburbs want compensation from Chicago

Le. C principle: Common ion effect Can help ppt Can prevent ppt Addition of phosphate should suppress solubility Removal of lead Under basic conditions Should also enhance solubility Removal of phosphate Under acidic conditions Should enhance solubility
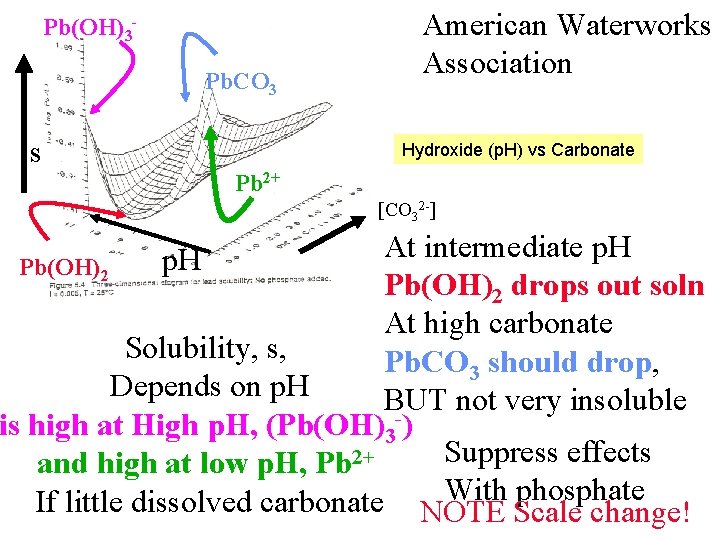
Pb(OH)3 Pb. CO 3 s American Waterworks Association Hydroxide (p. H) vs Carbonate Pb 2+ [CO 32 -] At intermediate p. H Pb(OH)2 drops out soln At high carbonate Solubility, s, Pb. CO 3 should drop, Depends on p. H BUT not very insoluble is high at High p. H, (Pb(OH)3 -) Suppress effects and high at low p. H, Pb 2+ With phosphate If little dissolved carbonate NOTE Scale change! p. H

MARCH 20/21, 2004 260 ppb Lead.
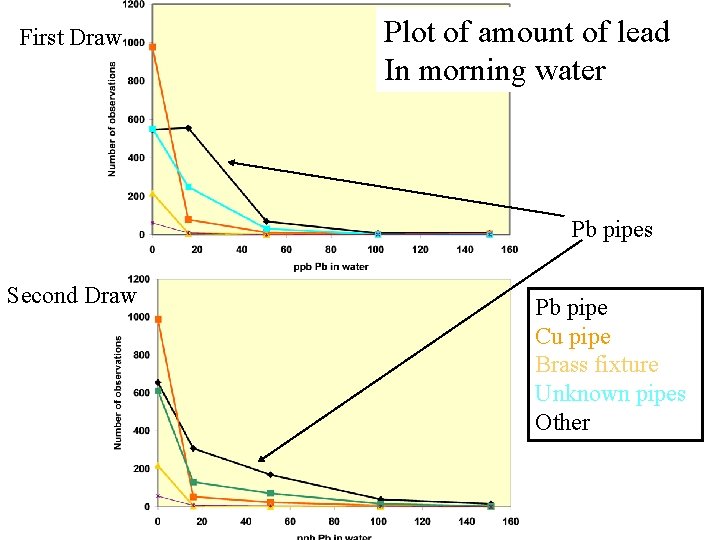
First Draw Plot of amount of lead In morning water Pb pipes Second Draw Pb pipe Cu pipe Brass fixture Unknown pipes Other

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Qualitative Analysis: Intro

Pb. S Muriatic acid “of or pertaining to brine Or salt” HCl

Qualitative Analysis First known text on chemical (as opposed of alchemical) analysis

Qualitative Analysis Oh Card me Plea S hydroxide Carbonate OHCO 3 -2 Phosphate Sulfide PO 43 S 2 - Cl. NH 3 OH- Move back and forth between precipitate And soluble species

Preceding example was: How to get rid of Pb 2+ by precipitation as a phosphate Next example: How to bring it back into solution a. With acid b. With a ligand
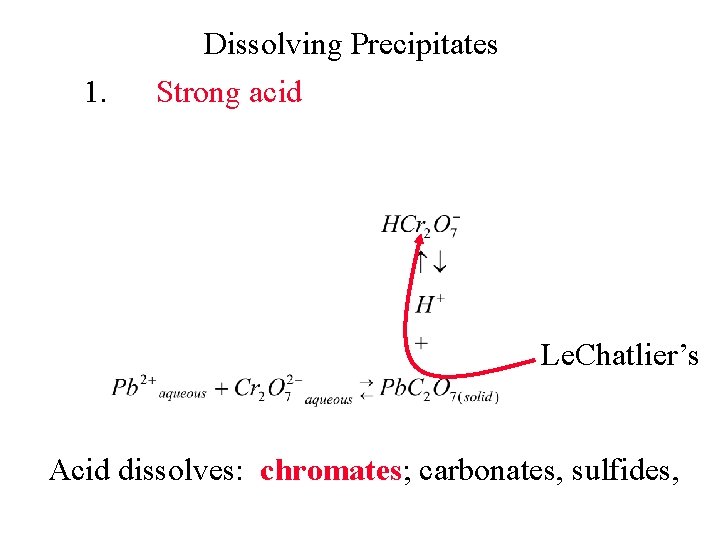
1. Dissolving Precipitates Strong acid Le. Chatlier’s Acid dissolves: chromates; carbonates, sulfides,

