A students work without solutions manual 10 problemsnight

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base Visualization Review Charge Density

Chemistry General FITCH Rules G 1: Suzuki is Success G 2. Slow me down G 3. Scientific Knowledge is Referential G 4. Watch out for Red Herrings G 5. Chemists are Lazy C 1. It’s all about charge C 2. Everybody wants to “be like Mike” C 3. Size Matters C 4. Still Waters Run Deep Piranhas lurk C 5. Alpha Dogs eat first What is an alpha dog? High charge, low volume
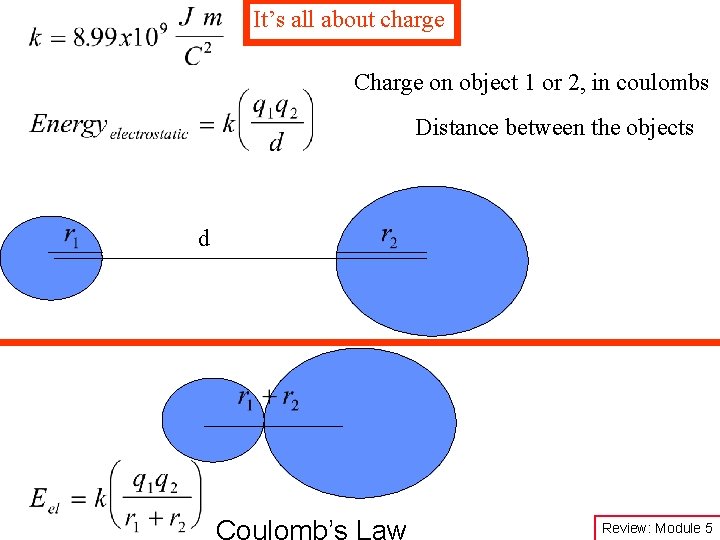
It’s all about charge Charge on object 1 or 2, in coulombs Distance between the objects d Coulomb’s Law Review: Module 5

Galen, 170 Marie the Jewess, 300 Charles Augustin James Watt Coulomb 1735 -1806 1736 -1819 Justus von Thomas Graham Liebig (1803 -1873 1805 -1869 Ludwig Boltzman 1844 -1906 Gilbert N Lewis 1875 -1946 Henri Louis Le. Chatlier 1850 -1936 Johannes Bronsted 1879 -1947 Jabir ibn Hawan, 721 -815 Luigi Galvani 1737 -1798 Richard AC E Erlenmeyer 1825 -1909 An alchemist Count Alessandro G A A Volta, 1747 -1827 James Joule (1818 -1889) Henri Bequerel 1852 -1908 Lawrence Henderson 1878 -1942 Galileo Galili Evangelista Torricelli 1564 -1642 1608 -1647 Amedeo Avogadro 1756 -1856 Rudolph Clausius 1822 -1888 Jacobus van’t Hoff 1852 -1911 Niels Bohr 1885 -1962 John Dalton 1766 -1844 William Thompson Lord Kelvin, 1824 -1907 Johannes Rydberg 1854 -1919 William Henry 1775 -1836 Johann Balmer 1825 -1898 J. J. Thomson 1856 -1940 Erwin Schodinger Louis de Broglie 1887 -1961 (1892 -1987) Fitch Rule G 3: Science is Referential Jean Picard 1620 -1682 Jacques Charles 1778 -1850 Francois-Marie Raoult 1830 -1901 Heinrich R. Hertz, 1857 -1894 Friedrich H. Hund 1896 -1997 Daniel Fahrenheit 1686 -1737 Max Planck 1858 -1947 Rolf Sievert, 1896 -1966 Blaise Pascal 1623 -1662 Georg Simon Ohm 1789 -1854 James Maxwell 1831 -1879 Robert Boyle, 1627 -1691 Isaac Newton 1643 -1727 Michael Faraday 1791 -1867 B. P. Emile Clapeyron 1799 -1864 Dmitri Mendeleev 1834 -1907 Svante Arrehenius Walther Nernst 1859 -1927 1864 -1941 Fritz London 1900 -1954 Wolfgang Pauli 1900 -1958 Johannes D. Van der Waals 1837 -1923 Marie Curie 1867 -1934 Anders Celsius 1701 -1744 Germain Henri Hess 1802 -1850 J. Willard Gibbs 1839 -1903 Fritz Haber 1868 -1934 Thomas M Lowry 1874 -1936 Werner Karl Linus Pauling Louis Harold Gray 1905 -1965 Heisenberg 1901 -1994 1901 -1976
Repulsion It Takes energy to push these guys together Positive energy + Attraction Negative energy + + -

Effect of ionic charge repulsive 4+ M 4+---M 4+ M+3—M 3+ M+2—M 2+ M+3—M 3+ M-2—M 2+ M+2—M 2+ M-3—M 3+ 1+--M+1 M 4+ M 4 ----M attractive Review: Module 5 As Charge increases The repulsive energy of like charged Or attractive energy of differently charged Ions increases It’s all about charge +1 charged ions are “losers” compared to more highly charged ions
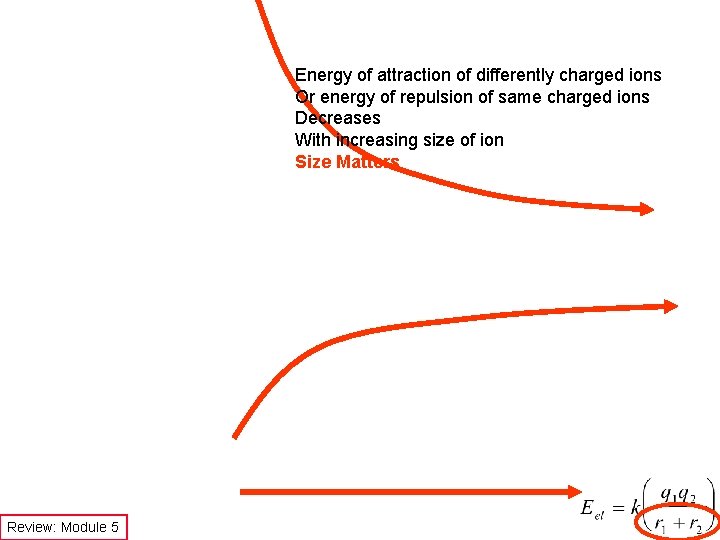
Energy of attraction of differently charged ions Or energy of repulsion of same charged ions Decreases With increasing size of ion Size Matters Review: Module 5
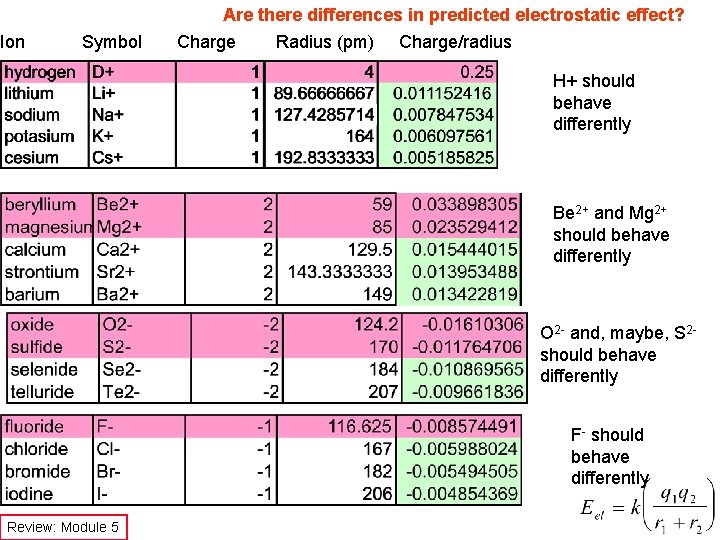
Ion Symbol Are there differences in predicted electrostatic effect? Charge Radius (pm) Charge/radius H+ should behave differently Be 2+ and Mg 2+ should behave differently O 2 - and, maybe, S 2 should behave differently F- should behave differently Review: Module 5
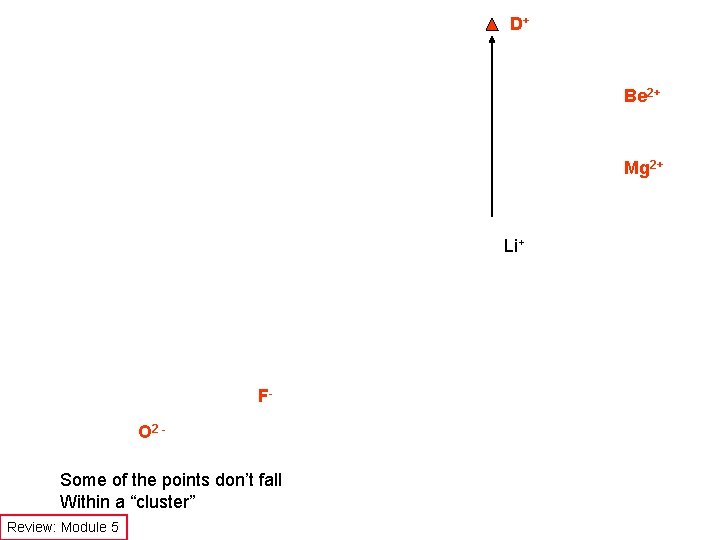
D+ Be 2+ Mg 2+ Li+ FO 2 Some of the points don’t fall Within a “cluster” Review: Module 5
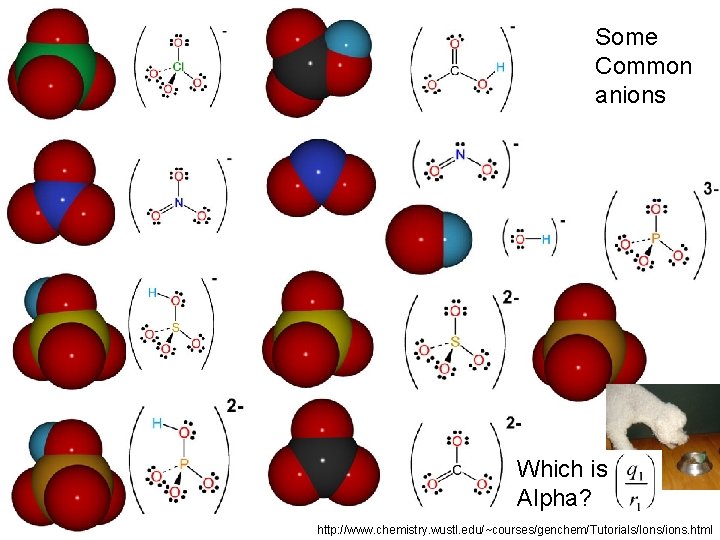
Some Common anions Which is Alpha? http: //www. chemistry. wustl. edu/~courses/genchem/Tutorials/Ions/ions. html
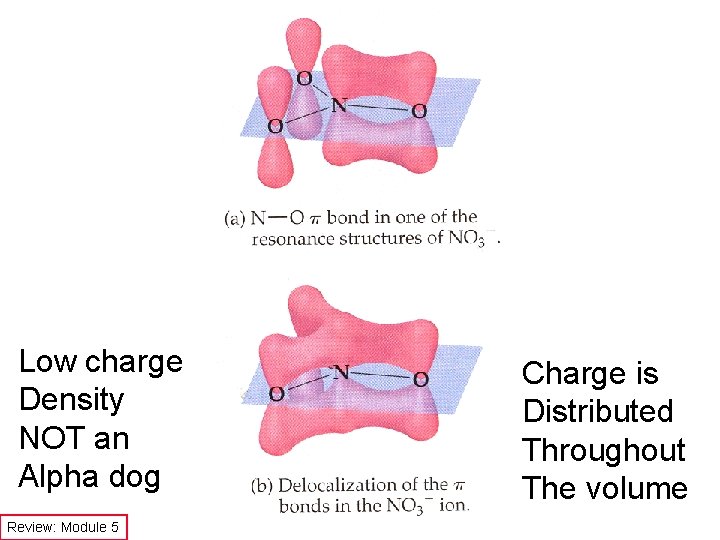
Low charge Density NOT an Alpha dog Review: Module 5 Charge is Distributed Throughout The volume

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base Visualization Review Biological Dependence on Acid/base reactions
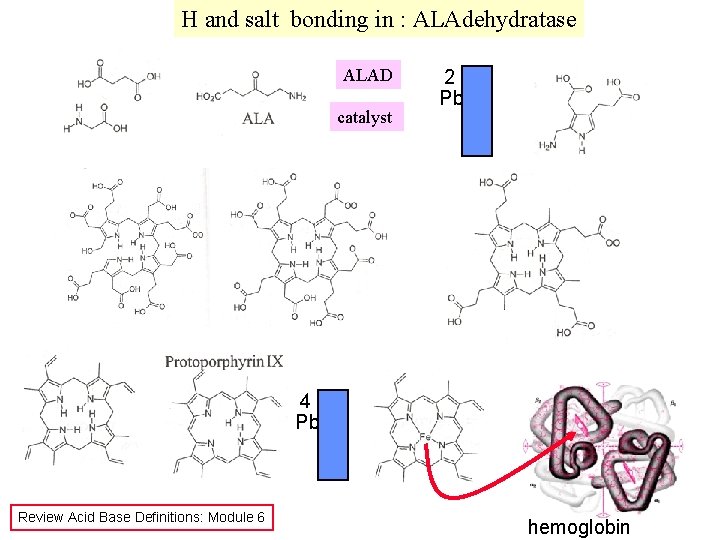
H and salt bonding in : ALAdehydratase ALAD catalyst 2 Pb 4 Pb Review Acid Base Definitions: Module 6 hemoglobin
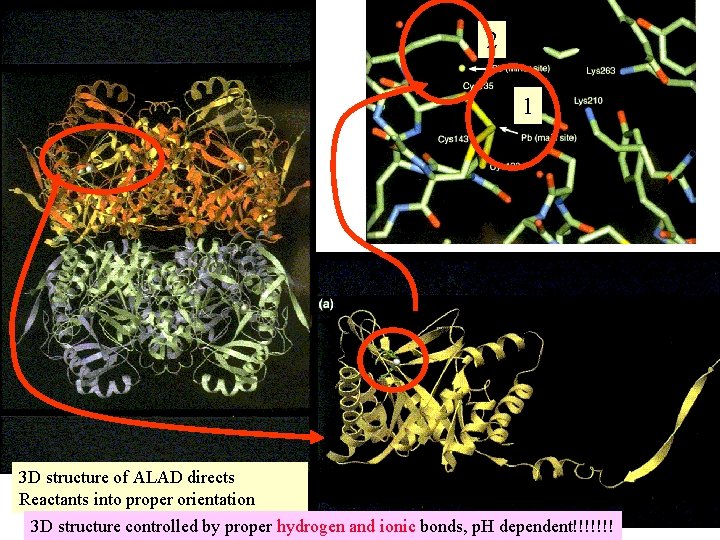
2 1 3 D structure of ALAD directs Reactants into proper orientation 3 D structure controlled by proper hydrogen and ionic bonds, p. H dependent!!!!!!!
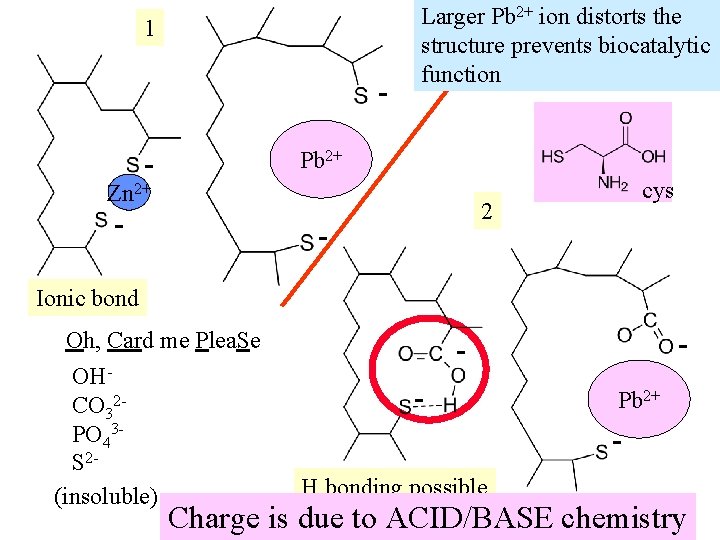
1 - Larger Pb 2+ ion distorts the structure prevents biocatalytic function Pb 2+ Zn 2+ - cys 2 - Ionic bond Oh, Card me Plea. Se OHCO 32 PO 43 S 2(insoluble) - - Pb 2+ H bonding possible Charge is due to ACID/BASE chemistry
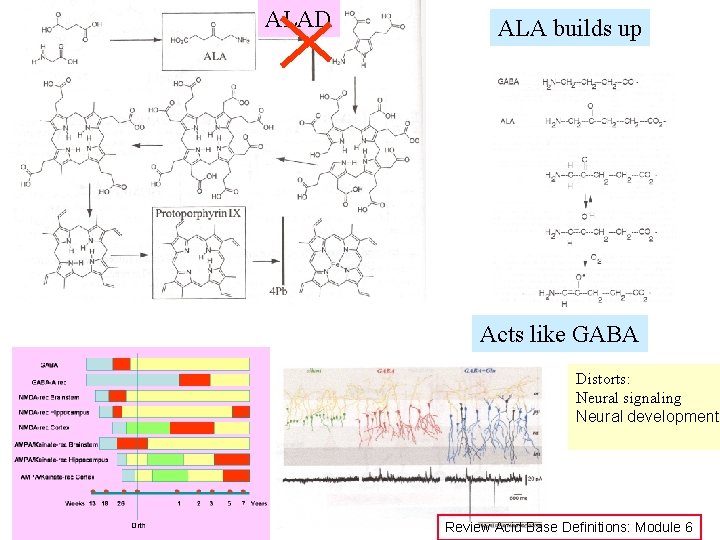
ALAD ALA builds up Acts like GABA Distorts: Neural signaling Neural development Review Acid Base Definitions: Module 6
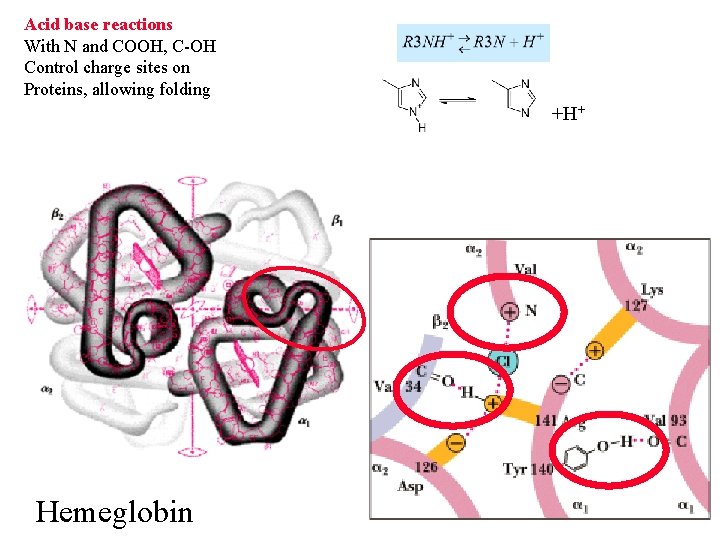
Acid base reactions With N and COOH, C-OH Control charge sites on Proteins, allowing folding +H+ Hemeglobin