1. Dissolving Precipitates Strong acid Le. Chatlier’s Acid dissolves: chromates, carbonates; sulfides
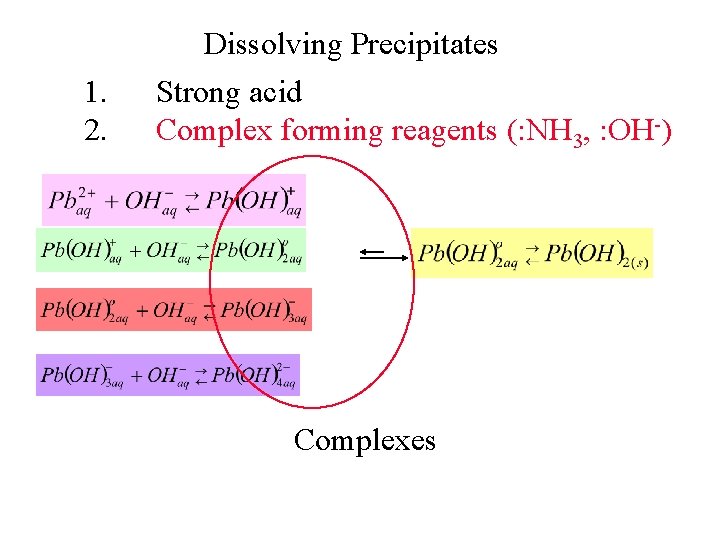
1. 2. Dissolving Precipitates Strong acid Complex forming reagents (: NH 3, : OH-) Complexes
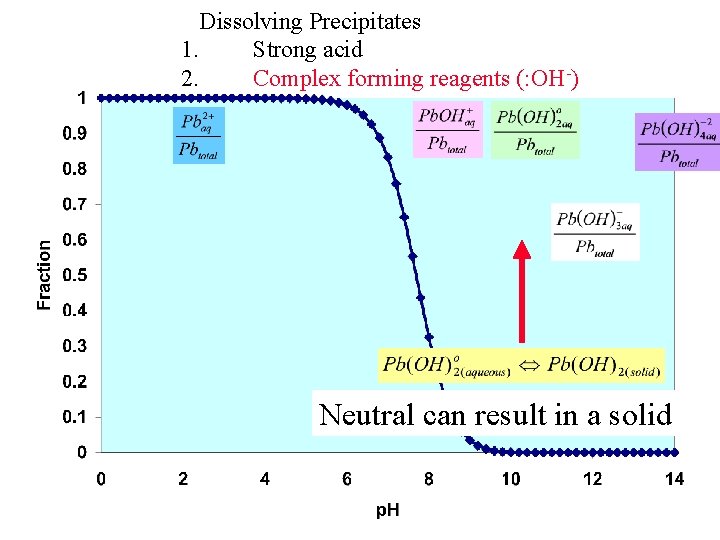
Dissolving Precipitates 1. Strong acid 2. Complex forming reagents (: OH-) Neutral can result in a solid

Ligands like : NH 3 and : OH- can bring various precipitates into solution Cation radius, CN CN: 4 tet; 6 oct Ag+ Cu 2+ 71, 87 Zn 2+ 74, 88 Cd 2+ 92, 109 Ni 2+ Al 3+ 67. 5 Sb 3+ 90 Sn 4+ Complex Kf Ag(NH 3)2+ Cu(NH 3)42+ Zn(NH 3)42+ Cd(NH 3)42+ Ni(NH 3)62+ 1. 8 x 107 2 x 1012 3. 6 x 108 2. 8 x 107 9 x 108 Hold Coordination Number constant What do you observe? Complex Kf Zn(OH)42 Cd(OH)42 - 3 x 1014 1. 2 x 109 Al(OH)4 Sb(OH)4 Sn(OH)62 - 1 x 1033 Size matters: the larger The cation (less charge dense) the smaller the Kf
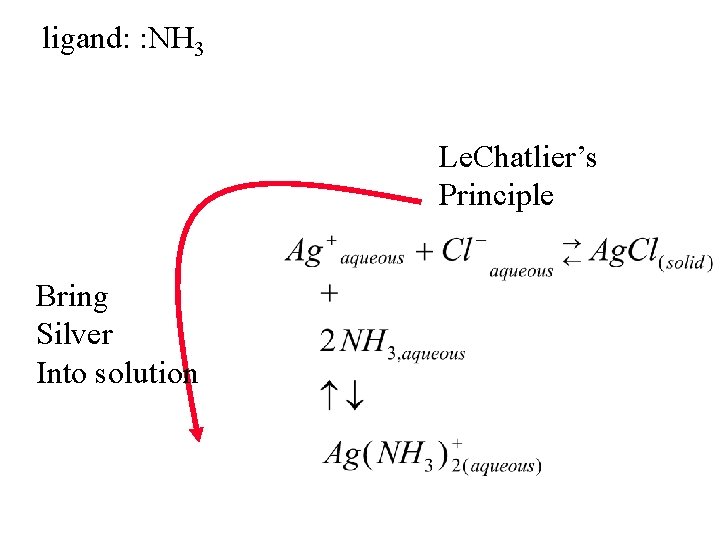
ligand: : NH 3 Le. Chatlier’s Principle Bring Silver Into solution

Example 4: Calculate moles Ag. Cl dissolved in 1 L of 6. 0 M NH 3 at a temperature of 298 K Know 6. 0 M NH 3 reaction stoic Init Change Equil Ag. Cls n. a. Don’t Know s Krx [NH 3] 2 6. 0 M -2 x 6. 0 -2 x Red Herring 298 K [Ag(NH 3)2+] 1 0 x x [Cl-] 1 0 x x