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base Visualization Review Acid/base definitions
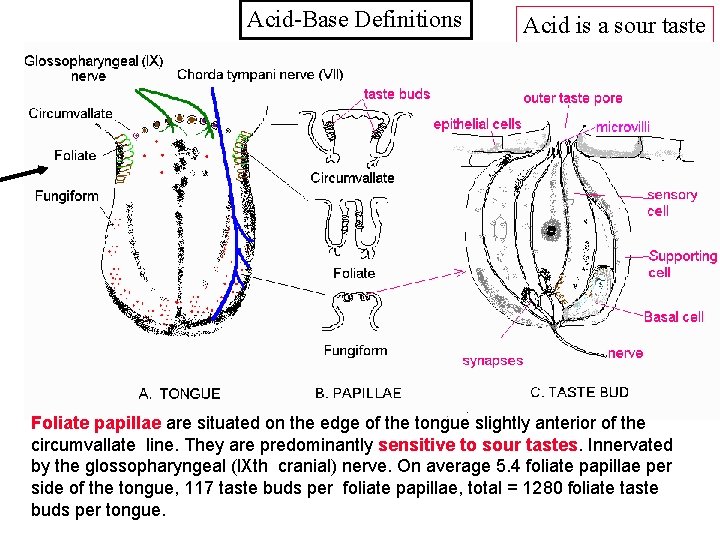
Acid-Base Definitions Acid is a sour taste Foliate papillae are situated on the edge of the tongue slightly anterior of the circumvallate line. They are predominantly sensitive to sour tastes. Innervated by the glossopharyngeal (l. Xth cranial) nerve. On average 5. 4 foliate papillae per side of the tongue, 117 taste buds per foliate papillae, total = 1280 foliate taste buds per tongue.
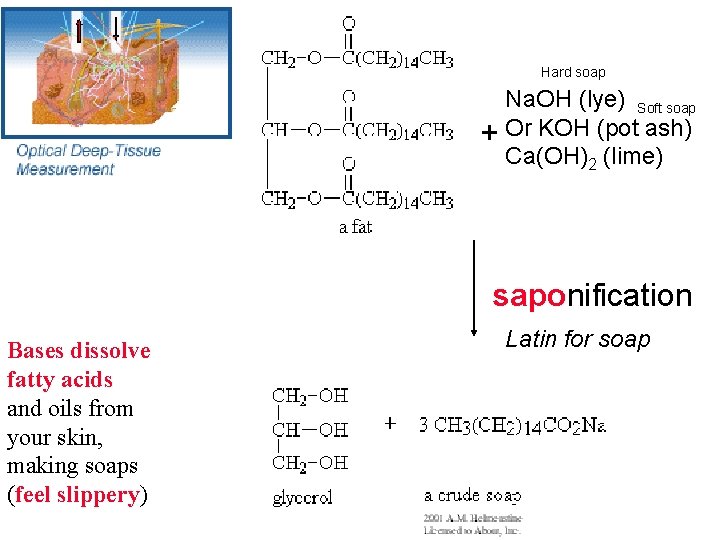
Hard soap Na. OH (lye) Soft soap + Or KOH (pot ash) Ca(OH)2 (lime) saponification Bases dissolve fatty acids and oils from your skin, making soaps (feel slippery) Latin for soap

In 1849 a Harvard chemistry professor, John Webster argued with a colleague, Dr. Parkman, about money he owed the colleague for some dinosaur bones. He killed his fellow Professor and attempted to hide the crime by dissolving the body in a vat Of “lime” (Ca(OH)2) and then burning the remaining bones. Review Acid Base Definitions: Module 6

Chemical Definition of Acid/Base Svante Arrhenius An acid is a species that produces proton ions in solution Solution X- or A- is an anion who is partnered with the cation, H+ Acid: HX Strong acid Weak acid In a STRONG acid the anion can not provide enough electrostatic attraction to hold onto the H+ (proton) In a WEAK acid the anion is very charge dense and holds onto the H+

Galen, 170 Marie the Jewess, 300 Charles Augustin James Watt Coulomb 1735 -1806 1736 -1819 Justus von Thomas Graham Liebig (1803 -1873 1805 -1869 Ludwig Boltzman 1844 -1906 Gilbert N Lewis 1875 -1946 Henri Louis Le. Chatlier 1850 -1936 Johannes Bronsted 1879 -1947 Jabir ibn Hawan, 721 -815 Luigi Galvani 1737 -1798 Richard AC E Erlenmeyer 1825 -1909 An alchemist Count Alessandro G A A Volta, 1747 -1827 James Joule (1818 -1889) Henri Bequerel 1852 -1908 Lawrence Henderson 1878 -1942 Galileo Galili Evangelista Torricelli 1564 -1642 1608 -1647 Amedeo Avogadro 1756 -1856 Rudolph Clausius 1822 -1888 Jacobus van’t Hoff 1852 -1911 Niels Bohr 1885 -1962 John Dalton 1766 -1844 William Thompson Lord Kelvin, 1824 -1907 Johannes Rydberg 1854 -1919 William Henry 1775 -1836 Johann Balmer 1825 -1898 J. J. Thomson 1856 -1940 Erwin Schodinger Louis de Broglie 1887 -1961 (1892 -1987) Fitch Rule G 3: Science is Referential Jean Picard 1620 -1682 Jacques Charles 1778 -1850 Francois-Marie Raoult 1830 -1901 Heinrich R. Hertz, 1857 -1894 Friedrich H. Hund 1896 -1997 Daniel Fahrenheit 1686 -1737 Max Planck 1858 -1947 Rolf Sievert, 1896 -1966 Blaise Pascal 1623 -1662 Georg Simon Ohm 1789 -1854 James Maxwell 1831 -1879 Robert Boyle, 1627 -1691 Isaac Newton 1643 -1727 Michael Faraday 1791 -1867 B. P. Emile Clapeyron 1799 -1864 Dmitri Mendeleev 1834 -1907 Svante Arrehenius Walther Nernst 1859 -1927 1864 -1941 Fritz London 1900 -1954 Wolfgang Pauli 1900 -1958 Johannes D. Van der Waals 1837 -1923 Marie Curie 1867 -1934 Anders Celsius 1701 -1744 Germain Henri Hess 1802 -1850 J. Willard Gibbs 1839 -1903 Fritz Haber 1868 -1934 Thomas M Lowry 1874 -1936 Werner Karl Linus Pauling Louis Harold Gray 1905 -1965 Heisenberg 1901 -1994 1901 -1976
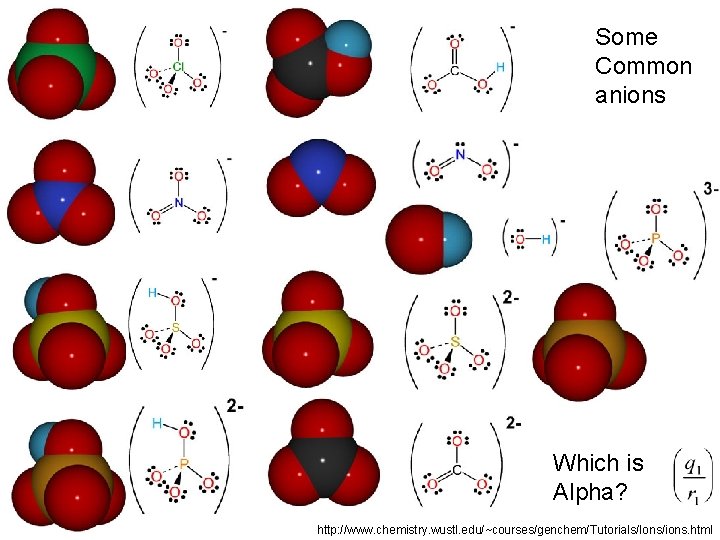
Some Common anions Which is Alpha? http: //www. chemistry. wustl. edu/~courses/genchem/Tutorials/Ions/ions. html