Example: Calculate moles Ag. Cl dissolved in 1 L of 6. 0 M NH 3 at a temperature of 298 K stoic init change equil Ag. Cls n. a. [NH 3] 2 6. 0 M -2 x 6. 0 -2 x [Ag(NH 3)2+] 1 0 x x [Cl-] 1 0 x x X= moles complex = moles Ag. Cl dissolved Alanah: Check Moles vs molarity
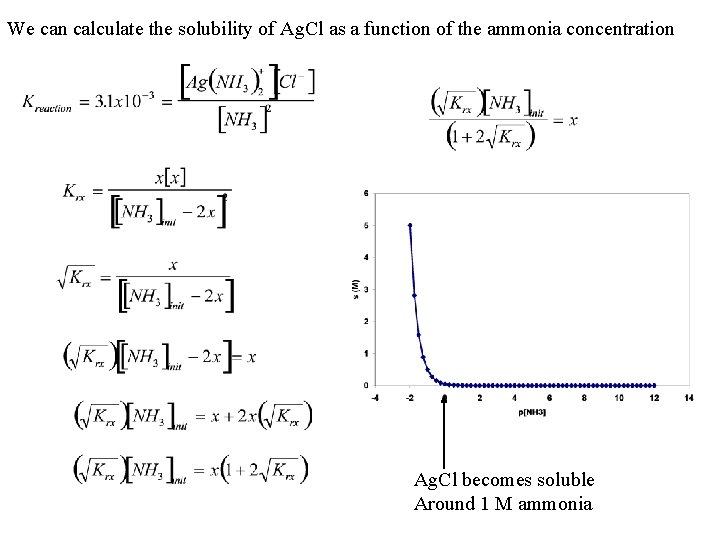
We can calculate the solubility of Ag. Cl as a function of the ammonia concentration Ag. Cl becomes soluble Around 1 M ammonia

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Qualitative Analysis: Cl to separate Pb, Hg 22+, Ag
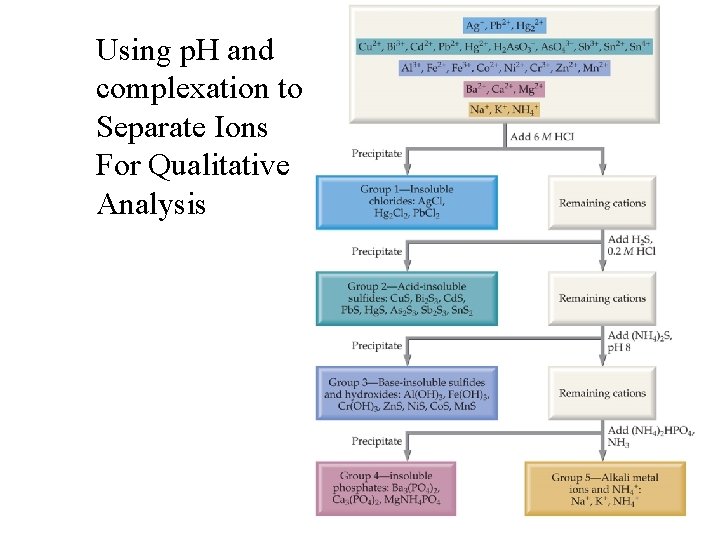
Using p. H and complexation to Separate Ions For Qualitative Analysis