We saw that q/r > ~0. 08 seemed to be “big” Which of these anions are not “alpha dogs” No Clean Socks Oh Card Me Plea. Se
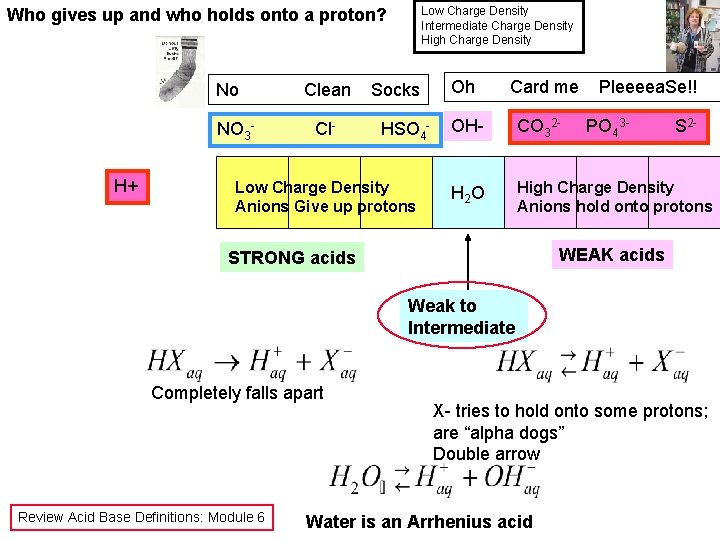
Low Charge Density Intermediate Charge Density High Charge Density Who gives up and who holds onto a proton? No NO 3 H+ Clean Cl- Socks HSO 4 - Low Charge Density Anions Give up protons Oh Card me Pleeeea. Se!! OH- CO 32 - H 2 O High Charge Density Anions hold onto protons PO 43 - S 2 - WEAK acids STRONG acids Weak to Intermediate Completely falls apart Review Acid Base Definitions: Module 6 X- tries to hold onto some protons; are “alpha dogs” Double arrow Water is an Arrhenius acid
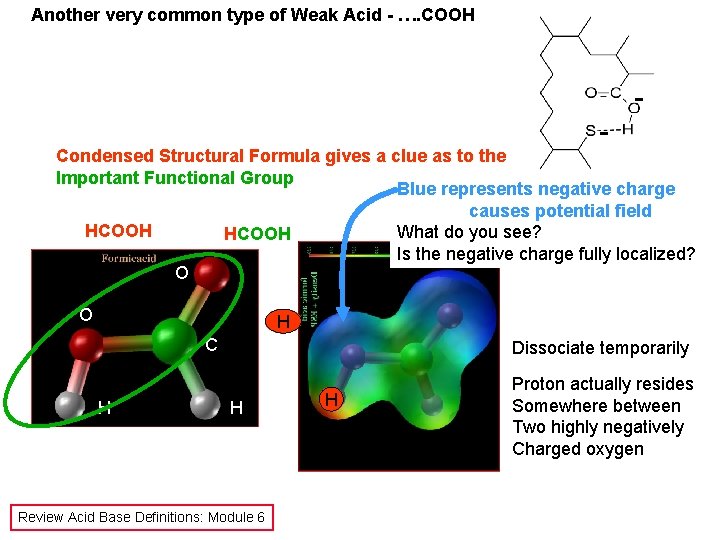
Another very common type of Weak Acid - …. COOH - - Condensed Structural Formula gives a clue as to the Important Functional Group Blue represents negative charge causes potential field HCOOH What do you see? HCOOH Is the negative charge fully localized? O O H C H Dissociate temporarily H Review Acid Base Definitions: Module 6 H Proton actually resides Somewhere between Two highly negatively Charged oxygen
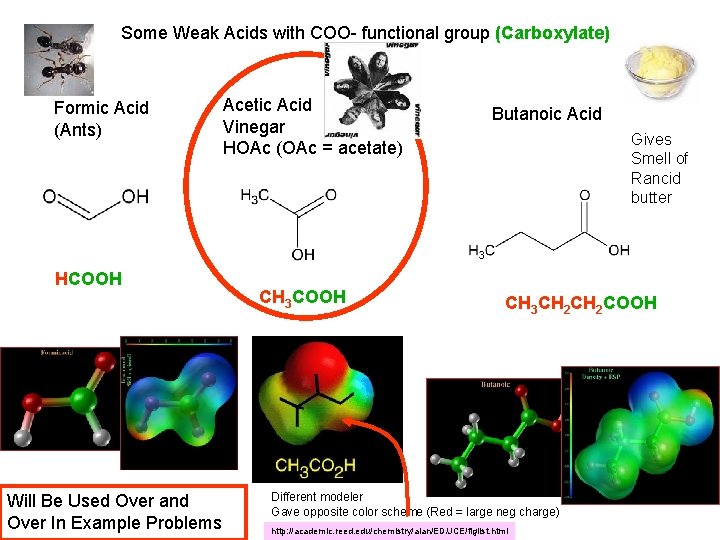
Some Weak Acids with COO- functional group (Carboxylate) Formic Acid (Ants) HCOOH Will Be Used Over and Over In Example Problems Acetic Acid Vinegar HOAc (OAc = acetate) CH 3 COOH Butanoic Acid Gives Smell of Rancid butter CH 3 CH 2 COOH Different modeler Gave opposite color scheme (Red = large neg charge) http: //academic. reed. edu/chemistry/alan/ED/JCE/figlist. html
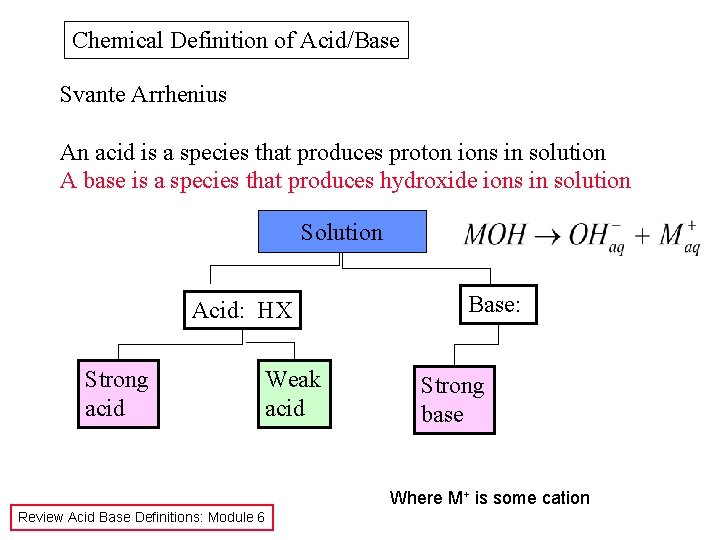
Chemical Definition of Acid/Base Svante Arrhenius An acid is a species that produces proton ions in solution A base is a species that produces hydroxide ions in solution Solution Acid: HX Strong acid Weak acid Base: Strong base Where M+ is some cation Review Acid Base Definitions: Module 6

Strong Bases (SB) hydroxides which have low charge density cations as partners 1. q is low (+1) Group 1 (Li+, Na+, K+) 2. r is large Group 2 cations Be 2+ Mg 2+ Ca 2+ Sr 2+ Ba 2+ Ra 2+ But only larger ones Not Ra 2+ because dispersion forces come into play
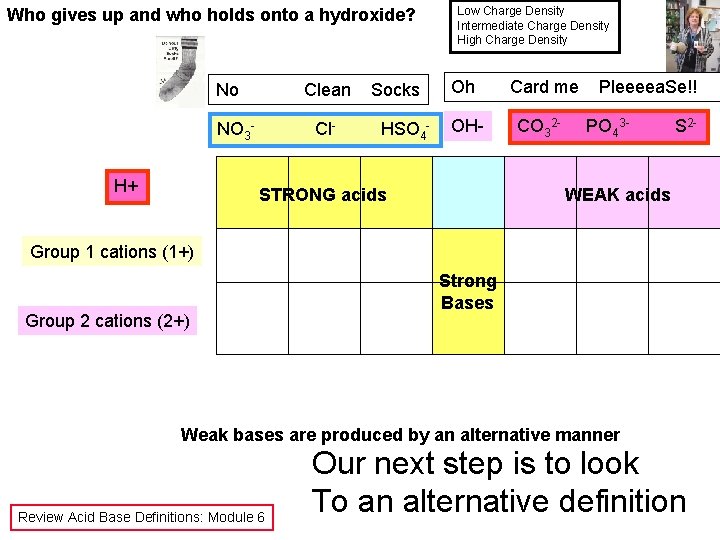
Who gives up and who holds onto a hydroxide? No Clean NO 3 H+ Cl- Socks HSO 4 - Low Charge Density Intermediate Charge Density High Charge Density Oh Card me OH- CO 32 - STRONG acids Pleeeea. Se!! PO 43 - S 2 - WEAK acids Group 1 cations (1+) Group 2 cations (2+) Strong Bases Weak bases are produced by an alternative manner Review Acid Base Definitions: Module 6 Our next step is to look To an alternative definition

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base. Visualization Review More Acid Base Definitions

Bronsted and Lowry definitions B- and X- are bases (anions) which can accept protons HX and HB are species which can donate protons A proton donor Bronsted acid A proton acceptor (Bronsted base)

Galen, 170 Marie the Jewess, 300 Charles Augustin James Watt Coulomb 1735 -1806 1736 -1819 Justus von Thomas Graham Liebig (1803 -1873 1805 -1869 Ludwig Boltzman 1844 -1906 Gilbert N Lewis 1875 -1946 Henri Louis Le. Chatlier 1850 -1936 Johannes Bronsted 1879 -1947 Jabir ibn Hawan, 721 -815 Luigi Galvani 1737 -1798 Richard AC E Erlenmeyer 1825 -1909 An alchemist Count Alessandro G A A Volta, 1747 -1827 James Joule (1818 -1889) Henri Bequerel 1852 -1908 Lawrence Henderson 1878 -1942 Galileo Galili Evangelista Torricelli 1564 -1642 1608 -1647 Amedeo Avogadro 1756 -1856 Rudolph Clausius 1822 -1888 Jacobus van’t Hoff 1852 -1911 Niels Bohr 1885 -1962 John Dalton 1766 -1844 William Thompson Lord Kelvin, 1824 -1907 Johannes Rydberg 1854 -1919 William Henry 1775 -1836 Johann Balmer 1825 -1898 J. J. Thomson 1856 -1940 Erwin Schodinger Louis de Broglie 1887 -1961 (1892 -1987) Fitch Rule G 3: Science is Referential Jean Picard 1620 -1682 Jacques Charles 1778 -1850 Francois-Marie Raoult 1830 -1901 Heinrich R. Hertz, 1857 -1894 Friedrich H. Hund 1896 -1997 Daniel Fahrenheit 1686 -1737 Max Planck 1858 -1947 Rolf Sievert, 1896 -1966 Blaise Pascal 1623 -1662 Georg Simon Ohm 1789 -1854 James Maxwell 1831 -1879 Robert Boyle, 1627 -1691 Isaac Newton 1643 -1727 Michael Faraday 1791 -1867 B. P. Emile Clapeyron 1799 -1864 Dmitri Mendeleev 1834 -1907 Svante Arrehenius Walther Nernst 1859 -1927 1864 -1941 Fritz London 1900 -1954 Wolfgang Pauli 1900 -1958 Johannes D. Van der Waals 1837 -1923 Marie Curie 1867 -1934 Anders Celsius 1701 -1744 Germain Henri Hess 1802 -1850 J. Willard Gibbs 1839 -1903 Fritz Haber 1868 -1934 Thomas M Lowry 1874 -1936 Werner Karl Linus Pauling Louis Harold Gray 1905 -1965 Heisenberg 1901 -1994 1901 -1976

Water can be one of the conjugate pairs The acid HX donates a proton to water it is a proton donor = acid The water molecule takes a proton from HX it is a proton acceptor = base Water is a Bronsted-Lowry Base
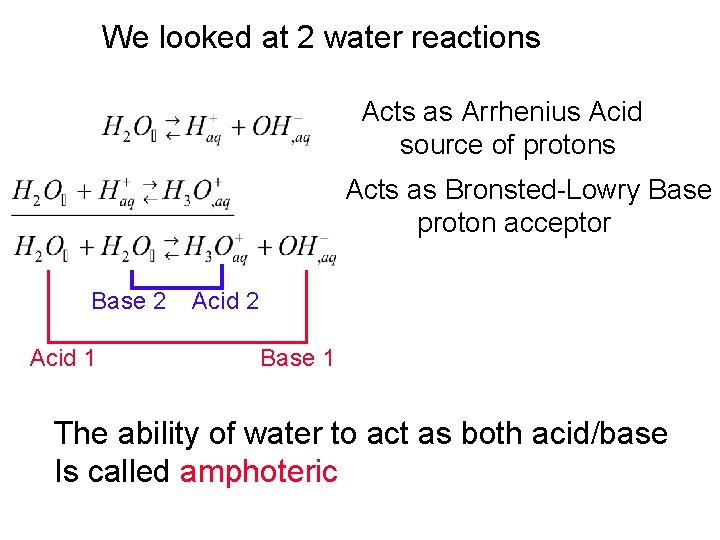
We looked at 2 water reactions Acts as Arrhenius Acid source of protons Acts as Bronsted-Lowry Base proton acceptor Base 2 Acid 1 Acid 2 Base 1 The ability of water to act as both acid/base Is called amphoteric