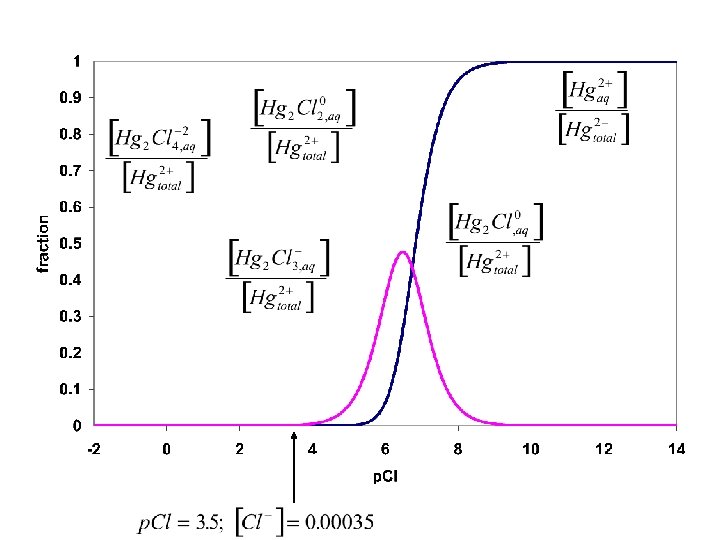
Separation in lab is not always the same As in the text. Text starts with 6 M Cl- = p. Cl = -. 778 The separation below Corresponds to What goes on in lab
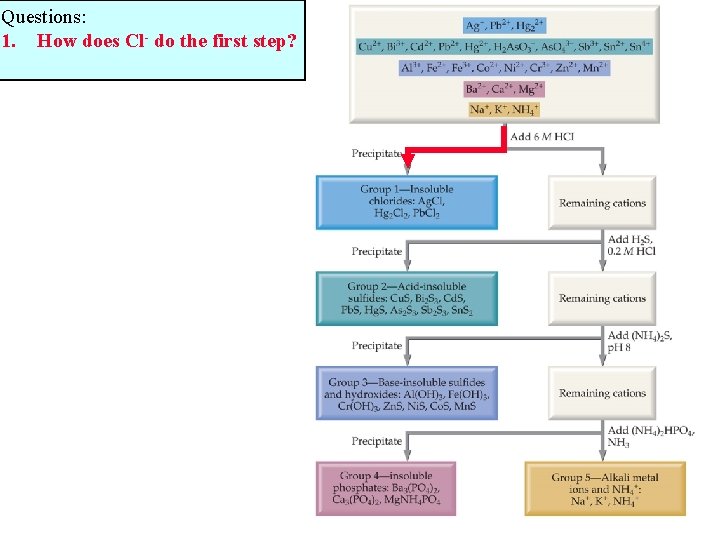
Questions: 1. How does Cl- do the first step?
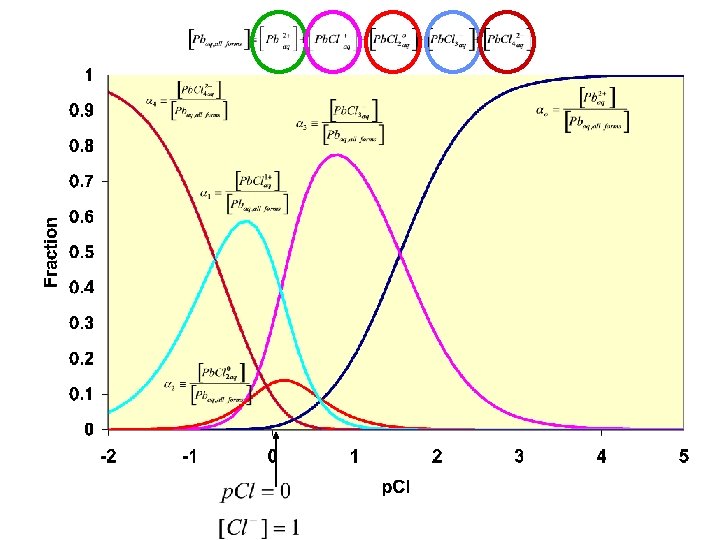
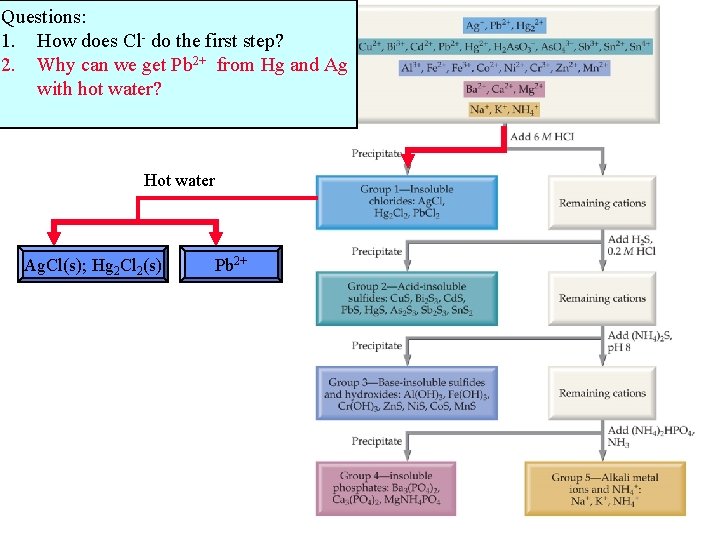
Questions: 1. How does Cl- do the first step? 2. Why can we get Pb 2+ from Hg and Ag with hot water? Hot water Ag. Cl(s); Hg 2 Cl 2(s) Pb 2+
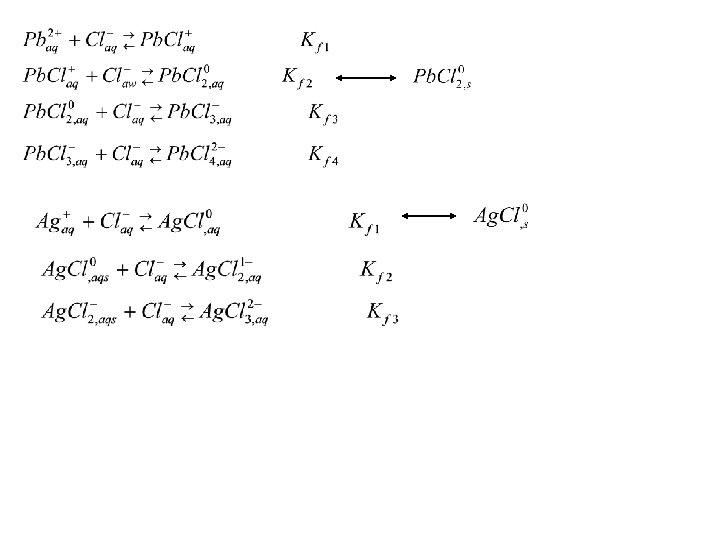
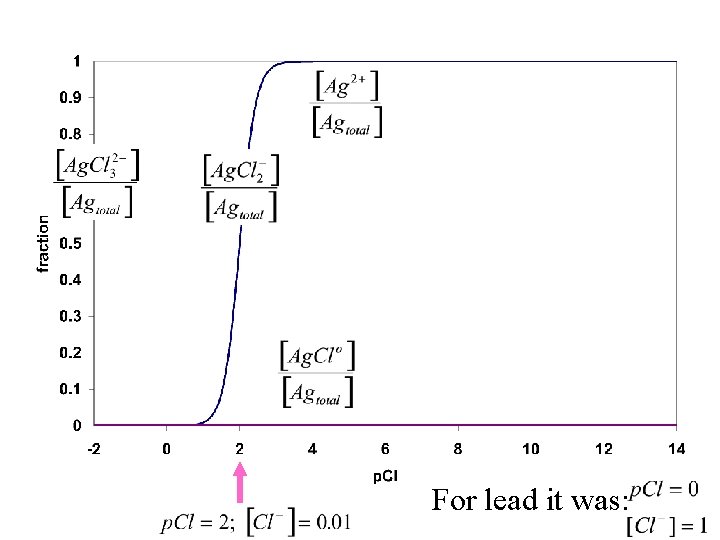
For lead it was:
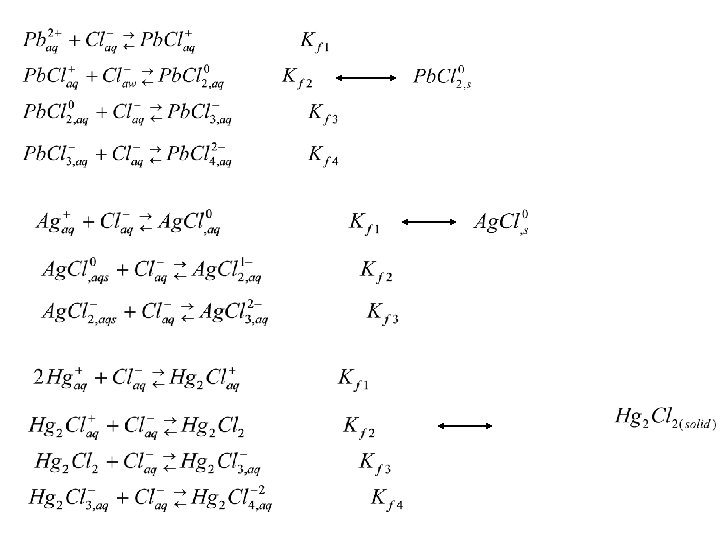
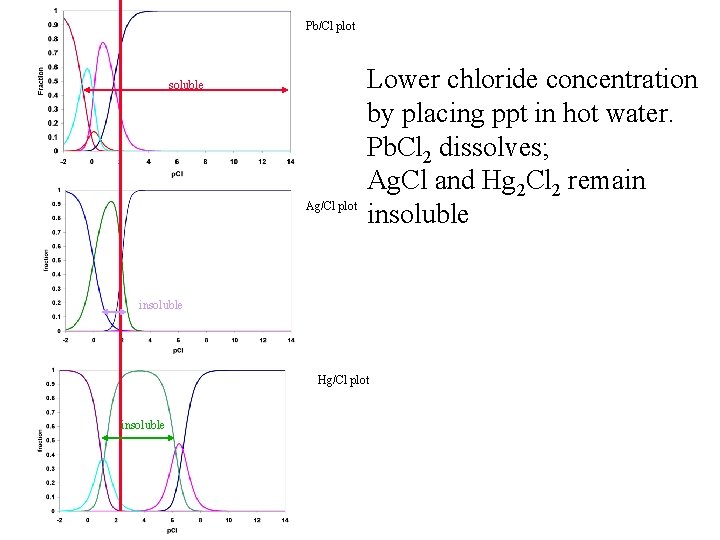
Pb/Cl plot soluble Ag/Cl plot Lower chloride concentration by placing ppt in hot water. Pb. Cl 2 dissolves; Ag. Cl and Hg 2 Cl 2 remain insoluble Hg/Cl plot insoluble
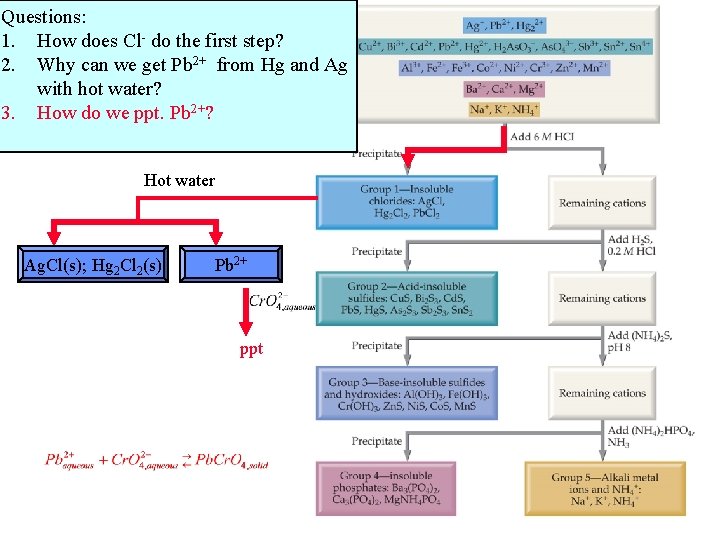
Questions: 1. How does Cl- do the first step? 2. Why can we get Pb 2+ from Hg and Ag with hot water? 3. How do we ppt. Pb 2+? Hot water Ag. Cl(s); Hg 2 Cl 2(s) Pb 2+ ppt

Candidates for precipitating Pb 2 from supernatent? Pb. F 2 Pb. Cl 2 Pb. I 2 Pb. SO 4 Pb. Cr. O 4 Pb. CO 3 Pb(OH)2 Pb. S Pb 3(PO 4)2 Ksp, Pb 4 x 10 -8 1. 6 x 10 -5 1. 4 x 10 -8 1. 3 x 10 -8 2 x 10 -16 1. 5 x 10 -15 1. 2 x 10 -15 7 x 10 -29 1 x 10 -54 Need very insoluble species, that doesn’t bring along Cd or Zn Cd - ? 5. 5 x 10 -13 6. 45 x 10 -6 _ _ Zn - OH , CO 32 - bring Cd 2+ _ _ S 2 -, PO 43 - bring Zn 2+ 6. 3 x 10 -17 1. 99 x 10 -25 3. 8 x 10 -36 Makes a very nice, older, chrome pigment!!

Example Calculation 5. Before lead in paint was discontinued, lead chromate was a common pigment in yellow paint. A 1. 0 L solution is prepared by mixing 0. 50 mg of lead nitrate with 0. 020 mg of potassium chromate. Will a precipitate form? P is like Q
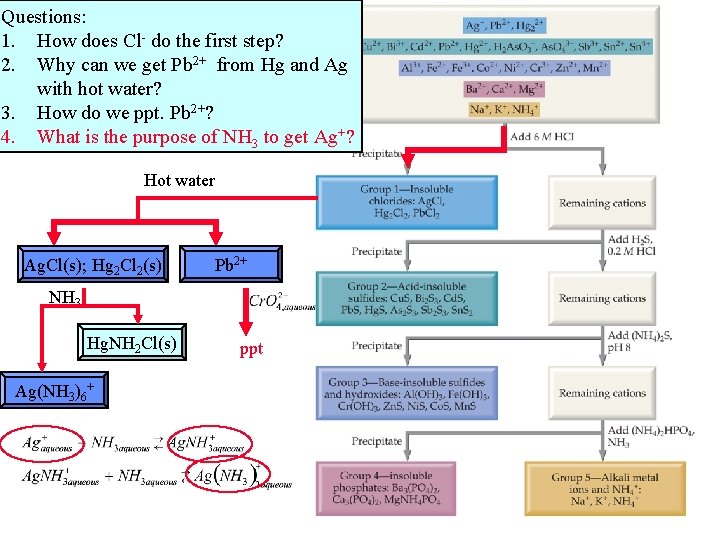
Questions: 1. How does Cl- do the first step? 2. Why can we get Pb 2+ from Hg and Ag with hot water? 3. How do we ppt. Pb 2+? 4. What is the purpose of NH 3 to get Ag+? Hot water Ag. Cl(s); Hg 2 Cl 2(s) Pb 2+ NH 3 Hg. NH 2 Cl(s) Ag(NH 3)6+ ppt