Some new vocabulary For Bronsted-Lowry Acid/Base Reactions

Mass balance for B 1. Occurs as both a “base” and “acid” form 2. The 2 forms are “linked” by the reaction 3. The 2 forms are “conjugated” 4. The anion B- is the “conjugate” base of HB Mass balance for X 1. Occurs as both a “base” and “acid” form 2. The 2 forms are “linked” by the reaction 3. The 2 forms are “conjugated” 4. The HX is the conjugate acid of X-
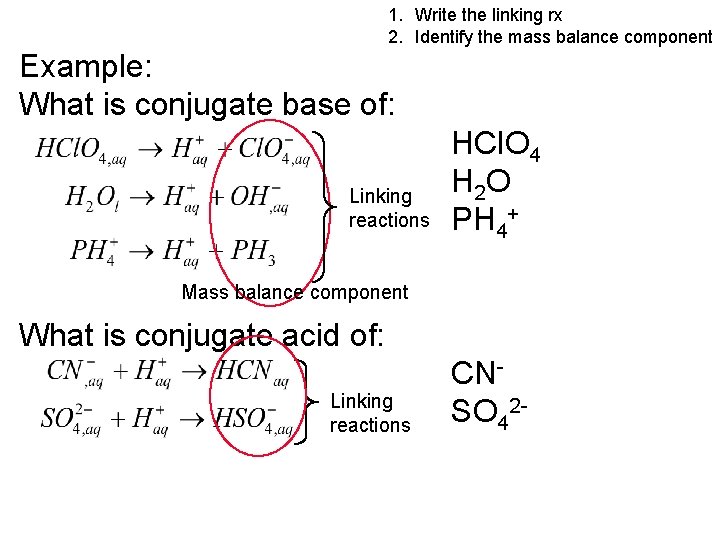
1. Write the linking rx 2. Identify the mass balance component Example: What is conjugate base of: Linking reactions HCl. O 4 H 2 O PH 4+ Mass balance component What is conjugate acid of: Linking reactions CNSO 42 -
Notice, that some anions occur in multiple rx
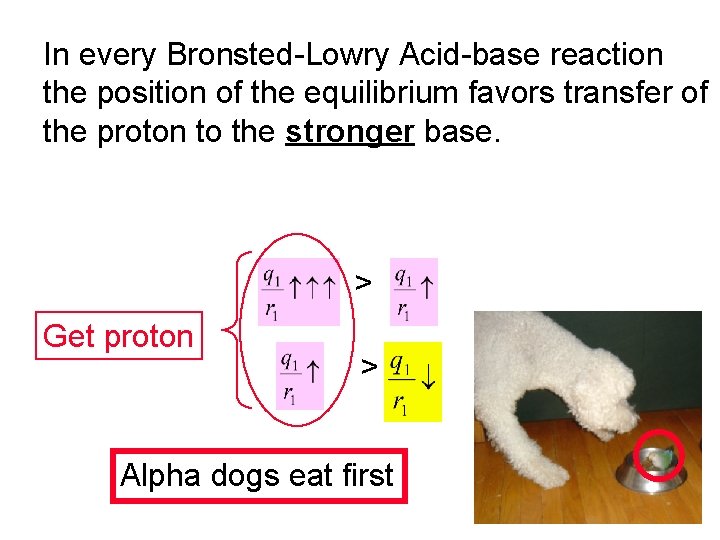
In every Bronsted-Lowry Acid-base reaction the position of the equilibrium favors transfer of the proton to the stronger base. > Get proton > Alpha dogs eat first
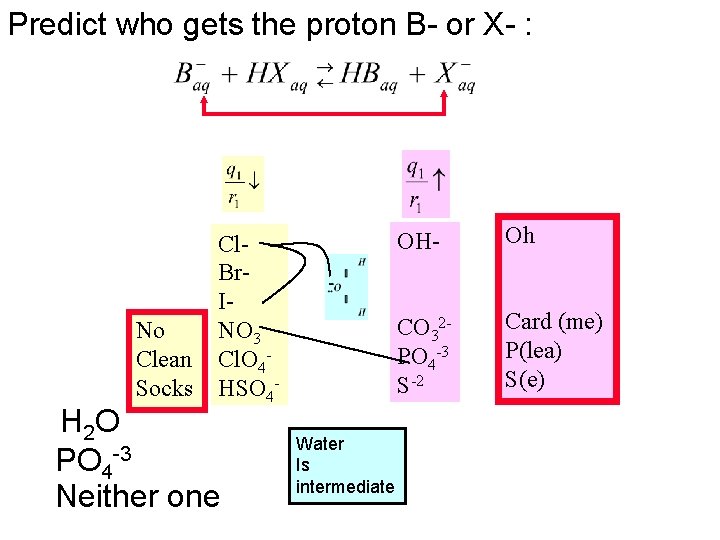
Predict who gets the proton B- or X- : No Clean Socks Cl. Br. INO 3 Cl. O 4 HSO 4 - H 2 O PO 4 -3 Neither one Water Is intermediate OH- Oh CO 32 PO 4 -3 S-2 Card (me) P(lea) S(e)

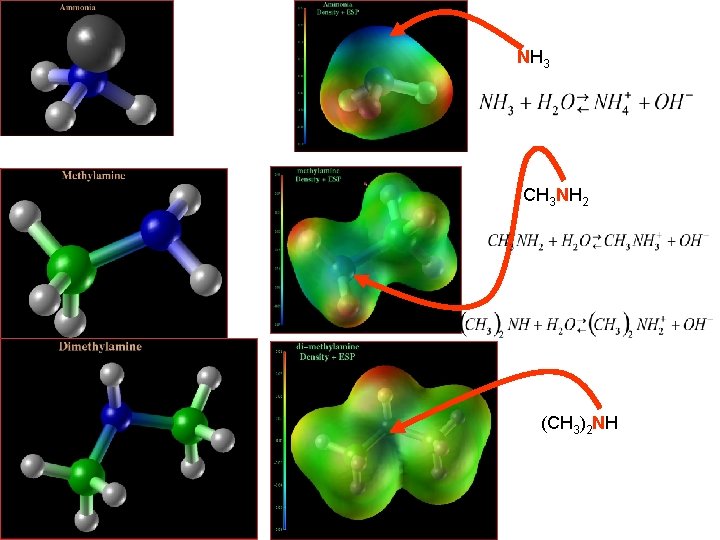
Weak bases are made by a BL reaction with water Who is a proton donor (BL acid)? Water Who is a proton acceptor (BL base)? Ammonia What functional groups are “R”?

http: //core. ecu. edu/phys/flurchickk/Atomic. Molecular. Systems/molecular. Structures. html net neutral Molecule ammonia, NH 3 Is the charge evenly distributed? Is there a region of high charge Density? In this model blue is negative charge causing a potential field and red is Positive charge opposite potential field Nitrogen typically contains a set of unbonded electrons which give Large charge density near the N atom Review Acid Base Definitions: Module 6

Write reaction to show the unbonded electrons on R (where R is N in ammonia) Write the reaction to show that electrons on R are attracted to proton on water Compounds in which H on NH 3 has been replaced by –C groups react Similarly NH 3 CH 3 NH 2 R C 2 H 5 NH 2 (C 2 H 5)2 NH C 6 H 5 NH 2
NH 3 CH 3 NH 2 (CH 3)2 NH
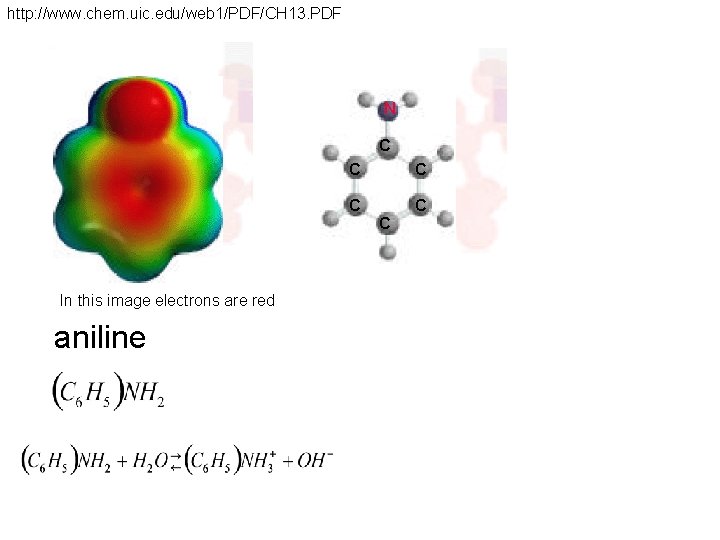
http: //www. chem. uic. edu/web 1/PDF/CH 13. PDF N C C C In this image electrons are red aniline
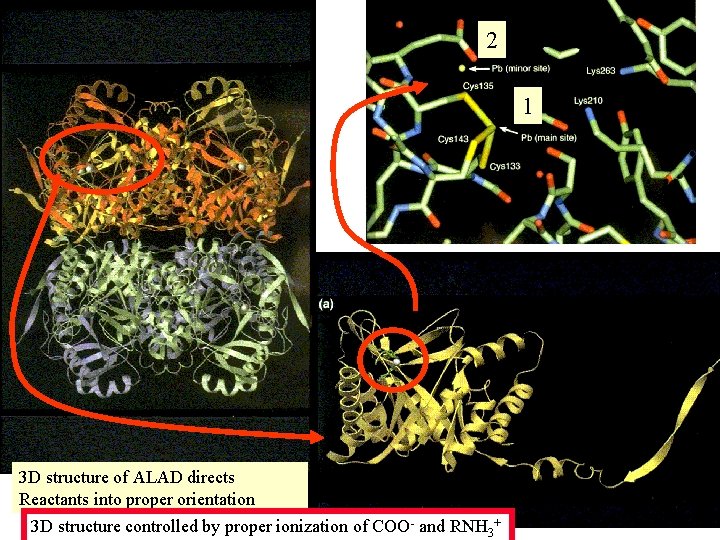
2 1 3 D structure of ALAD directs Reactants into proper orientation 3 D structure controlled by proper ionization of COO- and RNH 3+
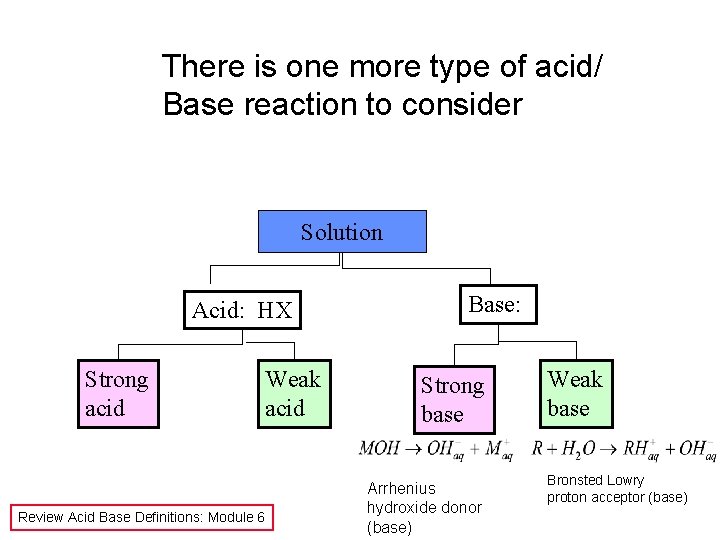
There is one more type of acid/ Base reaction to consider Solution Acid: HX Strong acid Weak acid Review Acid Base Definitions: Module 6 Base: Strong base Arrhenius hydroxide donor (base) Weak base Bronsted Lowry proton acceptor (base)