We did an example in which we calculated the Solubility of Ag. Cl in the presence of ammonia Which number is larger? Which has the higher solubility? water or ammonia
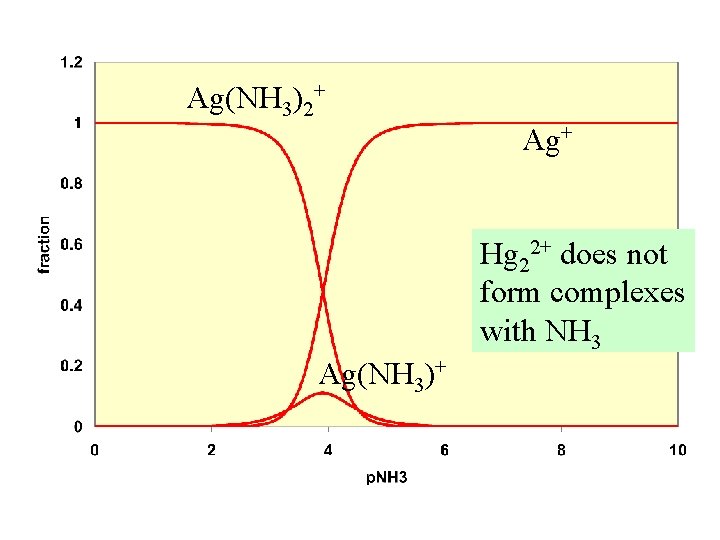
Ag(NH 3)2+ Ag+ Hg 22+ does not form complexes with NH 3 Ag(NH 3)+

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Qualitative Analysis: S to separate Cu 2+, Hg+, Bi 3+, Cd 2+
Questions: 1. How does H 2 S do the second step?

Cu 2+ Bi 3+ Hg 2+ Cd 2+ Al 3+ Fe 3+ Cr 3+ Mn 2+ Zn 2+ Ba 2+ Ca 2+ log. Ksp -48. 5 -52. 7 -27. 0 S 2 - Strong precipitators with S 2 -, will ppt at very low [S 2 -] Weak Precipitators with [S 2 -] Will ppt only at higher [S 2 -] How can we control [S 2 -] Easily? -10. 5 -24. 7 -
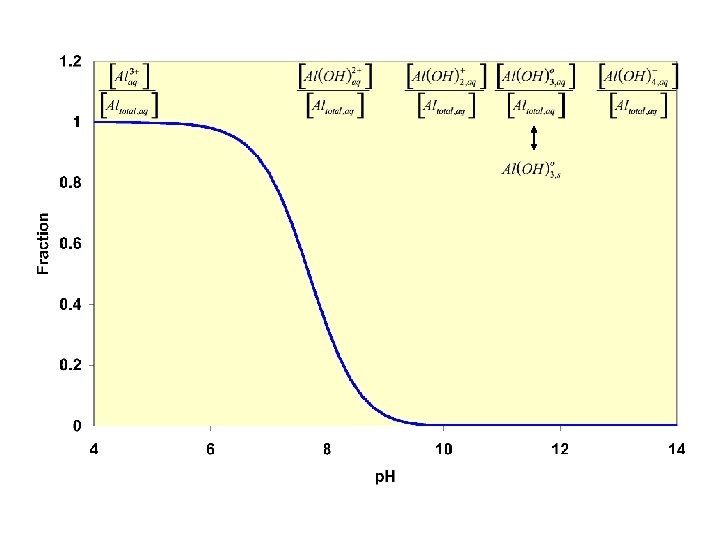
![Control [S 2 -] through p. H Who ppts here? Very insoluble MS species Control [S 2 -] through p. H Who ppts here? Very insoluble MS species](http://slidetodoc.com/presentation_image_h2/cbfece6c360b1d33b96f7e25cefc9638/image-71.jpg)
Control [S 2 -] through p. H Who ppts here? Very insoluble MS species Who ppt here? More soluble MS species

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Qualitative Analysis: S to separate Zn 2+, Co+2, Mn 2+, Ni 2+
Questions: 1. How does (NH 4)2 S do the first step? 2. Why is the solution basic (p. H 8) in next step?
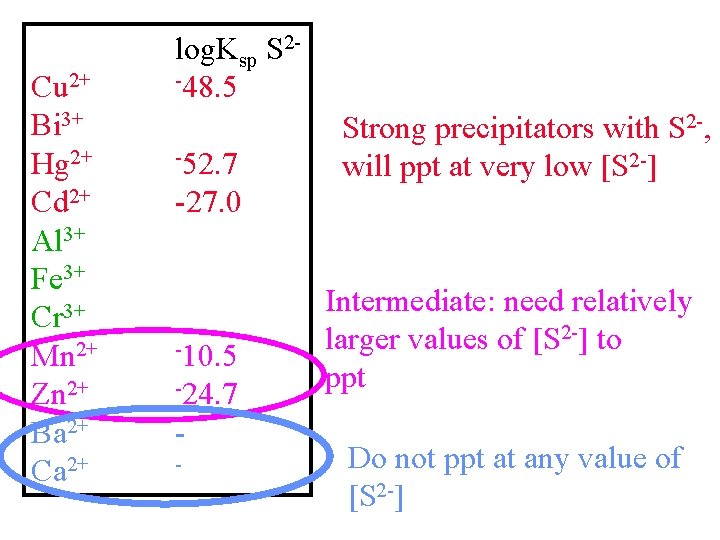
Cu 2+ Bi 3+ Hg 2+ Cd 2+ Al 3+ Fe 3+ Cr 3+ Mn 2+ Zn 2+ Ba 2+ Ca 2+ log. Ksp S 2 -48. 5 -52. 7 Strong precipitators with S 2 -, will ppt at very low [S 2 -] -27. 0 -10. 5 -24. 7 - Intermediate: need relatively larger values of [S 2 -] to ppt Do not ppt at any value of [S 2 -]
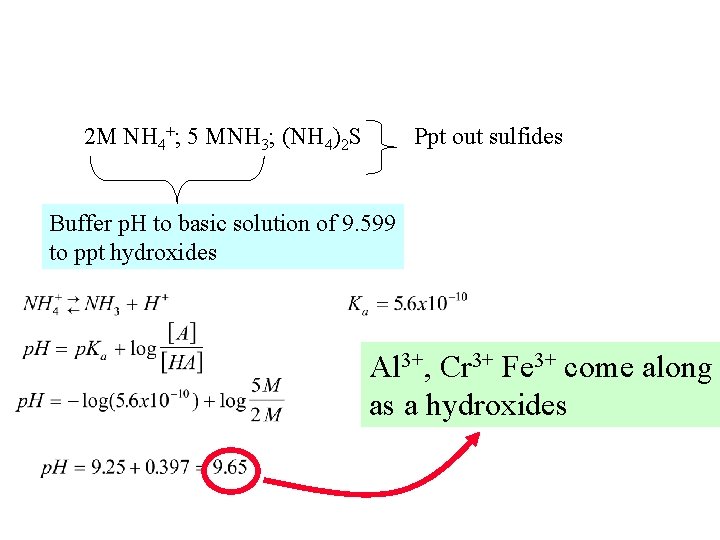
2 M NH 4+; 5 MNH 3; (NH 4)2 S Ppt out sulfides Buffer p. H to basic solution of 9. 599 to ppt hydroxides Al 3+, Cr 3+ Fe 3+ come along as a hydroxides
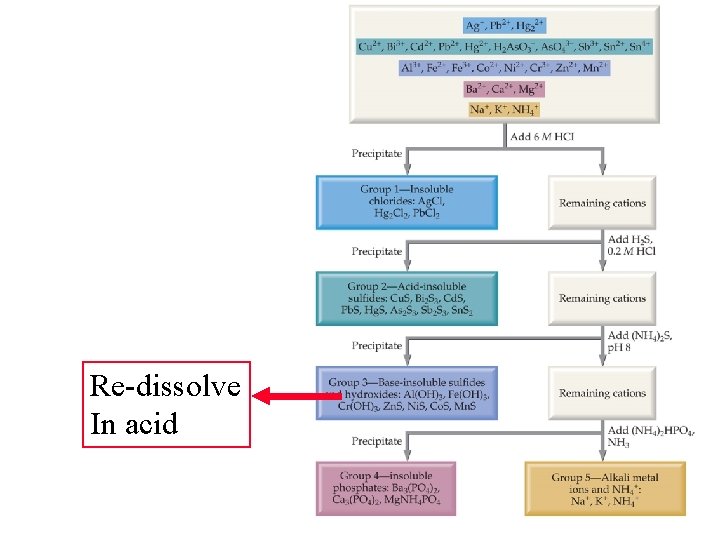
Re-dissolve In acid
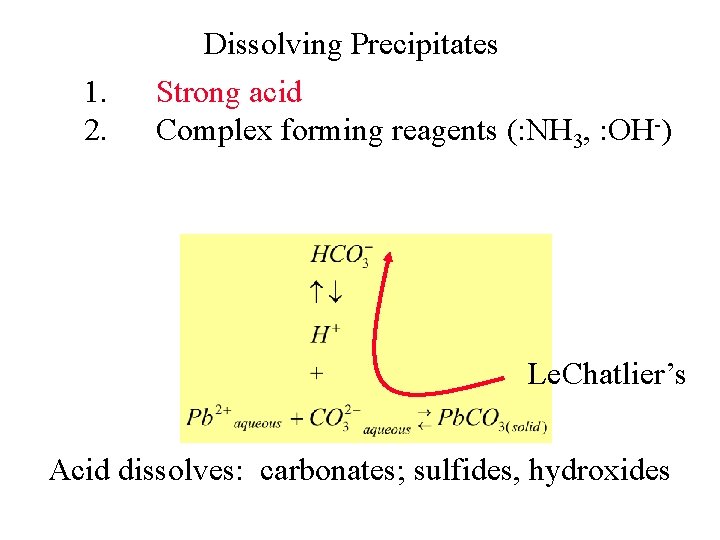
1. 2. Dissolving Precipitates Strong acid Complex forming reagents (: NH 3, : OH-) Le. Chatlier’s Acid dissolves: carbonates; sulfides, hydroxides

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Qualitative Analysis: Separating Zn 2+, And Al 3+, from the sulfides By OH complexation
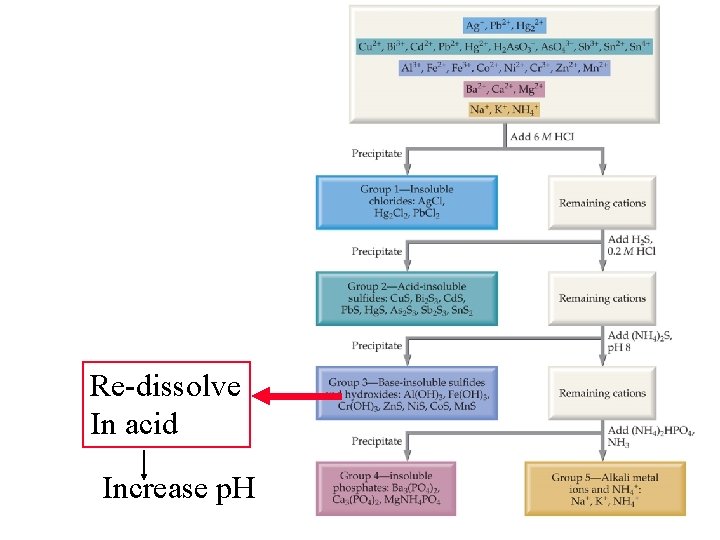
Re-dissolve In acid Increase p. H