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base Visualization One more kind of Acid/Base reaction
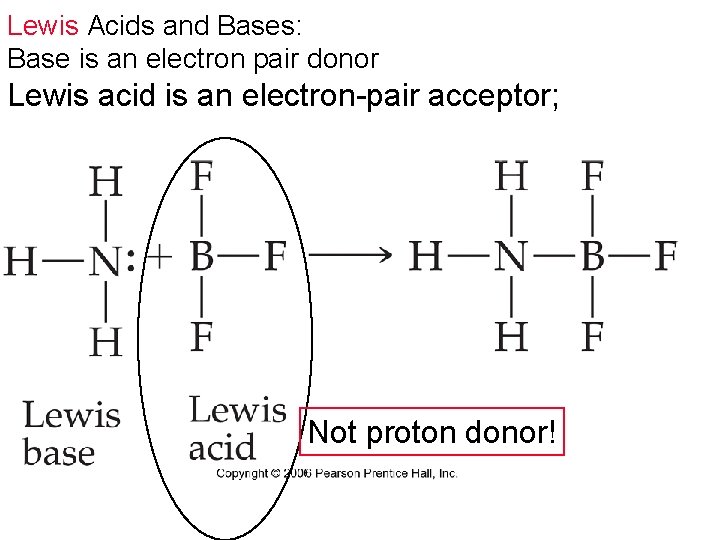
Lewis Acids and Bases: Base is an electron pair donor Lewis acid is an electron-pair acceptor; Not proton donor!

Highly charged Cations can Act as Lewis acids = stronger effect Aluminum ion is an electron pair acceptor = Lewis acid


ATOM SIZE (NOT ION) Na+ Size (A) 1. 34 Charge +1 Ca 2+ 1. 74 +2 Charge/size 0. 75 1. 15 Zn 2+ 1. 31 +2 2. 30 Produce protons Al 3+ 1. 18 +3 3. 39

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base. Visualization Salts
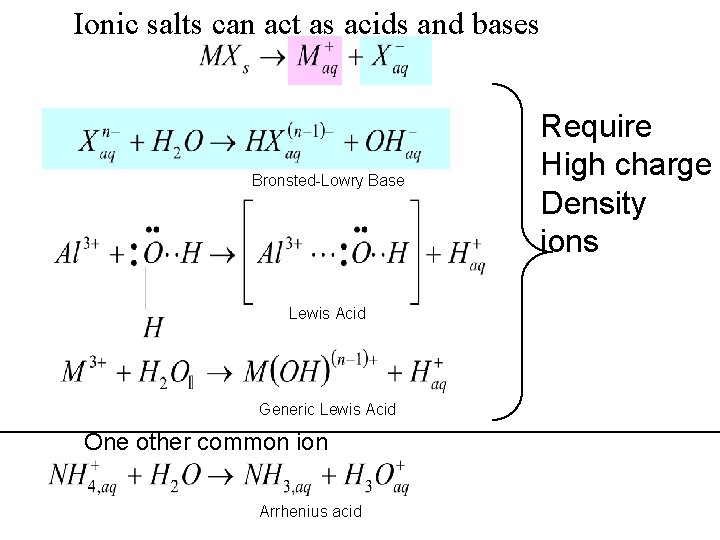
Ionic salts can act as acids and bases Bronsted-Lowry Base Lewis Acid Generic Lewis Acid One other common ion Arrhenius acid Require High charge Density ions

Predict what happens when the following salts Are added to water Na+ Cl- does not “grab” OH from water, no effect does not “grab” H from water, no effect neutral NH 4+Cl- Na+ CH 3 COO- Arrhenius Acid: forms protons acidic Bronsted Lowry Base: Proton acceptor basic

What about NH 4 F? Arrhenius Acid: forms protons acidic Bronsted Lowry Base: Proton acceptor basic Who controls the p. H? Here we need to define of our comparison numbers

Chemistry General FITCH Rules G 1: Suzuki is Success G 2. Slow me down G 3. Scientific Knowledge is Referential G 4. Watch out for Red Herrings G 5. Chemists are Lazy C 1. It’s all about charge C 2. Everybody wants to “be like Mike” C 3. Size Matters C 4. Still Waters Run Deep Piranhas lurk C 5. Alpha Dogs eat first
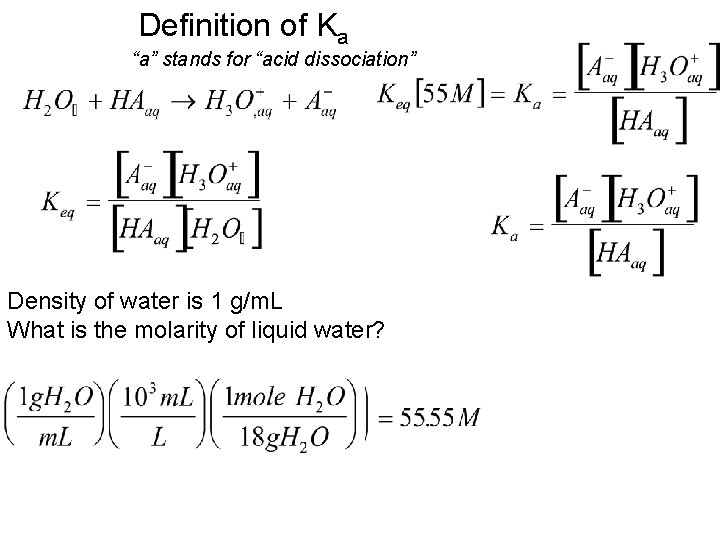
Definition of Ka “a” stands for “acid dissociation” Density of water is 1 g/m. L What is the molarity of liquid water?
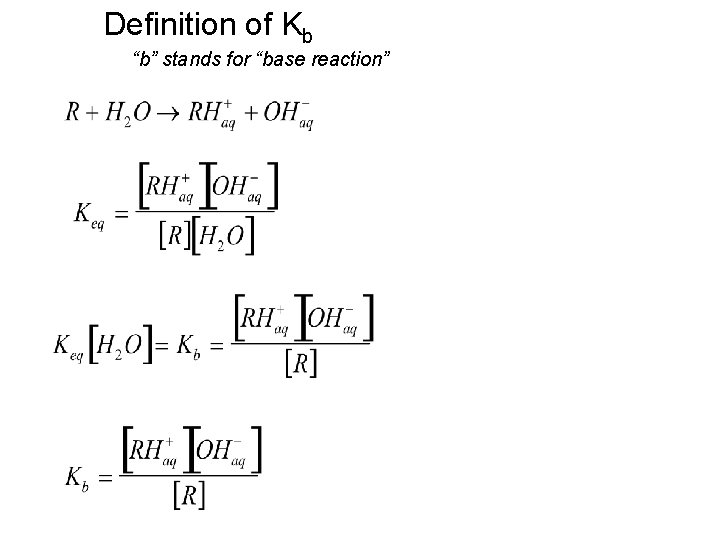
Definition of Kb “b” stands for “base reaction”

What about NH 4 F? Who controls the p. H? Since the concentrations of NH 4+ and F- are equal the reaction with the largest Keq will be in control Ammonium controls, solution goes acidic

What about NH 4 Cl. O? Who controls the p. H? Reaction with largest Keq Cl. O- controls, solution goes basic

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base. Visualization Using Ka and Kb Values for comparison