Example Calculation 6: Ag+ Zn 2+ Mn 2+ Cr 3+ Al 3+ Fe 3+ Sn 2+ Ag(OH)2 Zn(OH)42 Mn(OH)+ Cr(OH)2+ Al(OH)4 Fe(OH)2+ Sn(OH)3 - Kf 1 x 104 4. 6 x 1017 2. 51 x 103 6. 3 x 103 1 x 1033 1. 99 x 1022 2. 5 x 1025 Ksp Ag(OH) 1. 99 x 10 -9 Zn(OH)2 3. 16 x 10 -16 Mn(OH)2(s) 1. 58 x 10 -13 Cr(OH)3(s) 6. 3 x 10 -31 Al(OH)3(s) 4. 6 x 10 -33 Fe(OH)3(s) 4 x 10 -38 Sn(OH)2(s) 1. 2 x 10 -17 Ksp. Kf 1. 99 x 10 -5 145 3. 96 x 10 -10 1. 28 4. 6 7. 96 x 10 -16 3. 0 x 108 Which compounds can be brought into solution by raising the p. H?
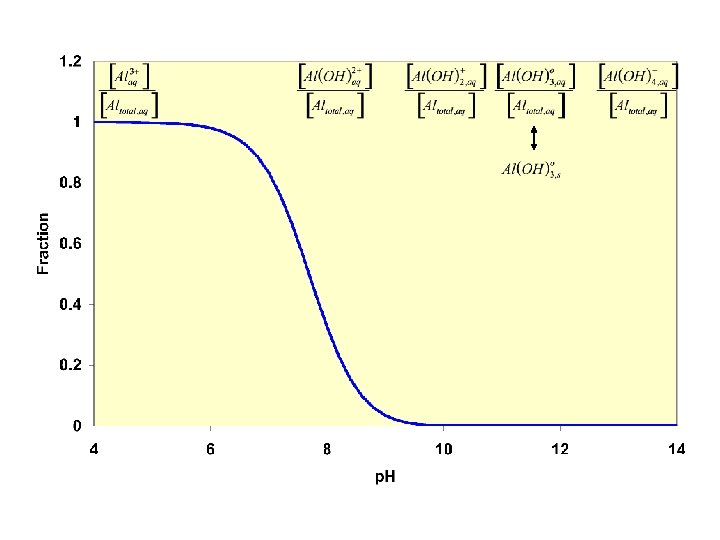

How do we distinguish Zn from Al? Between Al(OH)4 - and Zn(OH)4 only Zn has complexation with : NH 3 Drop p. H to re-ppt Al(OH)3, while simultaneously forming soluble Zn(NH 3)42+ 3 12 M NH 4+ M NH 3

1. 2. Dissolving Precipitates Strong acid Complex forming reagents (: NH 3, : OH-)
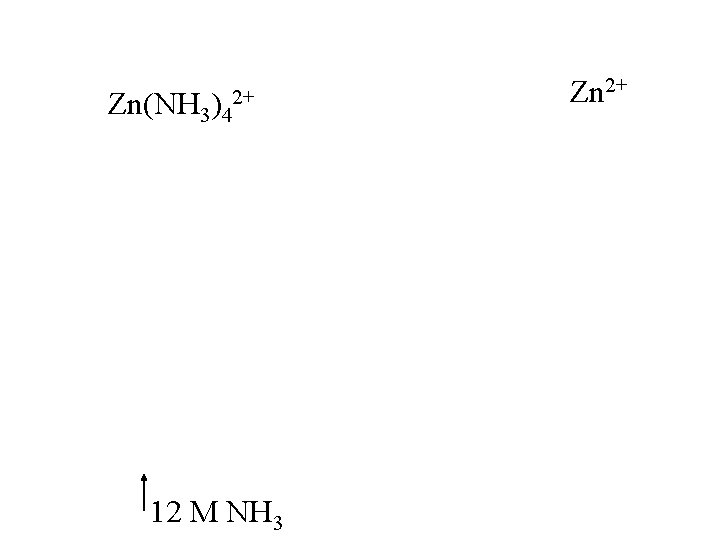
Zn(NH 3)42+ 12 M NH 3 Zn 2+

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #19: Precipitation Reactions Qualitative Analysis: PO 43 - to separate Ba 2+, Ca+2, Mg 2+
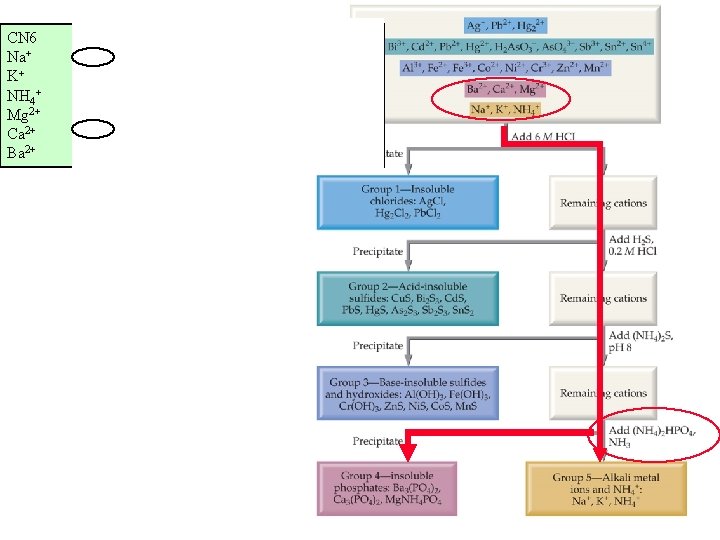
CN 6 Na+ K+ NH 4+ Mg 2+ Ca 2+ Ba 2+ r(pm) 116 152 q/r 0. 0086 0. 0065 - Ksp - 86 114 149 0. 023 0. 0175 0. 013 Ca 3(PO 4)2 Ba 3(PO 4)2 1. 2 x 10 -29 3. 4 x 10 -23

“A” students work (without solutions manual) ~7 problems/night. Solubility What you need To know

Summary Points Complexation vs Solubility Complexation based on electrostatic attraction lone pairs for central cation Size matters!; Charge matters! Can result in multiple points of binding Can result in charge -, o, + Based on charge can result in precipitation Use to manipulate and separate elements (biology too!) Ksp = solubility product; large - # implies not soluble Kf = formation constant; large +# implies strong binding Calculations proceed similarly to Ka EXCEPT – STOICHIOMETRY is trickier Ba 3(PO 4)2

“A” students work (without solutions manual) ~7 problems/night. END
- Slides: 91