No Clean Socks Oh Card me Plea. Se It all comes together Charge density relates to Ka values!
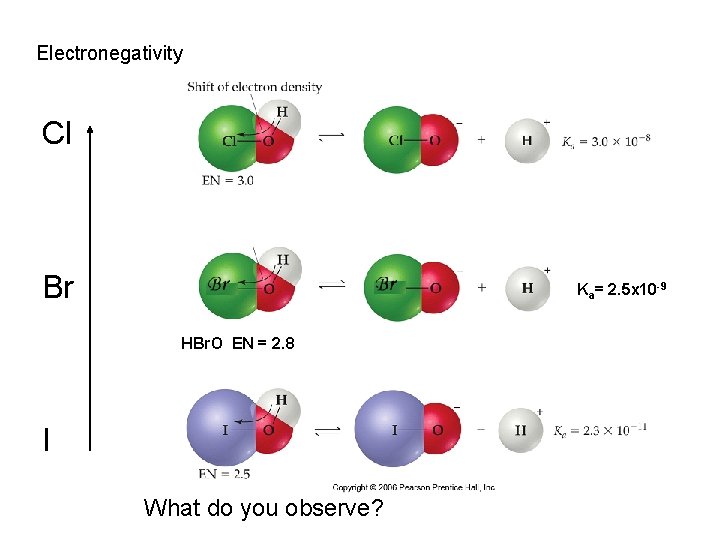
Electronegativity Cl Br Ka= 2. 5 x 10 -9 HBr. O EN = 2. 8 I What do you observe?
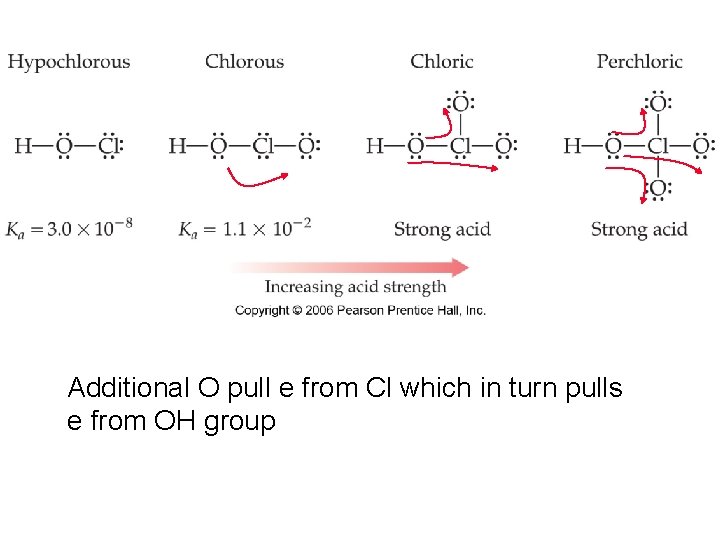
Additional O pull e from Cl which in turn pulls e from OH group
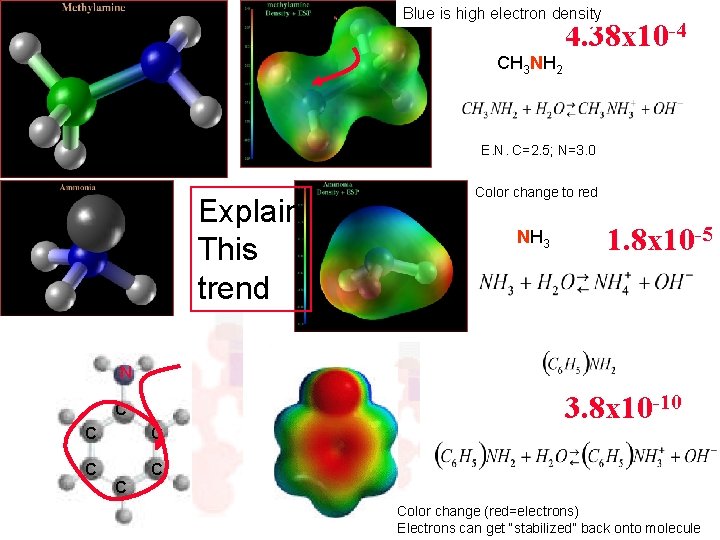
Blue is high electron density CH 3 NH 2 4. 38 x 10 -4 E. N. C=2. 5; N=3. 0 Explain This trend Color change to red NH 3 1. 8 x 10 -5 N C C C 3. 8 x 10 -10 C Color change (red=electrons) Electrons can get “stabilized” back onto molecule

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base. Visualization Polyprotic acids

Polyprotic acids can supply more than 1 proton Commonly: Ka 1 >>> Ka 2 which means commonly (but not always!) only the first one makes the proton contribution.
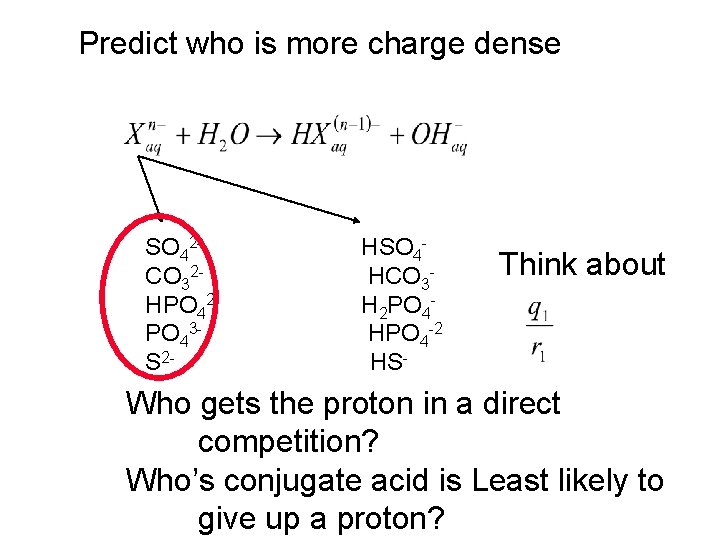
Predict who is more charge dense SO 42 CO 32 HPO 42 PO 43 S 2 - HSO 4 HCO 3 H 2 PO 4 HPO 4 -2 HS- Think about Who gets the proton in a direct competition? Who’s conjugate acid is Least likely to give up a proton?
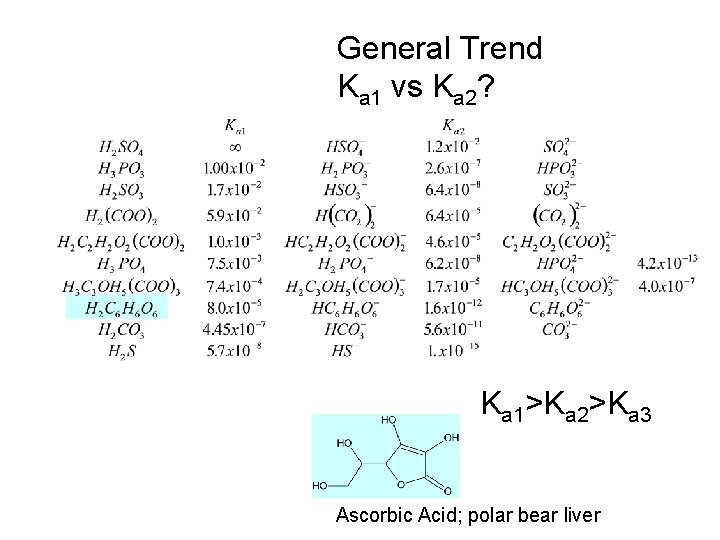
General Trend Ka 1 vs Ka 2? Ka 1>Ka 2>Ka 3 Ascorbic Acid; polar bear liver
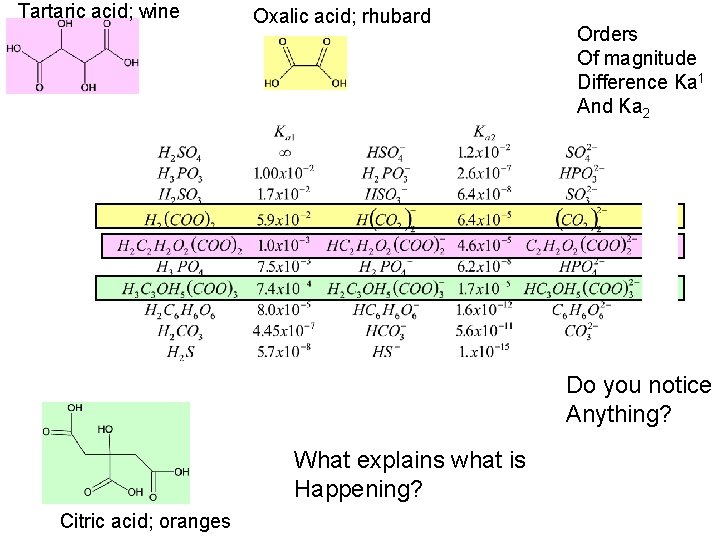
Tartaric acid; wine Oxalic acid; rhubard Orders Of magnitude Difference Ka 1 And Ka 2 Do you notice Anything? What explains what is Happening? Citric acid; oranges

“A” students work (without solutions manual) ~ 10 problems/night. Dr. Alanah Fitch Flanner Hall 402 508 -3119 afitch@luc. edu Office Hours Th&F 2 -3: 30 pm Module #17 A: Acid Base Visualization END

Who will get the proton, “A” or water? A HSO 4 NO 2 CH 3 COO- NH 3 water Ka Huge 4 x 10 -4 1. 8 x 10 -5 5. 6 x 10 -10 What do you observe? How does Ka change with structure? This kind of question WILL be on exam
- Slides: 75